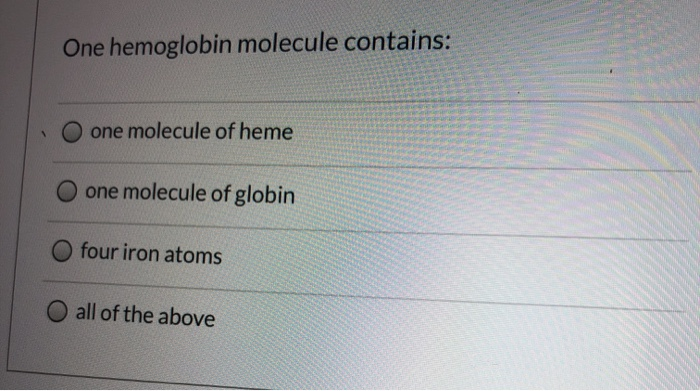
1 One hemoglobin molecule contains: one molecule of heme one molecule of globin four iron atoms...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
1 One hemoglobin molecule contains: one molecule of heme one molecule of globin four iron atoms all of the above 1 One hemoglobin molecule contains: one molecule of heme one molecule of globin four iron atoms all of the above
Expert Answer:
Answer rating: 100% (QA)
Answer D all of the above One hemoglobin molecule contains one molecule of h... View the full answer

Related Book For 

Posted Date:
Students also viewed these law questions
-
Assume that Bill's marginal tax rate is 40%. If corporate bonds pay 10% interest, what interest rate would a municipal bond have to offer for Bill to be indifferent between the two bonds? A. 10% B....
-
Consider the two languages L1=(a*+b)* and L2=(a*b*)* over the input alphabet {a, b}. Which of the following option describes the relation between the languages L1 and L2? a) LcL b) L2 cL c) L = L, d)...
-
5. For which of the following taxes is there no ceiling on the amount of employee annual earnings subject to the tax? a. only Medicare tax b. only unemployment compensation tax c. only Social...
-
make a small case or use a situation/problem from real life. You will discuss this situation together with a discussion that works through a solution of your own to the problem posed. Problems or...
-
The following information about the payroll for the week ended December 30 was obtained from the records of Greenfield Co.: Tax rates assumed: Social security, 6% on first $100,000 of employee annual...
-
Lai Hing Ltd. acquired all the assets and liabilities of Sound Ltd. on January 1, 2013. At this date, the assets and liabilities of Sound consisted of: In exchange for these net assets, Lai Hing...
-
Miguel read his credit card statement and discovered that a store charged him twice for the same item. His total bill was \($658.22\): 1. Calculate how much Miguel should subtract from his bill and...
-
Hannon Company makes swimsuits and sells these suits directly to retailers. Although Hannon has a variety of suits, it does not make the All-Body suit used by highly skilled swimmers. The market...
-
10) Your firm is planning to invest in an automated packaging plant. You find a comparable firm, Thurbinar Design, which is engaged in a similar line of business. Thurbinar has a stock price of $16...
-
Crane Medical manufactures hospital beds and other institutional furniture. The company's comparative balance sheet and income statement for 2019 and 2020 follow. Assets Current assets Crane Medical...
-
the purpose of the Income Statement and why it is important for businesses and investors. https://www.sec.gov/ixviewer/ix.html?doc=/Archives/edgar/data/320193/000032019323000106/aapl-20230930.htm 2....
-
12-32. Examine the following two-factor analysis of variance table: Source SS df Factor A 162.79 4 Factor B AB Interaction 262.31 12 Error Total 1,298.74 84 MS F-ratio 28.12 a. Complete the analysis...
-
A 20 g ball is fired from a compressed, vertical spring with a spring constant of 265 N/m. The spring is compressed 7 cm before the ball is fired. The spring shoots the ball straight up to a maximum...
-
a ) How many customers were studied: b ) About how many customers spent less than $ 5 0 weekly? c ) About 7 5 % of the customers spent less than what amount? d ) 4 0 of the customers studied spend...
-
On the first statistics exam, the coefficient of determination between the hours studied and the grade earned was 68%. The standard error of estimate was 6. There were 16 students in the class....
-
With the amount of data available to companies doubling every year, new sources of data, and innovations in data collection, possibilities for marketers to identify market niches and finely tune...
-
Income statement and balance sheet data for The Athletic Attic are provided below. THE ATHLETIC ATTIC Income Statements For the Years Ended December 31 2025 2024 $ 10,000,000 Net sales Cost of goods...
-
-x/2 x/4 If A = -x/2 and A-1 =6 then x equals
-
The reaction 8H2(g) + S8(l) 8H2S(g) is run at 125oC and a constant pressure of 12.0 atm. Assuming complete reaction, what mass of S8 would be required to produce 6.00 102 mL of H2S gas under these...
-
Use the electron-pair repulsion model to predict the geometry of the following molecules: a. GeCl2 b. NF3 c. SCl2 d. XeO4
-
What reaction was used by Priestley in preparing pure oxygen?
-
Today mutual funds are very popular investment options. However, since their origin in 1924, they have fallen in and out of favor. Write About It Research and write a one-page essay on the history of...
-
You own 12 20-year General Electric Capital Services corporate bonds with face values of $1,000. Their coupon rate is 7.5%. Their current yield is 5.2%. 1. Calculate (a) What is the current market...
-
The prospectus of a mutual fund is where you fi nd out about the structure, fees, and rules of investing in the fund.

Study smarter with the SolutionInn App


