1. In the potentiometric titration of an aqueous potassium chloride solution with 0.09603 M silver nitrate,...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
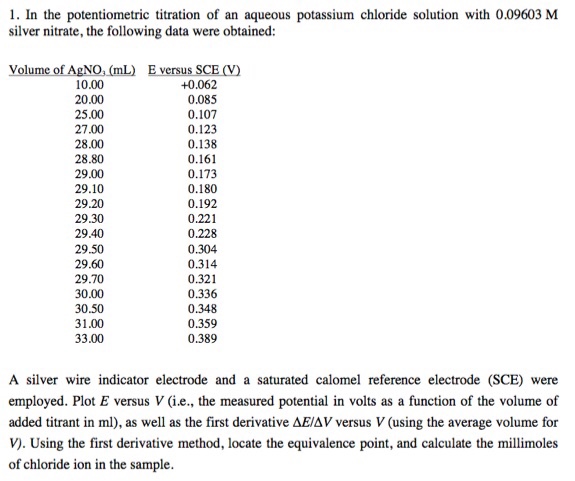
1. In the potentiometric titration of an aqueous potassium chloride solution with 0.09603 M silver nitrate, the following data were obtained: Volume of AgNO, (mL) E versus SCE (V) 10.00 20.00 25.00 27.00 28.00 28.80 29.00 29.10 29.20 29.30 29.40 29.50 29.60 29.70 30.00 30.50 31.00 33.00 +0.062 0.085 0.107 0.123 0.138 0.161 0.173 0.180 0.192 0.221 0.228 0.304 0.314 0.321 0.336 0.348 0.359 0.389 A silver wire indicator electrode and a saturated calomel reference electrode (SCE) were employed. Plot E versus V (i.e., the measured potential in volts as a function of the volume of added titrant in ml), as well as the first derivative AE/AV versus V (using the average volume for V). Using the first derivative method, locate the equivalence point, and calculate the millimoles of chloride ion in the sample. 1. In the potentiometric titration of an aqueous potassium chloride solution with 0.09603 M silver nitrate, the following data were obtained: Volume of AgNO, (mL) E versus SCE (V) 10.00 20.00 25.00 27.00 28.00 28.80 29.00 29.10 29.20 29.30 29.40 29.50 29.60 29.70 30.00 30.50 31.00 33.00 +0.062 0.085 0.107 0.123 0.138 0.161 0.173 0.180 0.192 0.221 0.228 0.304 0.314 0.321 0.336 0.348 0.359 0.389 A silver wire indicator electrode and a saturated calomel reference electrode (SCE) were employed. Plot E versus V (i.e., the measured potential in volts as a function of the volume of added titrant in ml), as well as the first derivative AE/AV versus V (using the average volume for V). Using the first derivative method, locate the equivalence point, and calculate the millimoles of chloride ion in the sample.
Expert Answer:
Answer rating: 100% (QA)
Prepare the following plot as below Volume AV mL of E vs AgNO3 V Vdown SCE V mL Vup 1000 2000 2500 2... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Silver nitrate reacts with strontium chloride in an aqueous precipitation reaction. What are the formulas of silver nitrate and strontium chloride? Write the molecular equation and net ionic equation...
-
Using standard electrode potentials, decide whether aqueous sodium hypochlorite solution will oxidize Br to Br2 in basic solution under standard conditions. See Appendix I for data.
-
An aqueous solution of ammonium chloride and barium hydroxide is heated, and the compounds react to give off ammonia gas. Barium chloride solution and water are also products. Write a balanced...
-
Wansley Portal Inc., a large Internet service provider, is evaluating the possible acquisition of Alabama Connections Company (ACC), a regional Internet service provider. Wansley's analysts project...
-
List any errors you can find in the following partial balancesheet: Tulips Company Balance Sheet December 31, 2012 Assets Current assets: Cash $138,000 Notes receivable $400,000 Less interest...
-
Identify the key primary research methods and the appropriate use of each one.
-
Sunmask is a cosmetics firm that has seen its stock price fall and its earnings decline in the past year. You have been hired as the new CEO of the company, and a careful analysis of Sunmasks current...
-
Beachfront property owners of the Village of Eden requested a seawall be constructed to protect their beach. The seawall was financed through a note payable, which was to be repaid from taxes raised...
-
NO PAPER WORK ONLY TYPED ANSWERS ASAP NO PLAGARISM DONT USE CHAT GPT I WILL REPORT YOU. A 12-year bond pays semi-annual coupons at j2 # 10.3% and has a yield rate of j2 = 7.1%. If the book value...
-
please help.. A recent study of the laundry habits of Americans included the time in minutes of the wash cycle. A sample of 40 observations follows. Click here for the Excel Data File Determine the...
-
Suppose that a price - taker firm has a marginal cost function given by: MC = 20 +0.5q. The firm could join cartel in its industry and agree to a quota of 5 units. The collusion drives the price of...
-
Consider the impact of digital and interactive storytelling on the evolution of literary forms. How are new technologies redefining the boundaries between author, text, and audience ?
-
PART 3 Paul Sabin organized Sabin Electronics 10 years ago to produce and sell several electronic devices on which he had secured patents. Although the company has been fairly profitable, it is now...
-
Champion Real Property has a decision to make between two property investment alternatives. The company requires a 1 0 % return on investment. Predicted data are provided below: Property Y Property Z...
-
Write_research method and description of respondents which may be used based on the study below: TITLE: Prevention of Drug Abuse Among Youth in Barangay D.I. Calihan, San Pablo City, Laguna....
-
Experimental System Parameter Identification. The following set of data was collected from a system. Plot the response. What is the steady state asymptote? Use the natural log analysis technique to...
-
1. Firms may hold financial assets to earn returns. How the firm would classify financial assets? What treatment will such financial assets get in the financial statements in accordance with US GAAP...
-
The equilibrium constant Kc for the reaction at 450oC is 0.159. Calculate the equilibrium composition when 1.00 mol N2 is mixed with 3.00 mol H2 in a 5.00-L vessel. N2(g) 3H2(g) 2NH3(g)
-
Part 1
-
When aluminum sulfate is dissolved in water, it produces an acidic solution. Suppose the pH of this solution is raised by the dropwise addition of aqueous sodium hydroxide. (a) Describe what you...
-
1. ETHICS Lee McNeely told Hardee's officials that he was interested in purchasing multiple restaurants in Arkansas. A Hardee's officer assured him that any of the company-owned stores in Arkansas...
-
Question: In which one or more of the following forms of organization is it true that none of the partners are liable for the debts of the partnership? A. General partnership B. Limited liability...
-
2. CPA QUESTION: Assuming all other requirements are met, a corporation may elect to be treated as an S corporation under the Internal Revenue Code if it has: (a) Both common and preferred...

Study smarter with the SolutionInn App


