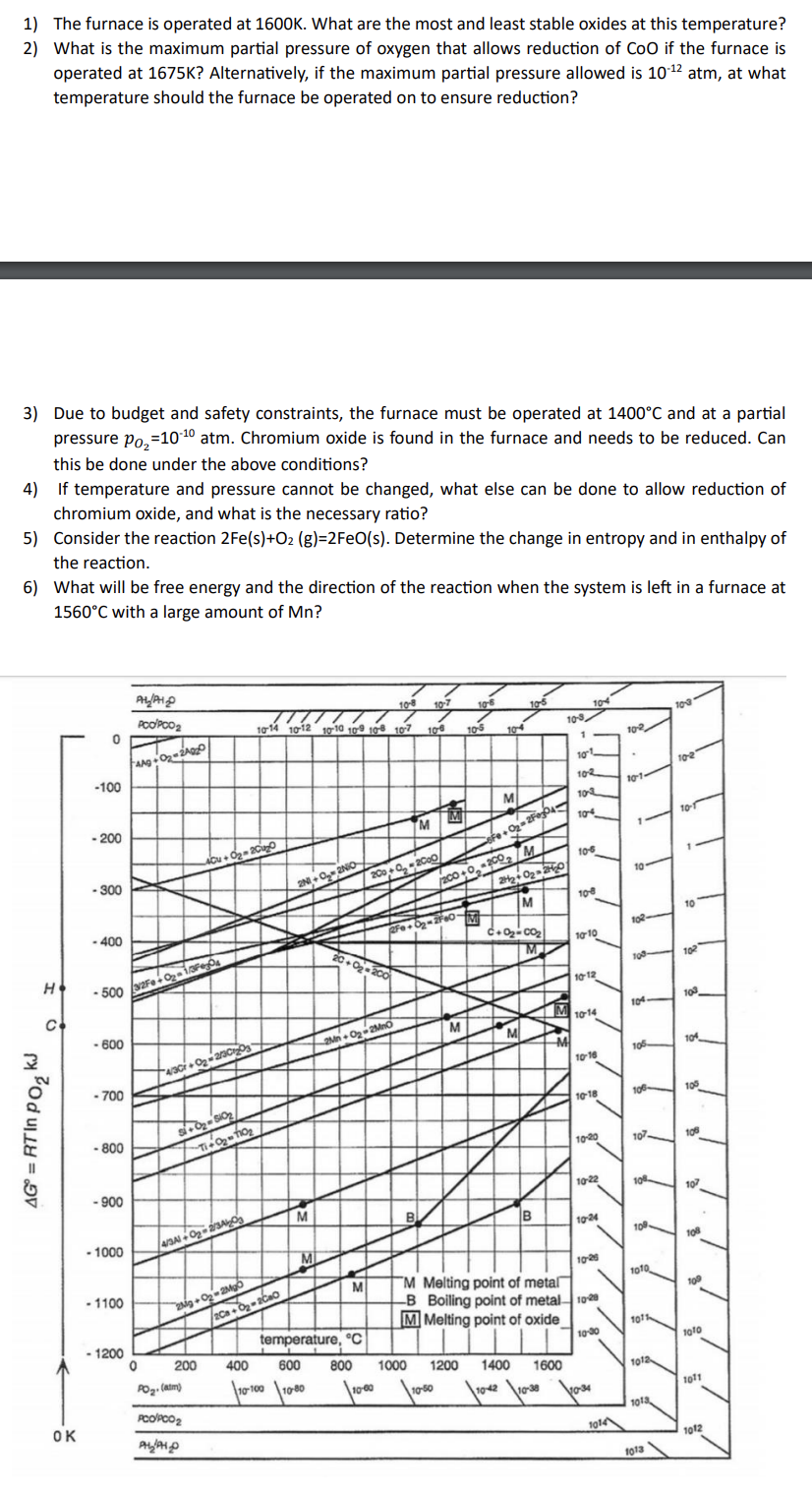
1) The furnace is operated at 1600K. What are the most and least stable oxides at...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
1) The furnace is operated at 1600K. What are the most and least stable oxides at this temperature? 2) What is the maximum partial pressure of oxygen that allows reduction of CoO if the furnace is operated at 1675K? Alternatively, if the maximum partial pressure allowed is 10-2 atm, at what temperature should the furnace be operated on to ensure reduction? 3) Due to budget and safety constraints, the furnace must be operated at 1400C and at a partial pressure Po=10-0 atm. Chromium oxide is found in the furnace and needs to be reduced. Can this be done under the above conditions? 4) If temperature and pressure cannot be changed, what else can be done to allow reduction of chromium oxide, and what is the necessary ratio? 5) Consider the reaction 2Fe(s)+O2 (g)-2FeO(s). Determine the change in entropy and in enthalpy of the reaction. 6) What will be free energy and the direction of the reaction when the system is left in a furnace at 1560C with a large amount of Mn? H . AG = RT IN PO KJ OK 0 -100 - 200 -300 - 400 - 600 -500 3/2Fe+0-1304 - 700 - 800 - 900 - 1000 - 1100 ALPH PCO/PCO Ang+02-24020 - 1200 0 4/30+02-2130203 si-0-S10/ 7-11-0-f10) 40u+0-200 4/3A1+0-2/3A 200 2g+0-2Mgo /20a+0-200 PO (atm) 10-8 10-7 10-6 10-14 10-12 10-10 109 108 107 108 105 104 PCO/PCO PH/PHO 2N+0-2NO M M 200+0-2000 20-0-200 M 2Fe+0=2 2Mn-0-2MnO TM temperature, C 400 600 800 1000 10-100 10-80 10-60 M 10-50 M Fe+0-2F0045 200+0-2002/M 1200 2H+0 20 M C+0-00 M M B 10-3 1 M 104 101 102 102 104 106 10-8 10-10 10-12 10-14 10-16 10-18 10-20 10-22 M Melting point of metal -B Boiling point of metal 1020 M Melting point of oxide 10-24 10:26 10-30 1400 1600 10-4210-38 1034 1014 102 10-1 10- 102 108 104 105 106 107- J 10% 109- 1010 1011 1012 1013 1013 10-3 10-2 10-1 10 102 103 ! 10% 1 100 108 18 107 108 109 1010 1071 1012 1) The furnace is operated at 1600K. What are the most and least stable oxides at this temperature? 2) What is the maximum partial pressure of oxygen that allows reduction of CoO if the furnace is operated at 1675K? Alternatively, if the maximum partial pressure allowed is 10-2 atm, at what temperature should the furnace be operated on to ensure reduction? 3) Due to budget and safety constraints, the furnace must be operated at 1400C and at a partial pressure Po=10-0 atm. Chromium oxide is found in the furnace and needs to be reduced. Can this be done under the above conditions? 4) If temperature and pressure cannot be changed, what else can be done to allow reduction of chromium oxide, and what is the necessary ratio? 5) Consider the reaction 2Fe(s)+O2 (g)-2FeO(s). Determine the change in entropy and in enthalpy of the reaction. 6) What will be free energy and the direction of the reaction when the system is left in a furnace at 1560C with a large amount of Mn? H . AG = RT IN PO KJ OK 0 -100 - 200 -300 - 400 - 600 -500 3/2Fe+0-1304 - 700 - 800 - 900 - 1000 - 1100 ALPH PCO/PCO Ang+02-24020 - 1200 0 4/30+02-2130203 si-0-S10/ 7-11-0-f10) 40u+0-200 4/3A1+0-2/3A 200 2g+0-2Mgo /20a+0-200 PO (atm) 10-8 10-7 10-6 10-14 10-12 10-10 109 108 107 108 105 104 PCO/PCO PH/PHO 2N+0-2NO M M 200+0-2000 20-0-200 M 2Fe+0=2 2Mn-0-2MnO TM temperature, C 400 600 800 1000 10-100 10-80 10-60 M 10-50 M Fe+0-2F0045 200+0-2002/M 1200 2H+0 20 M C+0-00 M M B 10-3 1 M 104 101 102 102 104 106 10-8 10-10 10-12 10-14 10-16 10-18 10-20 10-22 M Melting point of metal -B Boiling point of metal 1020 M Melting point of oxide 10-24 10:26 10-30 1400 1600 10-4210-38 1034 1014 102 10-1 10- 102 108 104 105 106 107- J 10% 109- 1010 1011 1012 1013 1013 10-3 10-2 10-1 10 102 103 ! 10% 1 100 108 18 107 108 109 1010 1071 1012
Expert Answer:
Answer rating: 100% (QA)
To address your questions 1 At 1600K the most stable oxide is usually the one with the highest negative Gibbs free energy of formation G Conversely th... View the full answer

Related Book For 

Posted Date:
Students also viewed these mechanical engineering questions
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
The gas laws are vitally important to scuba divers. The pressure exerted by 33 ft of seawater is equivalent to 1 atm pressure. (a) A diver ascends quickly to the surface of the water from a depth of...
-
Why have some firms gone to voluntary benefits as opposed to discretionary benefits?
-
Fill in the Table below for a combination of two unequal resistors of resistance R1 and R2. Assume the electric potential on the low-voltage end of the combination is VA volts and the potential at...
-
The diffusivity of toluene in air was determined experimentally by allowing liquid toluene to vaporize isothermally into air from a partially filled vertical tube 3 mm in diameter. At a temperature...
-
The electromagnetic waves that carry FM radio range in frequency from \(87.9 \mathrm{MHz}\) to \(107.9 \mathrm{MHz}\). What is the range of wavelengths of these radio waves? A. \(500-750...
-
Ronald Thump is interested in expanding his firm. After careful consideration, he has determined three areas in which he might invest additional funds: (1) product research and development, (2)...
-
6) Draw a timing diagram for x1, x2, x3, and f, for the circuit shown in the figure. x2 x3 x D D f
-
When you buy paint from the stores ( ( such as Home Depot or Lowe's ) , ) , the store employees are most likely to mix some basic colors available in the stores using a machine to get the exact color...
-
What is the purpose of an engagement letter? What should be included in an engagement letter?
-
Name two ratios that are used to measure financial leverage and write their equations.
-
Are you convinced by the economists' arguments of the extent to which factor substitution and technological advances can be used to ameliorate the scarcity of environmental resources? Provide...
-
What is the bid-ask spread?
-
Professional behaviour means that members of the professional body must: a. maintain their knowledge and skill at the required level. b. keep up to date with changes in regulations and standards. c....
-
Using the graph below, indicate domestic production and net imports. a. Would the United States want to raise or lower the world supply of the good? Why? b. How might that happen? Sw Pw D Quantity...
-
Define relevant costs and discuss: (1) whether all future costs are relevant for decision making and (2) whether variable costs are always relevant and fixed costs are always irrelevant
-
The boiling points of neon and krypton are 2245.9C and 2152.9C, respectively. Using these data, estimate the boiling point of argon.
-
Calculate the heats of combustion for the following reactions from the standard enthalpies of formation listed in Appendix 3: (a) 2H2(g) + O2(g) - 2H2O(l) (b) 2C2H2(g) + 5O2(g) - 4CO2(g) + 2H2O(l)
-
Active transport is the process in which a substance is transferred from a region of lower concentration to one of higher concentration. This is a non spontaneous process and must be coupled to a...
-
What were Aristotles two main classifications of motion?
-
Which depends on gravityweight or mass?
-
What is the name of the property of objects to maintain their states of motion?

Study smarter with the SolutionInn App


