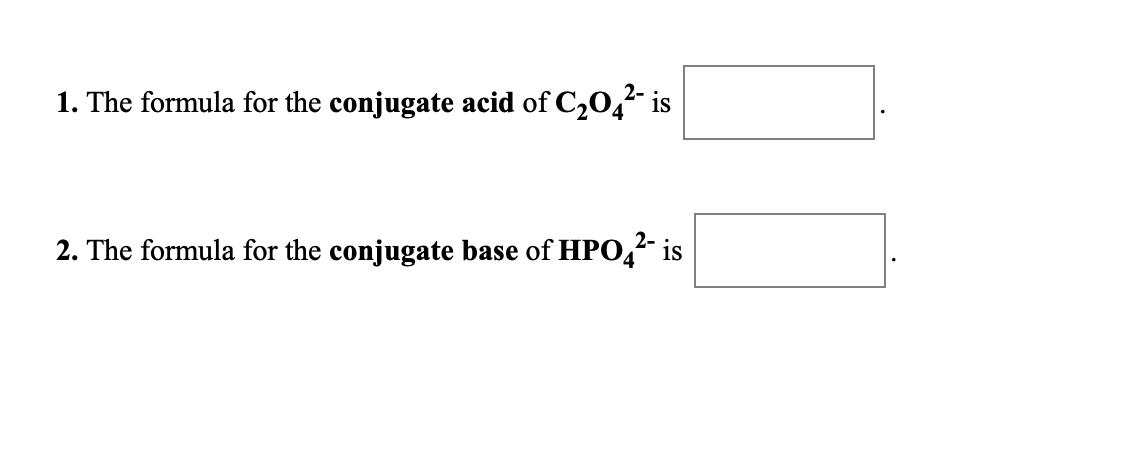
1. The formula for the conjugate acid of C20, is 2. The formula for the conjugate...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
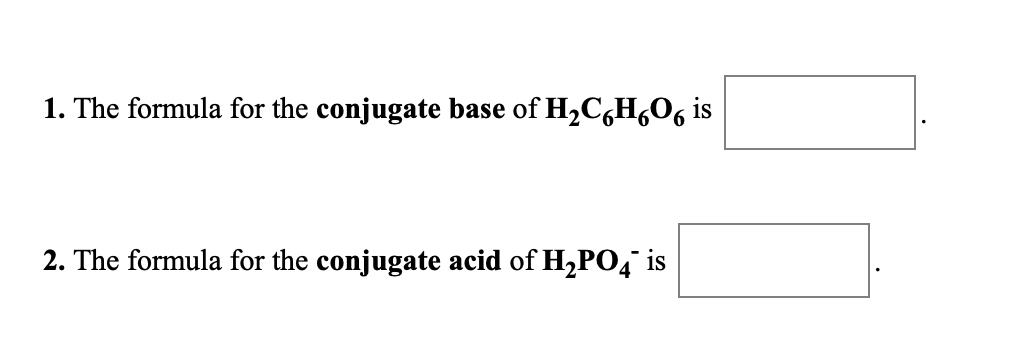
1. The formula for the conjugate acid of C20, is 2. The formula for the conjugate base of HPO, is 1. The formula for the conjugate base of HBr is + 2. The formula for the conjugate base of NH," is 1. The formula for the conjugate base of H2C,H,06 is 2. The formula for the conjugate acid of H2PO4 is 1. The formula for the conjugate acid of C20, is 2. The formula for the conjugate base of HPO, is 1. The formula for the conjugate base of HBr is + 2. The formula for the conjugate base of NH," is 1. The formula for the conjugate base of H2C,H,06 is 2. The formula for the conjugate acid of H2PO4 is 1. The formula for the conjugate acid of C20, is 2. The formula for the conjugate base of HPO, is 1. The formula for the conjugate base of HBr is + 2. The formula for the conjugate base of NH," is 1. The formula for the conjugate base of H2C,H,06 is 2. The formula for the conjugate acid of H2PO4 is 1. The formula for the conjugate acid of C20, is 2. The formula for the conjugate base of HPO, is 1. The formula for the conjugate base of HBr is + 2. The formula for the conjugate base of NH," is 1. The formula for the conjugate base of H2C,H,06 is 2. The formula for the conjugate acid of H2PO4 is 1. The formula for the conjugate acid of C20, is 2. The formula for the conjugate base of HPO, is 1. The formula for the conjugate base of HBr is + 2. The formula for the conjugate base of NH," is 1. The formula for the conjugate base of H2C,H,06 is 2. The formula for the conjugate acid of H2PO4 is 1. The formula for the conjugate acid of C20, is 2. The formula for the conjugate base of HPO, is 1. The formula for the conjugate base of HBr is + 2. The formula for the conjugate base of NH," is 1. The formula for the conjugate base of H2C,H,06 is 2. The formula for the conjugate acid of H2PO4 is
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these accounting questions
-
Write the formula for the conjugate acid of each of the following bases: (a) HS-, (b) HCO3-, (c) CO32-, (d) H2PO4-, (e) HPO42-, (f) PO43-, (g) HSO4-, (h) SO42-, (i) SO32-.
-
Write the formula for the conjugate acid of each of the following bases: (a) HS-, (b) HCO3-, (c) CO32-, (d) H2PO4, (e) HPO42-, (f) PO43-, (g) HSO4-, (h) SO42-, (i) SO32-.
-
Write the formula for the conjugate base of each of the following acids: (a) CH2ClCOOH, (b) HIO4, (c) H3PO4, (d) H2PO2-4, (e) HPO2-4 (f) H2SO4, (g) HSO4-, (h) HIO3, (i) HSO (j) NH4+, (k) H2S, (l)...
-
23 On January 1, 2021, a company had an existing deferred tax liability attributable to a temporary difference of $20,000. The tax rate at this time was 30%. During 2021, a new law was passed that...
-
For the data in Exercise 1, we want to test the null hypothesis that the mean age of houses in the two neighborhoods is the same. Assume that the data come from a population that is Normally...
-
Suppose there are two transformers between your house and the high-voltage transmission line that distributes the power. In addition, assume that your house is the only one using electric power. At a...
-
Consider the Michaelis-Menten model introduced in Eq. (12.23). Graph the expectation function for this model for \(\theta_{1}=200\) and \(\theta_{2}=0.04,0.06,0.08\), 0.10 . Overlay these curves on...
-
Joanne is a seventy-five-year-old widow who survives on her husbands small pension. Joanne has become increasingly forgetful, and her family worries that she may have Alzheimers disease (a brain...
-
1. Determine the Vave from 0 sec to 3 sec. 2. Determine the Vinst at t= 2 sec. 3. Explain why the answers to questions #1 and #2 are equal. 4. Determine the Vinst at t = 10 sec. 5. Determine the...
-
The general ledger of Zips Storage at January 1, 2024, includes the following account balances: Accounts Debits Credits Cash $26,300 17,100 Accounts Receivable Prepaid Insurance Land 15,400 165,000...
-
Use the calculator provided to solve the following problems: . Consider an E distribution with 48 numerator degrees of freedom and 22 denominator degrees of freedom. Compute P (FS1.91). Round your...
-
The right to die is a touchy subject for most people. In my personal experience people are always hesitant about the Living Will. It is such a serious document. Going through and deciding which...
-
A new client of ours, Guttman Conservation, has requested we sell to it on 60-day credit terms. It is being sued for defamation. I would like to know if this claim has any merit before we ship...
-
You are the Centre Manager for Bounce Fitness Brisbane. You are creating an operational plan to boost profit by introducing a new Bounce Fitness class. Access the following: Bounce Fitness...
-
The states is FLORIDA Research the court-connected ADR program in effect in the federal district that serves your area and provide the following information: What forms of ADR are used in this court...
-
Rights and Remedies upon Default In event of default Secured party may take possession of the goods and sell them to recover the amount owed Creditor usually hires bailiff Right to redeem: debtor...
-
The largest dollar amount that a buyer is willing to pay for a good is the O quantity demanded. O buyer's reservation price. O market price. O seller's reservation price.
-
Find the cross product a x b and verify that it is orthogonal to both a and b. a = (t, 1, 1/t), b = (t 2 , t 2 , 1)
-
Express the following numbers in scientific notation: (a) 0.000000027, (b) 356, (c) 47,764, (d) 0.096.
-
As an approximation, we can assume that proteins exist either in the native (or physiologically functioning) state and the denatured state Native denatured The standard molar enthalpy and entropy of...
-
A certain metal M forms a bromide containing 53.79 percent Br by mass. What is the chemical formula of the compound?
-
What is the change in value for this security over the four quarters recorded in the highlowclose graph?
-
Which of the 10 years had the most and least variability between the maximum sales of womens clothing and the minimum for mens, and what were those ranges?
-
What happened to the average sales figures for jewelry over the 10-year period?

Study smarter with the SolutionInn App



