1. The freezing point and boiling point of acetone are 178.5 K and 329.20 K, respectively,...
Fantastic news! We've Found the answer you've been seeking!
Question:
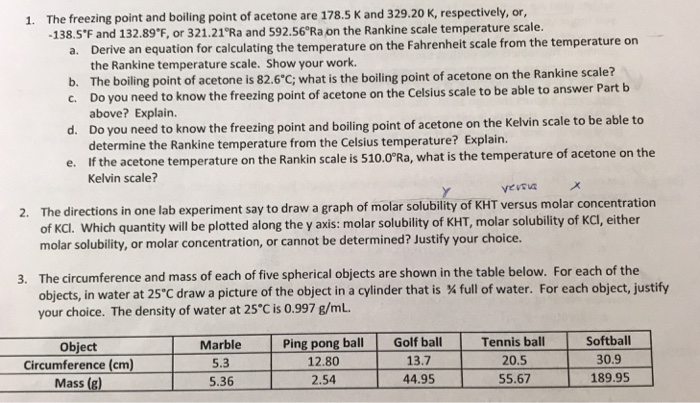
Transcribed Image Text:
1. The freezing point and boiling point of acetone are 178.5 K and 329.20 K, respectively, or, -138.5°F and 132.89°F, or 321.21°Ra and 592.56°Ra on the Rankine scale temperature scale. a. Derive an equation for calculating the temperature on the Fahrenheit scale from the temperature on the Rankine temperature scale. Show your work. b. The boiling point of acetone is 82.6°C; what is the boiling point of acetone on the Rankine scale? C. Do you need to know the freezing point of acetone on the Celsius scale to be able to answer Part b above? Explain. d. Do you need to know the freezing point and boiling point of acetone on the Kelvin scale to be able to determine the Rankine temperature from the Celsius temperature? Explain. e. If the acetone temperature on the Rankin scale is 510.0°Ra, what is the temperature of acetone on the Kelvin scale? Y versva 2. The directions in one lab experiment say to draw a graph of molar solubility of KHT versus molar concentration of KCI. Which quantity will be plotted along the y axis: molar solubility of KHT, molar solubility of KCl, either molar solubility, or molar concentration, or cannot be determined? Justify your choice. 3. The circumference and mass of each of five spherical objects are shown in the table below. For each of the objects, in water at 25°C draw a picture of the object in a cylinder that is % full of water. For each object, justify your choice. The density of water at 25°C is 0.997 g/mL. Object Circumference (cm) Mass (g) Marble 5.3 5.36 Ping pong ball 12.80 2.54 Golf ball 13.7 44.95 Tennis ball 20.5 55.67 Softball 30.9 189.95 1. The freezing point and boiling point of acetone are 178.5 K and 329.20 K, respectively, or, -138.5°F and 132.89°F, or 321.21°Ra and 592.56°Ra on the Rankine scale temperature scale. a. Derive an equation for calculating the temperature on the Fahrenheit scale from the temperature on the Rankine temperature scale. Show your work. b. The boiling point of acetone is 82.6°C; what is the boiling point of acetone on the Rankine scale? C. Do you need to know the freezing point of acetone on the Celsius scale to be able to answer Part b above? Explain. d. Do you need to know the freezing point and boiling point of acetone on the Kelvin scale to be able to determine the Rankine temperature from the Celsius temperature? Explain. e. If the acetone temperature on the Rankin scale is 510.0°Ra, what is the temperature of acetone on the Kelvin scale? Y versva 2. The directions in one lab experiment say to draw a graph of molar solubility of KHT versus molar concentration of KCI. Which quantity will be plotted along the y axis: molar solubility of KHT, molar solubility of KCl, either molar solubility, or molar concentration, or cannot be determined? Justify your choice. 3. The circumference and mass of each of five spherical objects are shown in the table below. For each of the objects, in water at 25°C draw a picture of the object in a cylinder that is % full of water. For each object, justify your choice. The density of water at 25°C is 0.997 g/mL. Object Circumference (cm) Mass (g) Marble 5.3 5.36 Ping pong ball 12.80 2.54 Golf ball 13.7 44.95 Tennis ball 20.5 55.67 Softball 30.9 189.95
Expert Answer:
Answer rating: 100% (QA)
1Fahranite and Rankine scale C F 32 59 1 Therefore we can convert F to C as K C 27315 2 Relati... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
You need to know whether the building of a new warehouse is justified under the following conditions. The proposal is for a warehouse costing $250,000. The warehouse has an expected useful life of...
-
Calculate the freezing point and boiling point of an antifreeze solution that is 50.0 g ethylene glycol (HOCH2CH2OH) in 50.0 g water. Ethylene glycol is a nonelectrolyte.
-
How many parameters do you need to know to completely describe the Otto cycle? How about the Diesel cycle?
-
Every real number is either a/an number or a/an_______ number.
-
Should professional accountants push for the development of a comprehensive framework for the reporting of corporate social performance? Why?
-
Minesweeper, the well-known computer game, is closely related to the wumpus world. A minesweeper world is a rectangular grid of N squares with M invisible mines scattered among them. Any square may...
-
The plaintiff, Thelma Agnes Smith, lived with the defendant out of wedlock for several years. When the relationship ended, she sued the defendant, seeking to enforce two written agreements with him...
-
Janus Products, Inc., is a merchandising company that sells binders, paper, and other school supplies. The company is planning its cash needs for the third quarter. In the past, Janus Products has...
-
Making Decisions with Confidence Intervals Assume you work for Kimberly Clark Corporation, the makers of Kleenex. The job you are presently working on requires you to decide how many Kleenexes are to...
-
Suppose that there are p = 4 quality characteristics, and in correlation form all four variables have variance unity and that all pairwise correlation coefficients are 0.9. The in-control value of...
-
Variable cost and fixed cost a. b. C. d. S f. g. a. b. C. T F A variable cost is a cost that varies with the level of production or output. A fixed cost is a cost that does not vary with the level of...
-
What Division and Section would you find information on brick masonry?
-
Describe a debit, credit , T -account, asset,liability, equity, revenue, and expense.
-
Consider the following income statement for Aldi-Woolworths Inc. (all figures in $ Millions): Year Total Sales Cost of goods sold Selling, General & Admin expenses Depreciation 2014 2015 2016 60,553...
-
What are quantitative ways to measure financial performance at Volkswagen?
-
. Clearview Window Company manufactures windows for the home-building industry. The window frames are produced in the Frame Division. The frames are then transferred to the Glass Division, where the...
-
The following are selected information from Entity A's accounting records: Cash Accounts receivable Inventory Prepaid insurance Accounts payable 20x1 510,000 25,000 65,000 5,000 15,000 20x0 120,000...
-
U.S. households have become smaller over the years. The following table from the 2010 GSS contains information on the number of people currently aged 18 years or older living in a respondent's...
-
A solution is prepared by dissolving table salt, sodium chloride, in water at room temperature. a. Assuming there is no significant change in the volume of water during the preparation of the...
-
You make solutions of ammonia and sodium hydroxide by adding the same moles of each solute to equal volumes of water. Which solution would you expect to have the higher pH?
-
Each of the following statements concerns a 0.10 M solution of a weak organic base, B. Briefly describe why each statement is either true or false. a. [B] is approximately equal to 0.10 M. b. [B] is...
-
In a talk at the White House in December 2009, President Barack Obama argued, Ultimately in this country we rise and fall together: banks and small businesses, consumers and large corporations. Why...
-
An article in the New York Times in mid-2012 noted, with the economy still struggling and new regulations meant to eliminate bad lending, bank loans continue to lag. a. What does the article mean by...
-
What is money? Why do we need money?

Study smarter with the SolutionInn App


