(1) Let h(x)=1+x+x+x* be the parity check polynomial of a C(7, 4) cyclic code. ,3 .4...
Fantastic news! We've Found the answer you've been seeking!
Question:
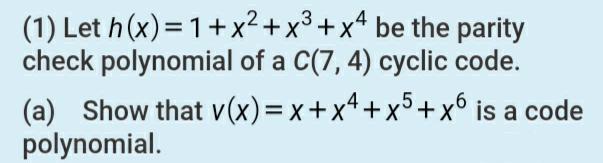
Transcribed Image Text:
(1) Let h(x)=1+x²+x³+x* be the parity check polynomial of a C(7, 4) cyclic code. ,3 .4 .5 (a) Show that v(x)=x+x+x°+x° is a code polynomial. (1) Let h(x)=1+x²+x³+x* be the parity check polynomial of a C(7, 4) cyclic code. ,3 .4 .5 (a) Show that v(x)=x+x+x°+x° is a code polynomial.
Expert Answer:
Answer rating: 100% (QA)
Consider the given equation hx 1 x 2 x 3 x 4 and C 74 vx x x ... View the full answer

Related Book For 

Organic Chemistry
ISBN: 978-1118133576
11th edition
Authors: Graham Solomons, Craig Fryhle, Scott Snyder
Posted Date:
Students also viewed these mathematics questions
-
Let X and Y be nonempty sets and let h : X Y R have bounded range in R. Let f : X R and g : Y R be defined by F(x) := sup{h(x; y) : y Y}; g(y) := inf{h(x; y) : x X}: Prove that Sup{g(y) : y Y}...
-
Let X and Y be nonempty sets and let h : X Ã Y R have bounded range in R. Let F : X R and G : Y R be defined by F(x) := sup{h(x; y) : y Y}; G(y) := sup{h(x; y) : x X}: Establish the Principle...
-
Let A be a 7 x 5 matrix with rank equal to 4 and let b be a vector in R8. The four fundamental subspaces associated with A are R(A), N(AT), R(AT), and N(A). (a) What is the dimension of N(AT) and...
-
Compute the least-squares regression line for predicting power (y) from wind speed (x).
-
Right-tailed test z = 2.46 = 0.01 Find the P-value for the hypothesis test with the standardized test statistic z. Decide whether to reject H0 for the level of significance .
-
Maggies Muffins Bakery generated $5 million in sales during 2018, and its year-end total assets were $2.5 million. Also, at year-end 2018, current liabilities were $1 million, consisting of $300,000...
-
How you may use computers for litigation support?
-
Consider the following project activities: a. Draw the project network diagram. b. Identify all the paths through the network and their lengths. c. Identify all the critical activities and path(s)....
-
Produces table lamps. The company uses a work order system. On July 28, he received two requests: ORDER 181: Comercial Gutirrez, 500 units of table lamps, Standard size, various styles and colors, at...
-
An investor, wants to know how two portfolios are performing in the market: 7% Y (3%) 12% Probability Market portfolio 0.40 0.20 15% 0.40 Beta factor 10% 18% Portfolio X 1.30 10% 8% 4% 0.75 1 The...
-
III. Full State Estimator: Let A = 10 a) Show (A, C) observable 0 1 b) Design a Full State Estimator: =[001],x(0)=1 [= A + Bu+K(y-) |=Cx; (0) = 0 with K so that the estimator error e=x-x eigenvalues...
-
What is a system call interface, and how does it maintain isolation between user space and kernel space? Give examples of how improper handling of system calls could lead to security vulnerabilities.
-
Pronghorn Corp reported net sales $675,000, cost of goods sold $438,750, operating expenses $154,000, and net income $69,525. Calculate the profit margin and gross profit rate. (Round Profit margin...
-
Year A B 0 ($32,000) ($27,500) 1 $15,000 $17,000 2 $22,000 $16,500 3 $19,000 $15,000 The process equipment will run for 3 years. With a MARR of 8%, calculate the following: a) Calculate the Future...
-
Wal-Mart has 138 million weekly customers. In 2006 Wal-Mart had annual sales of $312 billion and net income of more than $11.2 billion. There are about 5,300 Wal-Mart stores worldwide. During a...
-
Based on the following information, what is C/G/S for Moroni Industries. Inc. using FIFO under the periodic inventory method? Beginning Inventory 10 @ $120; Purchase 60 @ $112, Sale 40; Purchase 30 @...
-
The various elements of the Lucky Me Animal Rescue adoption event project budget and how the scope and schedule are used to create the budget. Note: You may assume any reasonable total budget. The...
-
Refrigerant-134a enters an adiabatic compressor as saturated vapor at 120 kPa at a rate of 0.3 m3/min and exits at 1-MPa pressure. If the isentropic efficiency of the compressor is 80 percent,...
-
A commercial synthesis of folic acid consists of heating the following three compounds with aqueous sodium bicarbonate. Propose reasonable mechanisms for the reactions that lead to folic acid. Hint:...
-
Specify the appropriate alkene and reagents for synthesis of each of the following alcohols by hydroboration-oxidation. (a) (b) (c) (d) (e) (f) CH 2 OH
-
(a) The most stable tautomeric form of guanine is the lactam form (or cyclic amide, see Section 17.8I). This is the form normally present in DNA, and, as we have seen, it pairs specifically with...
-
During 2020, Many Miles Trucking paid salary expense of \(\$ 40\) million. At December 31, Many Miles accrued salary expense of \(\$ 2\) million. Many Miles paid \(\$ 1.9\) million to its employees...
-
Ford Canada sells large fleets of vehicles to auto rental companies, such as Budget and Avis. Suppose Budget is negotiating a contract with Ford to purchase 1,000 Explorers by the end of next year....
-
Answer the following questions about prepaid expenses: a. On November 1, World Travel Ltd. prepaid \(\$ 6,000\) for three months' rent. Give the adjusting entry to record rent expense at December 31....

Study smarter with the SolutionInn App


