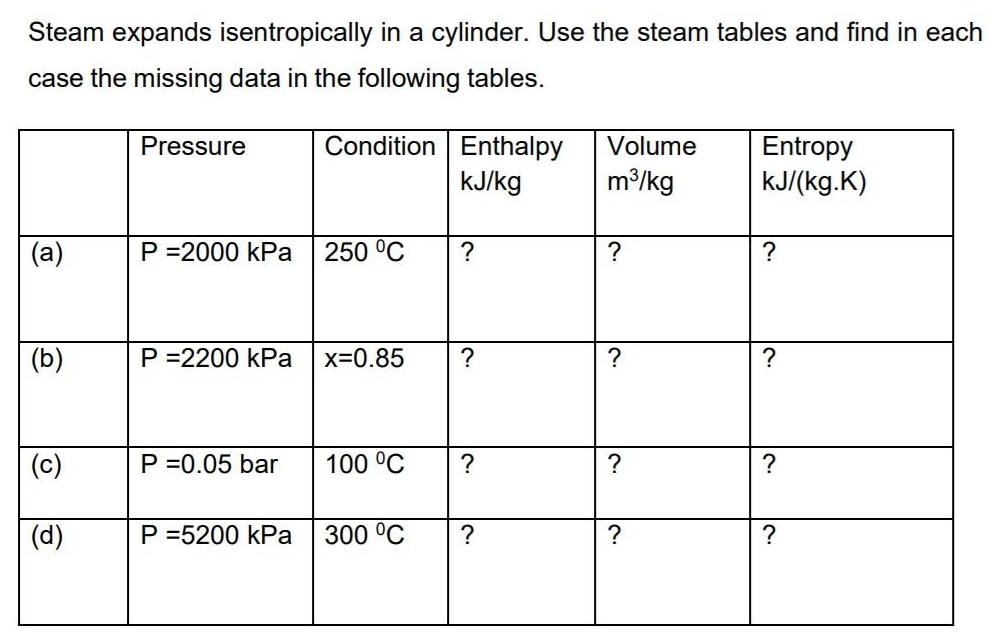
Steam expands isentropically in a cylinder. Use the steam tables and find in each case the...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Steam expands isentropically in a cylinder. Use the steam tables and find in each case the missing data in the following tables. Pressure Condition Enthalpy Volume Entropy kJ/(kg.K) KJ/kg m/kg (a) P =2000 kPa 250 °C ? ? (b) P =2200 kPa x=0.85 (c) P =0.05 bar 100 °C ? |(d) P =5200 kPa 300 °C Steam expands isentropically in a cylinder. Use the steam tables and find in each case the missing data in the following tables. Pressure Condition Enthalpy Volume Entropy kJ/(kg.K) KJ/kg m/kg (a) P =2000 kPa 250 °C ? ? (b) P =2200 kPa x=0.85 (c) P =0.05 bar 100 °C ? |(d) P =5200 kPa 300 °C
Expert Answer:
Answer rating: 100% (QA)
Solution 1 The data are taken from superheated steam properties in stea... View the full answer

Related Book For 

Introduction to Chemical Engineering Thermodynamics
ISBN: 978-0071247085
7th edition
Authors: J. M. Smith, H. C. Van Ness, M. M. Abbott
Posted Date:
Students also viewed these chemical engineering questions
-
Steam expands isentropically in a converging/diverging nozzle from inlet conditions of 1,400 kPa. 325oC and negligible velocity to a discharge pressure of 140 kPa. At the throat the cross-sectional...
-
Use the probability tables and sketch the graph of each of the following binomial distributions. Note on the graph where the mean of the distribution falls. a. n = 6 and p = .70 b. n = 20 and p = .50...
-
Enthalpy of steam at 75 kPa and 573 K is 3075 kJkg referred to liquid water at 273 K If the mean heat capacity of liquid water and water vapor are 42 and 197 kJkg K respectively, calculate the heat...
-
How do accounting differences impact the usefulness of financial ratio comparisons?
-
Sydney Siebenthaler, the audit manager for Jennifers Running Shirts Inc., has just returned from a continuing education class on audit sampling and now wants to use discovery sampling or sequential...
-
On January 1, 2007 the Powder Company listed the following stockholders equity section of its balance sheet: Contributed Capital Preferred stock, $100 par .............. $ 92,800 Common stock , $5...
-
Refer to the chapters opening feature, Sweet Success, describing the entrepreneurial efforts of Dylan Lauren and Jeff Rubin with Dylans Candy Bar. Required List the seven principles of internal...
-
1. Do you see any problems with Philip Austins plan for European expansion? Do you support his entrepreneurial approach to exporting? What should be the features of a more systematic approach to...
-
1.1 Smartphones and Spouses. Choose a random sample of 100 married couples. Ask each member of the couple how much time they spend looking at their smartphone while at home. Compare the mean times...
-
The accounts of Hernandez Computer Repair, Inc., and their normal balances at March 31, 2018, follow. The accounts are listed in no particular order. Requirements 1. Prepare the company?s trial...
-
Problem 8-49 (LO 8-1) (Algo) In 2021, Jasmine and Thomas, a married couple, had taxable income of $113,500. If they were to file separate tax returns, Jasmine would have reported taxable income of...
-
Presented below are variable costing income statements for Sunland Company and Carla Vista Company. They are in the same industry, with the same net incomes, but different cost structures. Sunland...
-
Calculate the fair market value of an investment that pays $180 at the end of each quarter for 10 years. Use a discount rate of j12=6%. Your Answer:
-
Beginning inventory, purchases, and sales for Item 88-HX are as follows: July 1 Inventory July 8 Sale 82 units @ $27 66 units July 15 Purchase July 27 Sale 91 units @ $30 76 units Assuming a...
-
Explain how an organization can use successful project management strategies to ensure its project remains on schedule and under budget. What examples of project management (places where there was an...
-
Read the Danshui Plant 2 case in your Course Pack, Using budget data, how many Apple iPhone 4's would have to have been completed for Danshui Plant 2 to break-even? Using budget data, what was the...
-
A) MFRS 107 requires entities to prepare a statement of cash flows as a component of financial statements. Required: Discuss TWO (2) objectives of Cash Flow Statements. (5 marks) (CLO3:PLO7:C3) B)...
-
Consider the combustion of methanol below. If 64 grams of methanol reacts with 160 grams of oxygen, what is the CHANGE in volume at STP. 2CH3OH(g) + 3O2(g) 2CO2(g) + 4H2O(1) The volume decreases by...
-
Natural gases arc rarely pure methane; they usually also contain other light hydrocarbons and nitrogen. Determine on expression for the standard heat of combustion as a function of composition for a...
-
Assuming Raoult's law to apply to the system n-pentane (1) In-hcptand(2), (a) What arc the values of x1 and y1 at t = 55C und P = 1/2 (P1sat + P2sat)? For these conditions plot the fraction of system...
-
A closed, nonconducting, horizontal cylinder is fitted with a nonconducting, friction* less, flouting piston which divides the cylinder into Sections A and B. The two sections contain equal masses of...
-
How do teams work?
-
Task characteristics, reward systems, and team size are all ____________ that can make a difference in group effectiveness. (a) group processes (b) group dynamics (c) group inputs (d) human resource...
-
When a new team member is anxious about questions such as Will I be able to influence what takes place? the underlying issue is one of ____________. (a) relationships (b) goals (c) processes (d)...

Study smarter with the SolutionInn App


