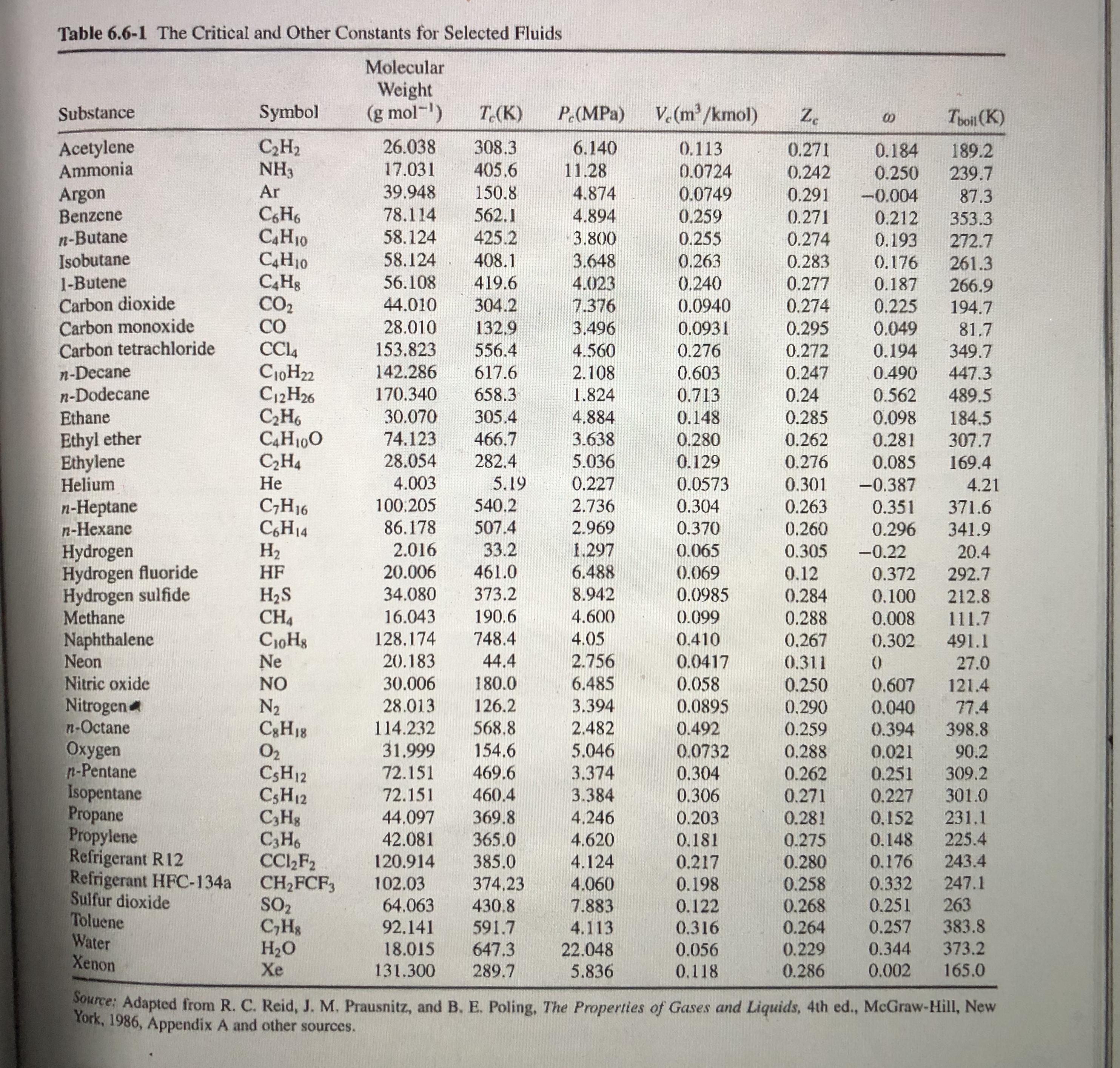
Table 6.6-1 The Critical and Other Constants for Selected Fluids Molecular Weight (g mol-) V. (m/kmol)...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
Table 6.6-1 The Critical and Other Constants for Selected Fluids Molecular Weight (g mol-) V. (m/kmol) Substance Symbol T.(K) P (MPa) Z. Toil (K) Acetylene Ammonia C,H2 NH3 26.038 308.3 405.6 6.140 0.113 0.271 0.184 0.250 189.2 17.031 11.28 0.0724 0.0749 0.242 239.7 Ar 39.948 150.8 Argon Benzene n-Butane Isobutane 1-Butene Carbon dioxide Carbon monoxide Carbon tetrachloride 4.874 0.291 -0.004 87.3 CH6 C4H10 C4H10 78,114 562.1 4.894 0.259 0.271 0.212 353.3 58.124 58.124 425.2 3.800 0.255 0.274 0.193 272.7 408.1 419.6 3.648 0.263 0.283 0.176 261.3 0.240 0.0940 56.108 4.023 0.277 0.187 266.9 194.7 CO2 CO 44.010 304.2 7.376 0.274 0.225 28.010 132,9 3.496 0.0931 0.295 0.049 81.7 349.7 153.823 556.4 4.560 0.276 0.272 0.194 n-Decane n-Dodecane Ethane C10H22 C12H26 C,H6 C4H100 C,H4 Не 142.286 617.6 2.108 0.603 0.247 0.490 447.3 170.340 658.3 1.824 0.713 0.24 0.562 489.5 30.070 305.4 4.884 0.148 0.285 0.098 184.5 307.7 Ethyl ether Ethylene Helium 74.123 466.7 3.638 5.036 0.280 0.262 0.281 28.054 282.4 0.129 0.276 0.085 169.4 4.003 5.19 0.227 0.0573 0.301 -0.387 4.21 540.2 n-Heptane n-Hexane C;H16 C,H14 H2 100.205 2.736 0.304 0.263 0.351 371.6 86.178 507.4 2.969 0.370 0.260 0.296 341.9 2.016 33.2 1.297 Hydrogen Hydrogen fluoride Hydrogen sulfide Methane Naphthalene Neon Nitric oxide Nitrogen n-Octane Oxygen n-Pentane Isopentane Propane Propylene Refrigerant R12 Refrigerant HFC-134a Sulfur dioxide Toluene Water Xenon 0.065 0.305 -0.22 20.4 | HF 20.006 461.0 6.488 (0.069 0.12 0.372 292.7 373.2 H2S CH4 CioHg Ne 34.080 8.942 0.0985 0.284 0.100 212.8 16.043 190.6 4.600 0.099 0.288 0.008 111.7 128,174 748.4 4.05 0.410 0.267 0.302 491.1 20.183 44.4 2.756 0.0417 0.058 0.0895 0.311 27.0 NO 30.006 180.0 6.485 0.250 0.607 121.4 N2 C3H18 O2 CSH12 CSH12 C,Hg C3H6 CC,F2 CH2FCF, SO2 C,Hs H2O Xe 28.013 126.2 3.394 0.290 0.040 77.4 114.232 568.8 2.482 0.492 0.259 0.288 0.394 398.8 31.999 154.6 5.046 0.0732 0.021 90.2 309.2 72.151 469.6 3.374 0.304 0.262 0.271 0.251 72.151 3.384 4,246 301.0 231.1 460.4 0.306 0.227 44.097 42.081 369.8 365.0 0.203 0.281 0,152 4.620 0.181 0.275 0.148 225.4 4.124 4.060 120.914 385.0 0.217 243.4 0.280 0.258 0.268 0.264 0.176 102.03 64.063 0.332 0.251 374,23 0.198 247.1 0.122 0.316 263 383.8 373.2 430.8 7.883 92.141 591.7 4.113 0.257 647.3 18.015 131.300 22.048 5.836 0.056 0.229 0.344 289.7 0.118 0.286 0.002 165.0 Source: Adapted from R. C. Reid, J. M. Prausnitz, and B. E. Poling, The Properties of Gases and Liquids, 4th ed., McGraw-Hill, New York, 1986, Appendix A and other sources. Table 6.6-1 The Critical and Other Constants for Selected Fluids Molecular Weight (g mol-) V. (m/kmol) Substance Symbol T.(K) P (MPa) Z. Toil (K) Acetylene Ammonia C,H2 NH3 26.038 308.3 405.6 6.140 0.113 0.271 0.184 0.250 189.2 17.031 11.28 0.0724 0.0749 0.242 239.7 Ar 39.948 150.8 Argon Benzene n-Butane Isobutane 1-Butene Carbon dioxide Carbon monoxide Carbon tetrachloride 4.874 0.291 -0.004 87.3 CH6 C4H10 C4H10 78,114 562.1 4.894 0.259 0.271 0.212 353.3 58.124 58.124 425.2 3.800 0.255 0.274 0.193 272.7 408.1 419.6 3.648 0.263 0.283 0.176 261.3 0.240 0.0940 56.108 4.023 0.277 0.187 266.9 194.7 CO2 CO 44.010 304.2 7.376 0.274 0.225 28.010 132,9 3.496 0.0931 0.295 0.049 81.7 349.7 153.823 556.4 4.560 0.276 0.272 0.194 n-Decane n-Dodecane Ethane C10H22 C12H26 C,H6 C4H100 C,H4 Не 142.286 617.6 2.108 0.603 0.247 0.490 447.3 170.340 658.3 1.824 0.713 0.24 0.562 489.5 30.070 305.4 4.884 0.148 0.285 0.098 184.5 307.7 Ethyl ether Ethylene Helium 74.123 466.7 3.638 5.036 0.280 0.262 0.281 28.054 282.4 0.129 0.276 0.085 169.4 4.003 5.19 0.227 0.0573 0.301 -0.387 4.21 540.2 n-Heptane n-Hexane C;H16 C,H14 H2 100.205 2.736 0.304 0.263 0.351 371.6 86.178 507.4 2.969 0.370 0.260 0.296 341.9 2.016 33.2 1.297 Hydrogen Hydrogen fluoride Hydrogen sulfide Methane Naphthalene Neon Nitric oxide Nitrogen n-Octane Oxygen n-Pentane Isopentane Propane Propylene Refrigerant R12 Refrigerant HFC-134a Sulfur dioxide Toluene Water Xenon 0.065 0.305 -0.22 20.4 | HF 20.006 461.0 6.488 (0.069 0.12 0.372 292.7 373.2 H2S CH4 CioHg Ne 34.080 8.942 0.0985 0.284 0.100 212.8 16.043 190.6 4.600 0.099 0.288 0.008 111.7 128,174 748.4 4.05 0.410 0.267 0.302 491.1 20.183 44.4 2.756 0.0417 0.058 0.0895 0.311 27.0 NO 30.006 180.0 6.485 0.250 0.607 121.4 N2 C3H18 O2 CSH12 CSH12 C,Hg C3H6 CC,F2 CH2FCF, SO2 C,Hs H2O Xe 28.013 126.2 3.394 0.290 0.040 77.4 114.232 568.8 2.482 0.492 0.259 0.288 0.394 398.8 31.999 154.6 5.046 0.0732 0.021 90.2 309.2 72.151 469.6 3.374 0.304 0.262 0.271 0.251 72.151 3.384 4,246 301.0 231.1 460.4 0.306 0.227 44.097 42.081 369.8 365.0 0.203 0.281 0,152 4.620 0.181 0.275 0.148 225.4 4.124 4.060 120.914 385.0 0.217 243.4 0.280 0.258 0.268 0.264 0.176 102.03 64.063 0.332 0.251 374,23 0.198 247.1 0.122 0.316 263 383.8 373.2 430.8 7.883 92.141 591.7 4.113 0.257 647.3 18.015 131.300 22.048 5.836 0.056 0.229 0.344 289.7 0.118 0.286 0.002 165.0 Source: Adapted from R. C. Reid, J. M. Prausnitz, and B. E. Poling, The Properties of Gases and Liquids, 4th ed., McGraw-Hill, New York, 1986, Appendix A and other sources.
Expert Answer:

Related Book For 

Introduction to Statistical Quality Control
ISBN: 978-1118146811
7th edition
Authors: Douglas C Montgomery
Posted Date:
Students also viewed these chemical engineering questions
-
Table 6.6-1 The Critical and Other Constants for Selected Fluids Molecular Weight (g mol-) V. (m/kmol) Substance Symbol T.(K) P (MPa) Z. Toil (K) Acetylene Ammonia C,H2 NH3 26.038 308.3 405.6 6.140...
-
Carbon dioxide gas enters a pipe at 3 MPa and 500 K at a rate of 2 kg/s. CO2 is cooled at constant pressure as it flows in the pipe and the temperature CO2 drops to 450 K at the exit. Determine the...
-
Carbon dioxide enters an adiabatic nozzle at 8 MPa and 450 K with a low velocity and leaves at 2 MPa and 350 K. Using the generalized enthalpy departure chart, determine the exit velocity of the...
-
3) Sauseda Corporation has two operating divisions-an Inland Division and a Coast Division. The company's Customer Service Department provides services to both divisions. The variable costs of the...
-
Bouderi's portfolio was hedged with a 50 percent hedge ratio. Examining just his U.S. equity exposure of A$285,000, suppose he had been fully hedged at the A$:US$ = 0.56 forward rate. Over his...
-
A set of four integers has a mode of 7 and a median of 4. What is the greatest possible average (arithmetic mean) of this set? A) 3.50 B) 3.75 C) 4.00 D) 4.25
-
The following data available for four stroke petrol engine : bore \(=20 \mathrm{~cm}\) stroke to bore ratio \(=\) 1.5 , speed \(=350 \mathrm{rpm}, \mathrm{p}_{\mathrm{m}}=2.75 \mathrm{bar}\), net...
-
The accounts in the ledger of Tempus Fugit Delivery Service contain the following balances on July 31, 2014. Instructions Prepare a trial balance with the accounts arranged as illustrated in the...
-
The total sales of a company is $ 1 , 0 0 0 K . The company aims to increase its profit by 2 5 % . There are two ways to increase profit by 2 5 % , either by purchasing the raw materials at a lower...
-
A manufacturer of a new Smart Watch claims that the probability of its watch running more than 1 minute slow or 1 minute fast after 1 year of use is .05. A consumer protection agencyhas purchased...
-
Assume today is December 15th, 2022. Kenny and Melissa Background Kenny, age 62, and Melissa, age 23, have been dating for about a year and a half. Kenny and Melissa met when Kenny was on a vacation...
-
A steel block is placed on a frictionless table and attached to a spring which is fixed to the table. The mass is then stretched and released so that it moves back and forth horizontally. If the mass...
-
ABC Corporation, a global leader in the technology sector, has a complex financial structure with subsidiaries operating in various countries. The company decides to conduct a comprehensive audit to...
-
Fifteen identical particles have various speeds: one has a speed of 2.00 m/s, two have speeds of 3.00 m/s, three have speeds of 5.00 m/s, four have speeds of 8.00 m/s, three have speeds of 9.00 m/s,...
-
Ethical considerations play a paramount role in the practice of auditing, a profession entrusted with ensuring the accuracy and reliability of financial information. Auditors face a myriad of ethical...
-
An object moves in 2 dimensions with a constant acceleration of (2 m/s)i + (4 m/s). At t=0 s, they component of the velocity is 5 m/s. Knowing that the x component of the velocity is 30 m/s after 5...
-
Barga Company's net sales for Year 1 and Year 2 are $665,000 and $742,000, respectively. Its year-end balances of accounts receivable follow: Year 1, $58,000; and Year 2, $95,000. a. Complete the...
-
We all experience emotions, but some people disguise their true feelings better than others. Do you think this is a helpful or harmful thing to do? Under what conditions do you think it would be most...
-
The data in Table 9E.1 represent individual observations on molecular weight taken hourly from a chemical process. The target value of molecular weight is 1,050 and the process standard deviation is...
-
Consider the control chart shown in Exercise 5.17. Would the use of warning limits reveal any potential out-of-control conditions?
-
A supplier ships a product in lots of size N = 8,000. We wish to have an AOQL of 3%, and we are going to use single sampling. We do not know the suppliers process fallout but suspect that it is at...
-
Draw a class diagram for resource rental pattern and its installation for library service by using analogy approach.
-
What is Fowlers account pattern? Explain with the help of a diagram.
-
Draw a class diagram for account with entry pattern.

Study smarter with the SolutionInn App


