(1) Predict the major organic product of these reactions. In some cases, the formula of the...
Fantastic news! We've Found the answer you've been seeking!
Question:
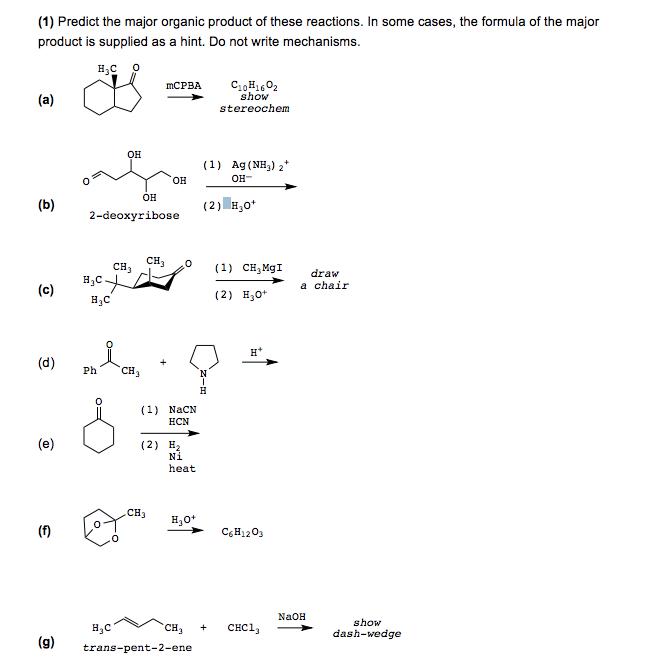
Transcribed Image Text:
(1) Predict the major organic product of these reactions. In some cases, the formula of the major product is supplied as a hint. Do not write mechanisms. C,H02 show stereochem MCPBA (a) он (1) Ag(NH,) 2* OH OH- он (b) (2) H,0* 2-deoxyribose CH3 сн, H,C- н,с (1) CH,MgI draw a chair (c) (2) H,0* H* (d) Ph CH3 (1) NACN НCN (e) (2) H2 Ni heat Cнз H,0* (f) C,H1203 NAOH show Cн, снC1, dash-wedge (g) trans-pent-2-ene (1) Predict the major organic product of these reactions. In some cases, the formula of the major product is supplied as a hint. Do not write mechanisms. C,H02 show stereochem MCPBA (a) он (1) Ag(NH,) 2* OH OH- он (b) (2) H,0* 2-deoxyribose CH3 сн, H,C- н,с (1) CH,MgI draw a chair (c) (2) H,0* H* (d) Ph CH3 (1) NACN НCN (e) (2) H2 Ni heat Cнз H,0* (f) C,H1203 NAOH show Cн, снC1, dash-wedge (g) trans-pent-2-ene
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Predict the major organic product of each of the following reactions. In spite of the structural complexity of some of the starting materials, the functional group transformations are all of the type...
-
Predict the major organic product of each of the following reactions. In spite of the structural complexity of some of the starting materials, the functional group transformations are all of the type...
-
Write the structure of the major organic product formed in the reaction of hydrogen chloride with each of the following: (a) 2-Methyl-2-butene (b) 2-Methyl-1-butene (c) cis-2-Butene
-
PC Connection is a leading mail order retailer of personal computers. A recent financial report issued by the company revealed the following information: Merchandise inventory (beginning of the...
-
When an order is placed with a company, there is a probability of 0.2 that it is an express order. Estimate the probability that 90 or more of the next 400 orders will be express orders.
-
Consider the following MIPS loop: 1. Assume that the register $t1 is initialized to the value 10. What is the value in register $s2 assuming $s2 is initially zero? 2. For each of the loops above,...
-
Discuss the sampling strategy and technique to be used. Develop a hypothetical research scenario that would necessitate the use of the Action Research Approach and a Posttest With a Historical...
-
At December 31, 2011, Craig Corporation reported these plant assets. During 2012, the following selected cash transactions occurred.Apr. 1 Purchased land for $2,600,000.May 1 Sold equipment that cost...
-
It took 6 seconds for the sound of a whistle to reach an observer 5400 feet away from the whistle. At what rate did the sound travel in feet per minute?
-
Lydia Hartley, manager of UltraProducts New Zealand Division, is trying to set the production schedule for the last quarter of the year. The New Zealand Division had planned to sell 100,000 units...
-
Two particles collide with a normal direction defined: 0.8i+0.6j (eT-0.6i+-0.8j). The first mass has a mass of 7 kg and a velocity of 8i m/s. The second mass has a mass of 9 kg and a velocity of...
-
9. In the management of business, managers develop and rely on various types of plans. Which type of plan is associated with the accomplishment of long-term goals? * Tactical plan Contingency plan ...
-
Based on the following information, Salaries and Wages: $18,944 Utilities: $4,726 Net Rental Income: $262,968 Real Estate Taxes: $28,273 General & Administrative: $18,921 Other Income: $15,435...
-
I have a question on setters and getters and the 'this' keyword. So if I have a public void named public void setTime(int time), then can I have anything in the method I want like the calculations...
-
A bond with a par value of $100 is currently trading at a price of $104. The bond has a coupon rate of 4.16%, and it has exactly 10 years remaining until maturity. What is the bonds current yield? An...
-
Woods Forestry Inc. (WFI) is a privately owned integrated forestry company. The shares are currently for sale for $40 million. Bob met with the shareholder and managers of WFI and prepared a summary...
-
Mateo is a single taxpayer who had the following income and loss during 2023: Wages of $60,000. Dividend income of $5,000. Rental loss of $9,000. The loss was from a rental property that Mateo owned....
-
Linda Lopez opened a beauty studio, Lindas Salon, on January 2, 2011. The salon also sells beauty supplies. In January 2012, Lopez realized she had never filed any tax reports for her business and...
-
Which one of the dichlorobenzene isomers does not have a dipole moment? Which one has the largest dipole moment? Compare your answers with the dipole moments calculated using the molecular-modeling...
-
When compounds of the type represented by A are allowed to stand in pentane, they are converted to a constitutional isomer. Hydrolysis of either A or B yields RNHCH2CH2OH and p-nitrobenzoic acid....
-
Suggest a suitable series of reactions for carrying out each of the following synthetic transformations: CH CH32 CO-H to SO3H CH3 CO H CH to (CH3)3 CH C to 0 OCH OCH O-N to OCH C(CH)s
-
When a pair of charged particles are brought twice as close to each other, the force between them becomes (a) twice as strong. (b) four times as strong. (c) half as strong. (d) one-quarter as strong.
-
The electric force of attraction between an electron and a proton is greater on (a) the proton. (b) the electron. (c) neither the proton nor the electron; both are the same. (d) none of these
-
When you double the voltage in a simple electric circuit, you double (a) the current. (b) the resistance. (c) both the current and the resistance. (d) neither the current nor the resistance.

Study smarter with the SolutionInn App


