1. Indicate if the reaction is Felkin-Ahn or Cram and Chelation model. 2. Show Newman Projection...
Fantastic news! We've Found the answer you've been seeking!
Question:
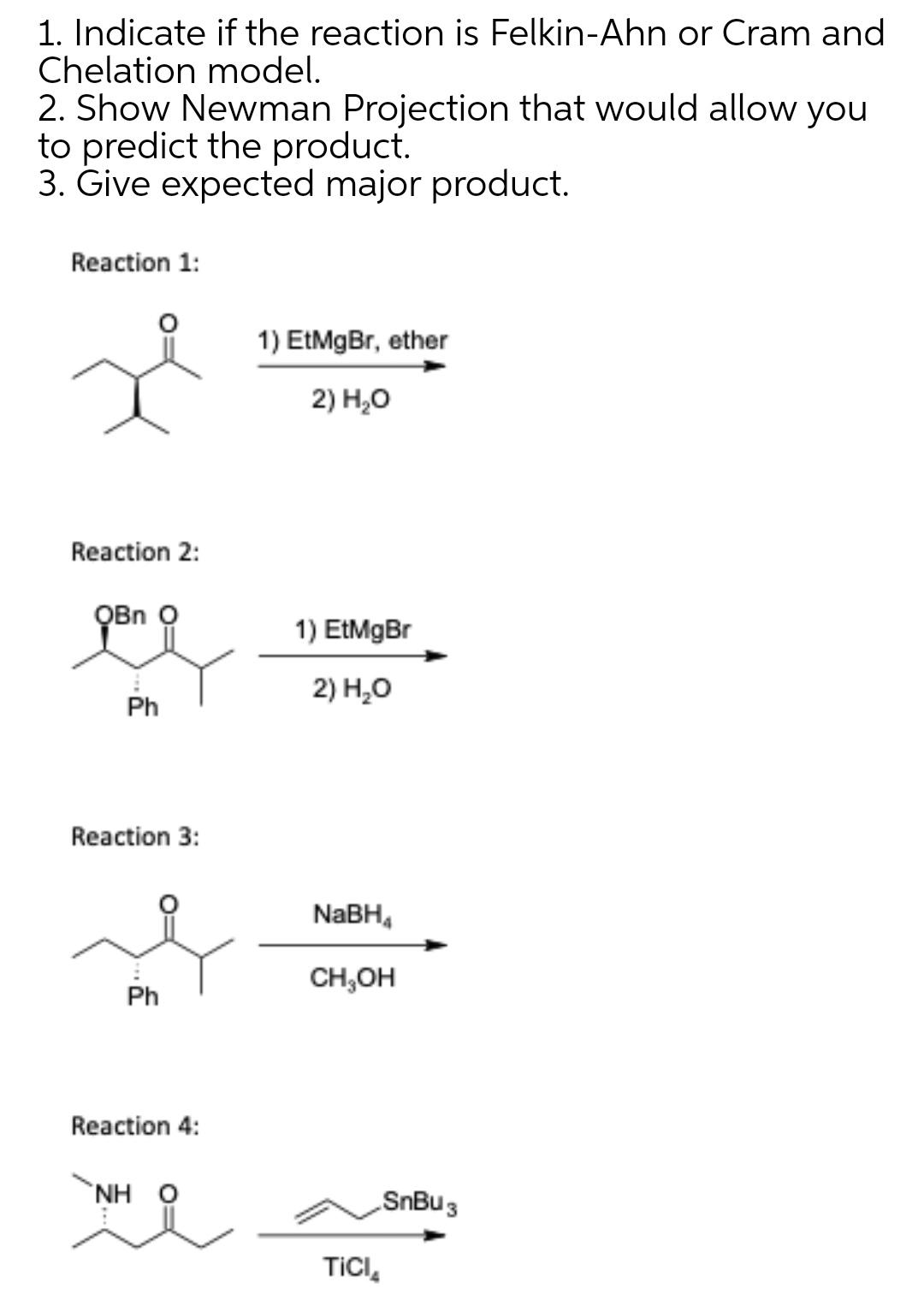
Transcribed Image Text:
1. Indicate if the reaction is Felkin-Ahn or Cram and Chelation model. 2. Show Newman Projection that would allow you to predict the product. 3. Give expected major product. Reaction 1: 1) EtMgBr, ether 2) H,о Reaction 2: QBn O 1) EtMgBr 2) Н,О Ph Reaction 3: NABH, CH,OH Ph Reaction 4: NH O SnBu 3 TICI, 1. Indicate if the reaction is Felkin-Ahn or Cram and Chelation model. 2. Show Newman Projection that would allow you to predict the product. 3. Give expected major product. Reaction 1: 1) EtMgBr, ether 2) H,о Reaction 2: QBn O 1) EtMgBr 2) Н,О Ph Reaction 3: NABH, CH,OH Ph Reaction 4: NH O SnBu 3 TICI,
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
If the reaction is carried out at constant V, how does the total pressure change if T increases? H 2 (g) + Cl 2 (g) 2HCl(g) at equilibrium. Assume ideal gas behavior.
-
Select reaction conditions that would allow you to carry out each of the following stereospecific transformations: a. b. CH3 (R)-1,2-propanediol CH3 (S)-1,2-propanediol
-
Design a SIP experiment that would allow you to determine which organisms in a lake water sample were capable of oxidizing the hydrocarbon hexane (C6H14). Assume that four different species could do...
-
The following is the documentation of the payroll cycle at McQuarrie Enterprises, a ladies clothing wholesaler. There are approximately 50 staff members at McQuarrie Enterprises. Every employee has a...
-
The president and vice president of USA Corporation receive benefits that are unavailable to other employees. These benefits include free parking, payment of monthly expenses in a local club,...
-
Though income is usually accounted for on an accruals concept, dividends proposed by an entity in which shares are held are not accounted for until received. The accounting convention used is: (a)...
-
A sample of 32 students took a class designed to improve their SAT math scores. Following are their scores before and after the class. Can you conclude that the mean increase in score is less than 15...
-
Steelcase Inc. is one of the largest manufactures of office furniture in the United States. In Grand Rapids, Michigan, it produces filing cabinets in two departments: Fabrication and Trim Assembly....
-
It is recommended to start by reviewing the 2019 file as an example to see what was done in the previous year. Similar work should be completed for 2020. 2. The client was unable to prepare bank...
-
Effective financial statement analysis requires an understanding of a firms economic characteristics. The relations between various financial statement items provide evidence of many of these...
-
Suppose we have obtained the tableau in Table 75 for a maximization problem. State conditions on au 02, 83, b, 01, and 02 that are required to make the following statements true: a. The current...
-
Find all possible I/O equations. \(\left\{\begin{array}{l}\ddot{x}_{1}+x_{1}-x_{3}-\left(\dot{x}_{2}-\dot{x}_{1} ight)-\frac{1}{2}\left(x_{2}-x_{1} ight)=f(t) \\...
-
Find all possible I/O equations. \(\left\{\begin{array}{l}\frac{1}{2} \ddot{q}_{1}+\dot{q}_{1}+2\left(q_{1}-q_{2} ight)+\frac{1}{3} q_{1}=v(t) \\ \dot{q}_{2}-2\left(q_{1}-q_{2} ight)=0\end{array},...
-
Find all possible I/O equations. \(\left\{\begin{array}{l}\ddot{x}_{1}+\dot{x}_{1}+3\left(x_{1}-x_{2} ight)=f_{1}(t) \\ \ddot{x}_{2}-3\left(x_{1}-x_{2} ight)=f_{2}(t)\end{array}, f_{1}(t), f_{2}(t)=...
-
Find all possible I/O equations. \(\left\{\begin{array}{l}\ddot{\theta}_{1}+\frac{2}{3} \dot{\theta}_{1}+\frac{1}{3}\left(\theta_{1}-\theta_{2} ight)=u(t) \\...
-
Find all possible I/O equations. \(\left\{\begin{array}{l}\ddot{x}_{1}+\dot{x}_{1}+2\left(x_{1}-x_{2} ight)=0 \\ \ddot{x}_{2}+\dot{x}_{2}-2\left(x_{1}-x_{2} ight)=f(t)^{\prime}\end{array}, f(t)=...
-
Papst Company is preparing its cash budget for the month of May. The following information is available concerning its accounts receivable (based on sales made to customers on open account): Actual...
-
A bubble-point liquid feed is to be distilled as shown in Figure. Use the Edmister group method to estimate the mole-fraction compositions of the distillate and bottoms. Assume initial overhead and...
-
Chemical degradation of chlorophyll gives a number of substances including phytol. The constitution of phytol is given by the name 3,7,11,15-tetramethyl-2-hexadecen-1-ol. How many stereoisomers have...
-
Suggest reasonable explanations for each of the following observations: (a) The C==O stretching frequency of ,-unsaturated ketones (about 1675 cm-1) is less than that of typical dialkyl ketones (1710...
-
Give the mechanistic symbols (SN1, SN2, E1, E2) that are most consistent with each of the following statements: (a) Methyl halides react with sodium ethoxide in ethanol only by this mechanism. (b)...
-
What are the responsibilities of the governments toward the stateless? Do the United States and other powerful nations have responsibilities toward groups such as the Rohingya? Following the...
-
The United States and Canada are two of only 33 countries that have jus soli, the right to citizenship of any individual born on their soil. Why do you think this right is so limited, and do you...
-
What are stratified societies and what kinds of social systems are they identified with?

Study smarter with the SolutionInn App


