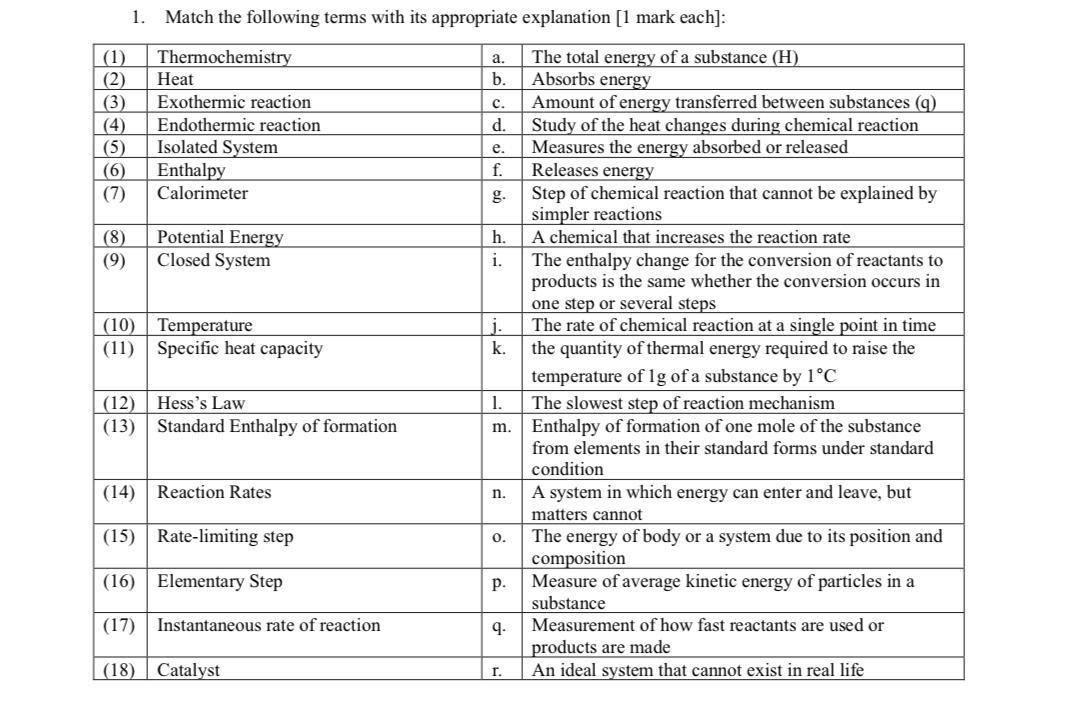
1. Match the following terms with its appropriate explanation [1 mark each]: (1) Thermochemistry (2) Heat...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
1. Match the following terms with its appropriate explanation [1 mark each]: (1) Thermochemistry (2) Heat (3) Exothermic reaction Endothermic reaction (4) (6) (5) Isolated System Enthalpy Calorimeter (7) (8) (9) Potential Energy Closed System (10) Temperature (11) Specific heat capacity (12) Hess's Law (13) Standard Enthalpy of formation (14) Reaction Rates (15) Rate-limiting step (16) Elementary Step (17) Instantaneous rate of reaction (18) Catalyst a. b. C. Amount of energy transferred between substances (q) d. Study of the heat changes during chemical reaction Measures the energy absorbed or released Releases energy Step of chemical reaction that cannot be explained by simpler reactions e. f. g. h. i. j. k. 1. m. n. 0. P. 9. The total energy of a substance (H) Absorbs energy r. A chemical that increases the reaction rate The enthalpy change for the conversion of reactants to products is the same whether the conversion occurs in one step or several steps The rate of chemical reaction at a single point in time the quantity of thermal energy required to raise the temperature of 1g of a substance by 1C The slowest step of reaction mechanism Enthalpy of formation of one mole of the substance from elements in their standard forms under standard condition A system in which energy can enter and leave, but matters cannot The energy of body or a system due to its position and composition Measure of average kinetic energy of particles in a substance Measurement of how fast reactants are used or products are made An ideal system that cannot exist in real life 1. Match the following terms with its appropriate explanation [1 mark each]: (1) Thermochemistry (2) Heat (3) Exothermic reaction Endothermic reaction (4) (6) (5) Isolated System Enthalpy Calorimeter (7) (8) (9) Potential Energy Closed System (10) Temperature (11) Specific heat capacity (12) Hess's Law (13) Standard Enthalpy of formation (14) Reaction Rates (15) Rate-limiting step (16) Elementary Step (17) Instantaneous rate of reaction (18) Catalyst a. b. C. Amount of energy transferred between substances (q) d. Study of the heat changes during chemical reaction Measures the energy absorbed or released Releases energy Step of chemical reaction that cannot be explained by simpler reactions e. f. g. h. i. j. k. 1. m. n. 0. P. 9. The total energy of a substance (H) Absorbs energy r. A chemical that increases the reaction rate The enthalpy change for the conversion of reactants to products is the same whether the conversion occurs in one step or several steps The rate of chemical reaction at a single point in time the quantity of thermal energy required to raise the temperature of 1g of a substance by 1C The slowest step of reaction mechanism Enthalpy of formation of one mole of the substance from elements in their standard forms under standard condition A system in which energy can enter and leave, but matters cannot The energy of body or a system due to its position and composition Measure of average kinetic energy of particles in a substance Measurement of how fast reactants are used or products are made An ideal system that cannot exist in real life
Expert Answer:
Answer rating: 100% (QA)
here is the correct matching of the terms with their appropriate explanations 1 d Thermochemistry St... View the full answer

Related Book For 

Accounting Information Systems
ISBN: 978-0133428537
13th edition
Authors: Marshall B. Romney, Paul J. Steinbart
Posted Date:
Students also viewed these chemical engineering questions
-
Multiple Choice Question 1 Raw materials and merchandise inventories are included in (a) Expenses (b) Fixed assets (c) Cash (d) Current assets (e) None of these 2 According to IAS 2, inventories are...
-
Use the following table to compute the operating income if 130,000 units are sold. Less: Requirement 3. What would the company's monthly operating income be if the company had sales of $4,500,000?...
-
Presented below is selected information related to the financial instruments of Dawson Company at December 31, 2020. This is Dawson Company?s first year of operations. Instructions a. Dawson elects...
-
Destin Products makes digital watches. Destin is preparing a product-life cycle budget for a new watch, MX3. Development on the new watch is to start shortly. Estimates for MX3 are as follows: 1....
-
The State of Washington established a program to provide legal services for the needy. The program, called Interest on Lawyers' Trust Accounts (IOLTA), required that clients deposit their legal...
-
A stock price is governed by geometric Brownian motion with \(\mu=.20\) and \(\sigma=.40\). The initial price is \(S(0)=1\). Evaluate the four quantities E[In S(1)], E[S(1)], stdev[In S(1)]...
-
1. Is this company running afoul of the Fair Labor Standards Act (FLSA)? Refer back to the discussion earlier in this chapter. Would this company be able to document that the store managers are...
-
A monopolist has the long run cost function C(Q) = 502. Inverse market demand is PD (Q) = 360-2Q. The firm practices first degree (perfect) price discrimination. a. Find the monopolist's profit...
-
Concord Corporation had 126,000 shares of stock outstanding on January 1, 2017. On May 1, 2017, Concord issued 57,600 shares. On July 1, Concord purchased 9,000 treasury shares, which were reissued...
-
With 500 kr, 400 kr, 300 kr, 200 kr and 100 kr as the sales prices the company expects to produce respectively 1000, 2000, 3000, 4000 and 500 units of a product. The company has proportional variable...
-
Can you try to define the liquidity of stocks quantitatively? (It means how to measure the liquidity.) How can we improve the liquidity of stocks?
-
Compute how much will be included and excluded in the gross income given the following scenarios. 1. A call center agent receiving a monthly salary of P21,000 with mandatory annual deductions of...
-
4. Explain how leverages intensify your profits when you are right about future movements of stock prices.
-
A computer retailer offers a payment plan at 6.5% annual interest, compounded monthly. Customers can pay for a gaming system by monthly payments of $156.20 for 1 year. What is the cost of the gaming...
-
Cassa Nova is delinquent on his alimony payments to his spouse, Hillary. Hillary hasdemanded copies of Cassas tax returns and has a court date scheduled for June 10,2018 to demand that payments are...
-
What impact has the Internet had on the globalization of small firms? How do you think small companies will use the Internet for business in the future?
-
Don Richardson, JEM Corporations vice president of marketing is part of a management team that for several months has been discussing plans to develop a new line of business. Rumors about the major...
-
What are the advantages and disadvantages of having the person responsible for information security report directly to the chief information officer (CIO), who has overall responsibility for all...
-
The basic REA template includes links between two events and links between events and resources and between events and agents. Why do you think the basic REA template does not include direct links...
-
Two loudspeakers \(42.0 \mathrm{~m}\) apart and facing each other emit identical \(115 \mathrm{~Hz}\) sinusoidal sound waves in a room where the sound speed is \(345 \mathrm{~m} / \mathrm{s}\). Susan...
-
A child's train whistle replicates a classic conductor's whistle from the early 1900 s. This whistle has two open-open tubes that produce two different frequencies. When you hear these two different...
-
A student waiting at a stoplight notices that her turn signal, which has a period of \(0.85 \mathrm{~s}\), makes one blink exactly in sync with the turn signal of the car in front of her. The blinker...

Study smarter with the SolutionInn App


