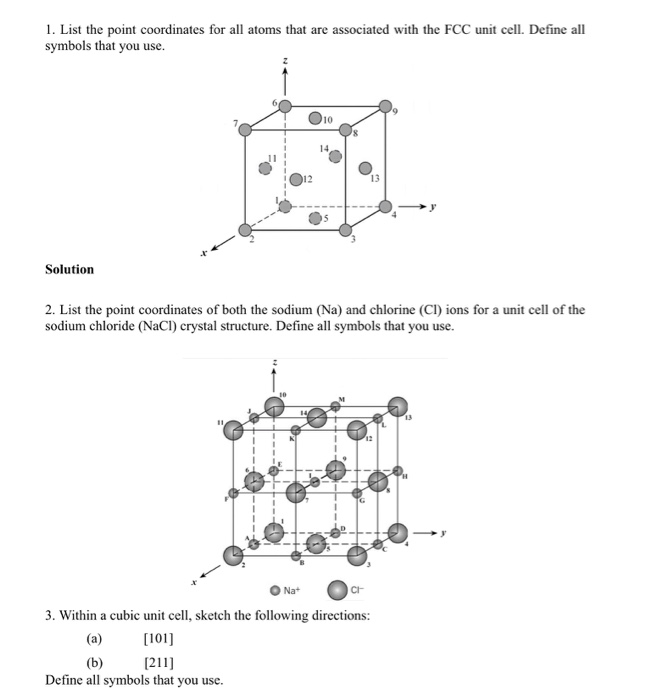
1. List the point coordinates for all atoms that are associated with the FCC unit cell....
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
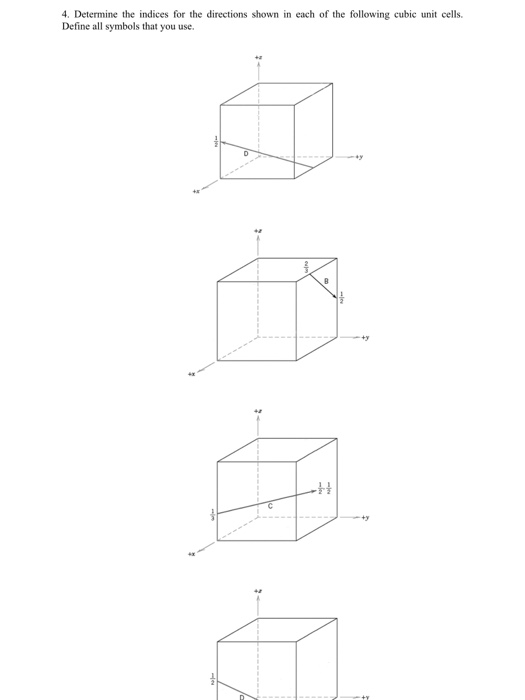
1. List the point coordinates for all atoms that are associated with the FCC unit cell. Define all symbols that you use. Solution 12 Na+ 10 2. List the point coordinates of both the sodium (Na) and chlorine (CI) ions for a unit cell of the sodium chloride (NaCl) crystal structure. Define all symbols that you use. 13 CI 3. Within a cubic unit cell, sketch the following directions: [101] (b) [211] Define all symbols that you use. 4. Determine the indices for the directions shown in each of the following cubic unit cells. Define all symbols that you use. 2 H a Solution 5. Sketch within a cubic unit cell the following planes: (a) (107) (b) (271) Solution 6. Derive linear density expressions for FCC [100] and [111] directions in terms of the atomic radius R. Solution D 7. Derive planar density expressions for FCC (100) and (111) planes in terms of the atomic radius R. Solution 8. Determine the expected diffraction angle for the first-order diffraction from the (111) set of planes for FCC nickel (Ni) when monochromatic radiation of wavelength 0.1937 nm is used. Solution 9. The metal niobium (Nb) has a BCC crystal structure. If the angle of diffraction for the (211) set of planes occurs at 75.99° (first-order diffraction) when monochromatic x-radiation having a wavelength of 0.1659 nm is used, compute the following: (a) the interplanar spacing for this set of planes and (b) the atomic radius for the Nb atom. Solution 1. List the point coordinates for all atoms that are associated with the FCC unit cell. Define all symbols that you use. Solution 12 Na+ 10 2. List the point coordinates of both the sodium (Na) and chlorine (CI) ions for a unit cell of the sodium chloride (NaCl) crystal structure. Define all symbols that you use. 13 CI 3. Within a cubic unit cell, sketch the following directions: [101] (b) [211] Define all symbols that you use. 4. Determine the indices for the directions shown in each of the following cubic unit cells. Define all symbols that you use. 2 H a Solution 5. Sketch within a cubic unit cell the following planes: (a) (107) (b) (271) Solution 6. Derive linear density expressions for FCC [100] and [111] directions in terms of the atomic radius R. Solution D 7. Derive planar density expressions for FCC (100) and (111) planes in terms of the atomic radius R. Solution 8. Determine the expected diffraction angle for the first-order diffraction from the (111) set of planes for FCC nickel (Ni) when monochromatic radiation of wavelength 0.1937 nm is used. Solution 9. The metal niobium (Nb) has a BCC crystal structure. If the angle of diffraction for the (211) set of planes occurs at 75.99° (first-order diffraction) when monochromatic x-radiation having a wavelength of 0.1659 nm is used, compute the following: (a) the interplanar spacing for this set of planes and (b) the atomic radius for the Nb atom. Solution
Expert Answer:

Related Book For 

Materials Science and Engineering An Introduction
ISBN: 978-0470419977
8th edition
Authors: William D. Callister Jr., David G. Rethwisch
Posted Date:
Students also viewed these accounting questions
-
List the point coordinates for all atoms that are associated with the FCC unit cell.
-
List the point coordinates for all atoms that are associated with the FCC unit cell(figure). (b) (a) (c)
-
List the point coordinates of all atoms that are associated with the diamond cubic unit cell.
-
Let S represent the amount of steel produced (in tons). Steel production is related to the amount of labor used (L) and the amount of capital used (C) by the following function: S = 20 L0.30 C 0.70...
-
On July 21, Boehmer, a customer of Birmingham Trust, secured a loan from that bank for the principal sum of $5,500 to purchase a boat allegedly being built for him by A.C. Manufacturing Company, Inc....
-
An executive at a large package delivery company has complained that by shifting some of its business to independent channel members, the company has lost control of the customer. What do you think...
-
Martin is sitting in his bedroom surrounded by dozens of papersgas receipts for his car, a checkbook, a couple of unopened and unpaid bills, paycheck stubs from his work at a day-care center, and...
-
Prepare journal entries to record the transactions in E12- 3. In E12- 3. For each of the following transactions, identify the net asset classification ( unrestricted, temporarily restricted,...
-
Please help me. Company XYZ produces and sells wireless earphones. The selling price per unit is $5 and the total fixed costs are $20,000. Assuming a variable cost ratio of 40%, what is the breakeven...
-
You are the manager of the Mighty Fine mutual fund. The following table reflects the activity of the fund during the last quarter. The fund started the quarter on January 1 with a balance of $40...
-
The following is a partial balance sheet for Andy and Kim Garza. Complete the balance sheet by determining their total assets, total current liabilities, long-term liabilities, and net worth. Balance...
-
Fixed costs remain constant at$450,000 per month. Duringhigh-output months variable costs are$300,000, and duringlow-output months variable costs are$125,000. What are the respective high and...
-
A corporate bond portfolio has a modified duration of 5.35. Current interest rates are flat at 4.0%. 1. Estimate the approximate percentage change in the value of the portfolio if interest rates rise...
-
Both competition and individualism are deeply held values in American society. Have I stressed collaboration in a way that omits these values from the students lives or in a way that weaves them...
-
The company has an operating profit of 159290 euros and a net profit of 45000 euros. It is subject to a corporate income tax rate of 30%. Its net financial debts amount to 750,000 euros and its...
-
KK Ltd. runs a departmental store which grants 10 focuses for each acquisition of ' 500 which can be limited by the clients for additional shopping with a similar dealer. Unutilised focuses will pass...
-
Using single-subject evaluation with an ABA design for an adolescent victim of sexual exploitation exhibiting high-risk sexualized behaviors (ex., prostitution), what is a specific problem that can...
-
Find an equation of the given line. Slope is -2; x-intercept is -2
-
For each of the following crystal structures, represent the indicated plane in the manner of Figures 3.11 and 3.12, showing both anions and cations: (a) (100) plane for the rock salt crystal...
-
(a) From Figure 7.25, compute the length of time required for the average grain diameter to increase from 0.01 to 0.1 mm at 500C for this brass material. (b) Repeat the calculation at 600C.
-
The number-average molecular weight of a polypropylene is 1,000,000 g/mol. Compute the degree of polymerization.
-
2. YOU BE THE JUDGE WRITING PROBLEM Jerome Schneider wrote several books on how to avoid taxes. These books were sold on Amazon.com. Amazon permits visitors to post comments about items for sale....
-
1. ETHICS Matt Drudge published a report on his Web site (http://www.drudgereport .com) that White House aide Sidney Blumenthal "has a spousal abuse past that has been effectively covered up.......
-
3. During the course of 10 months, Joseph Melle sent more than 60 million unsolicited e-mail advertisements to AOL members. What charges could be brought against him?

Study smarter with the SolutionInn App


