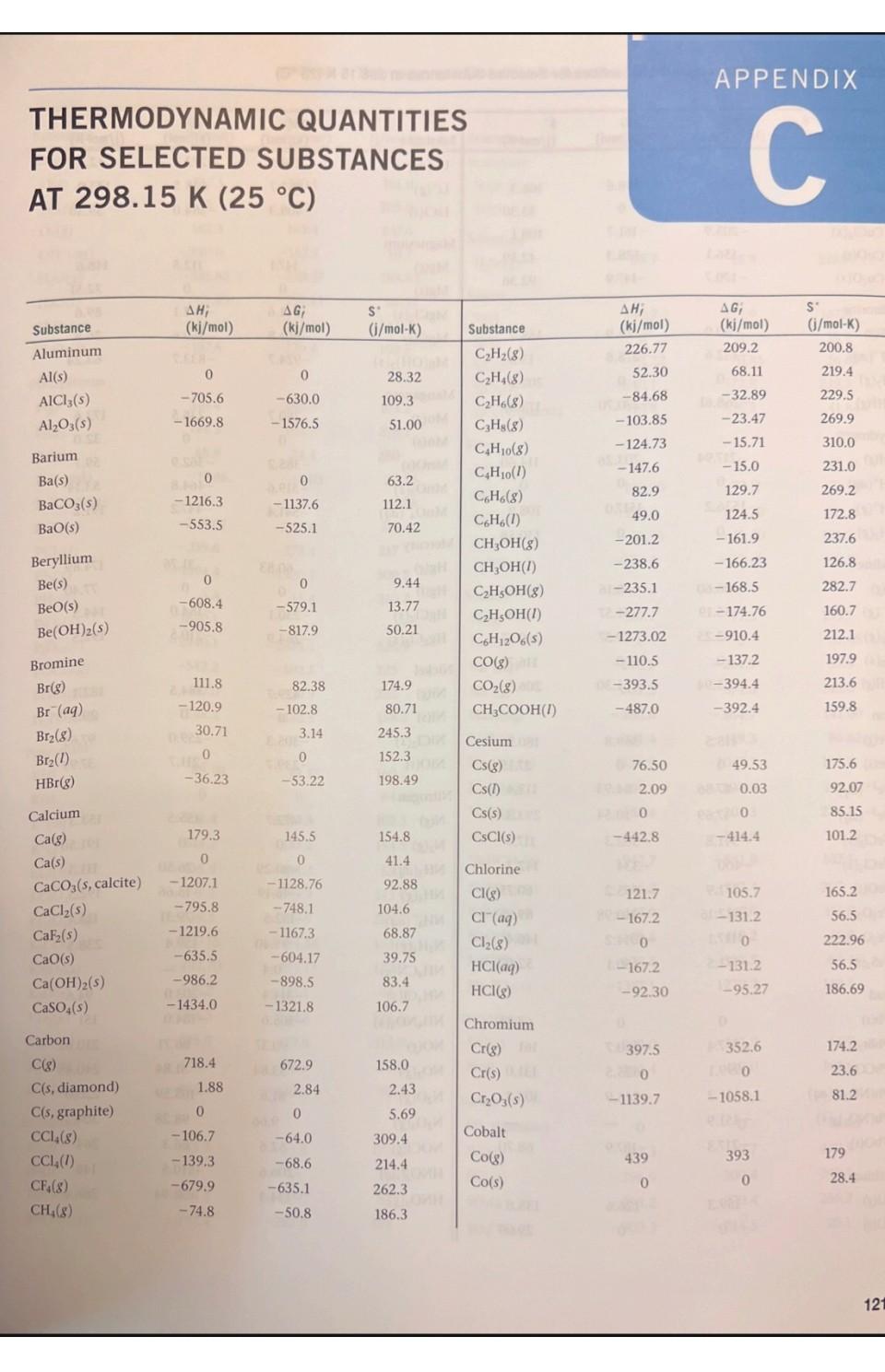
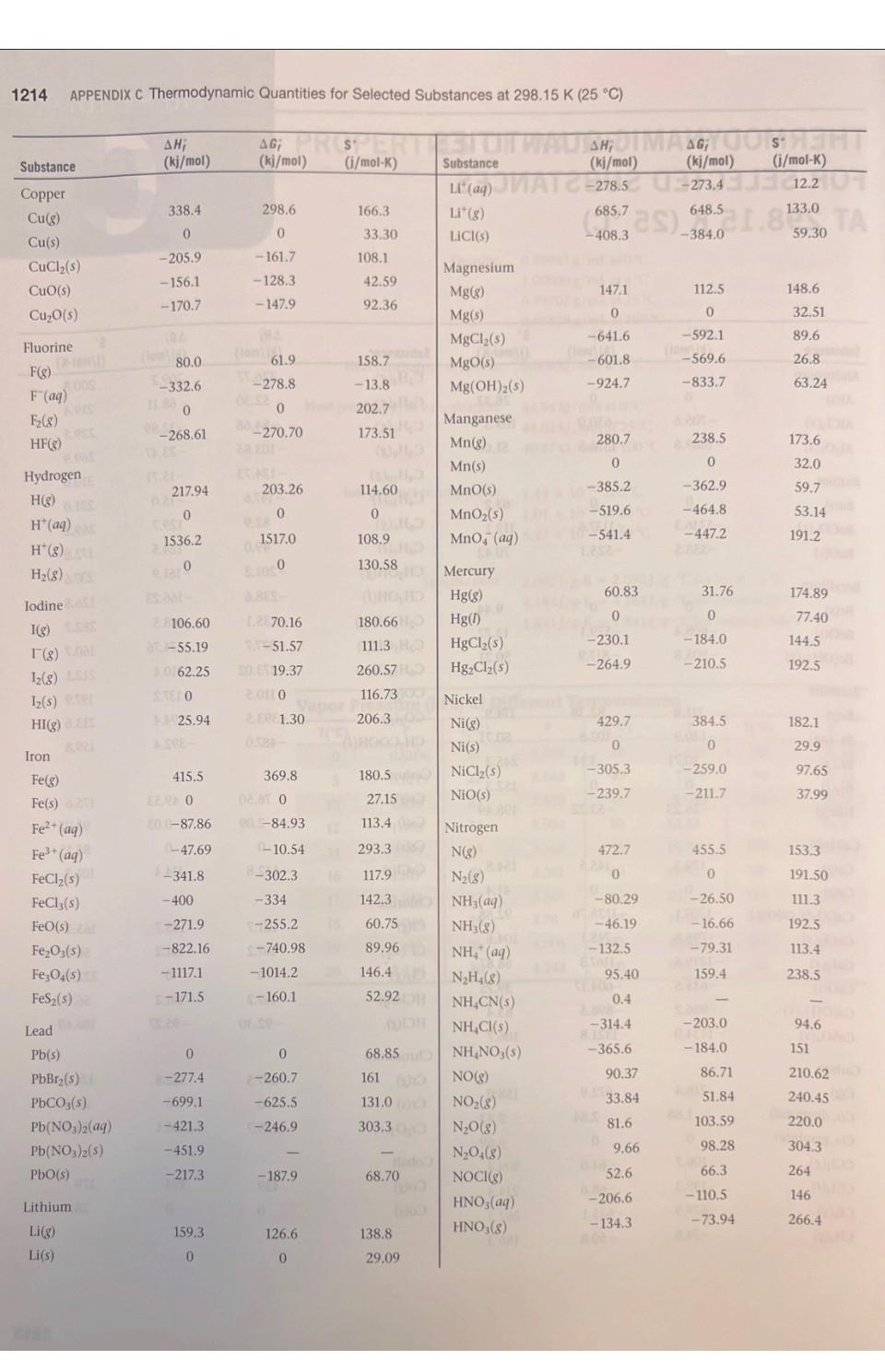
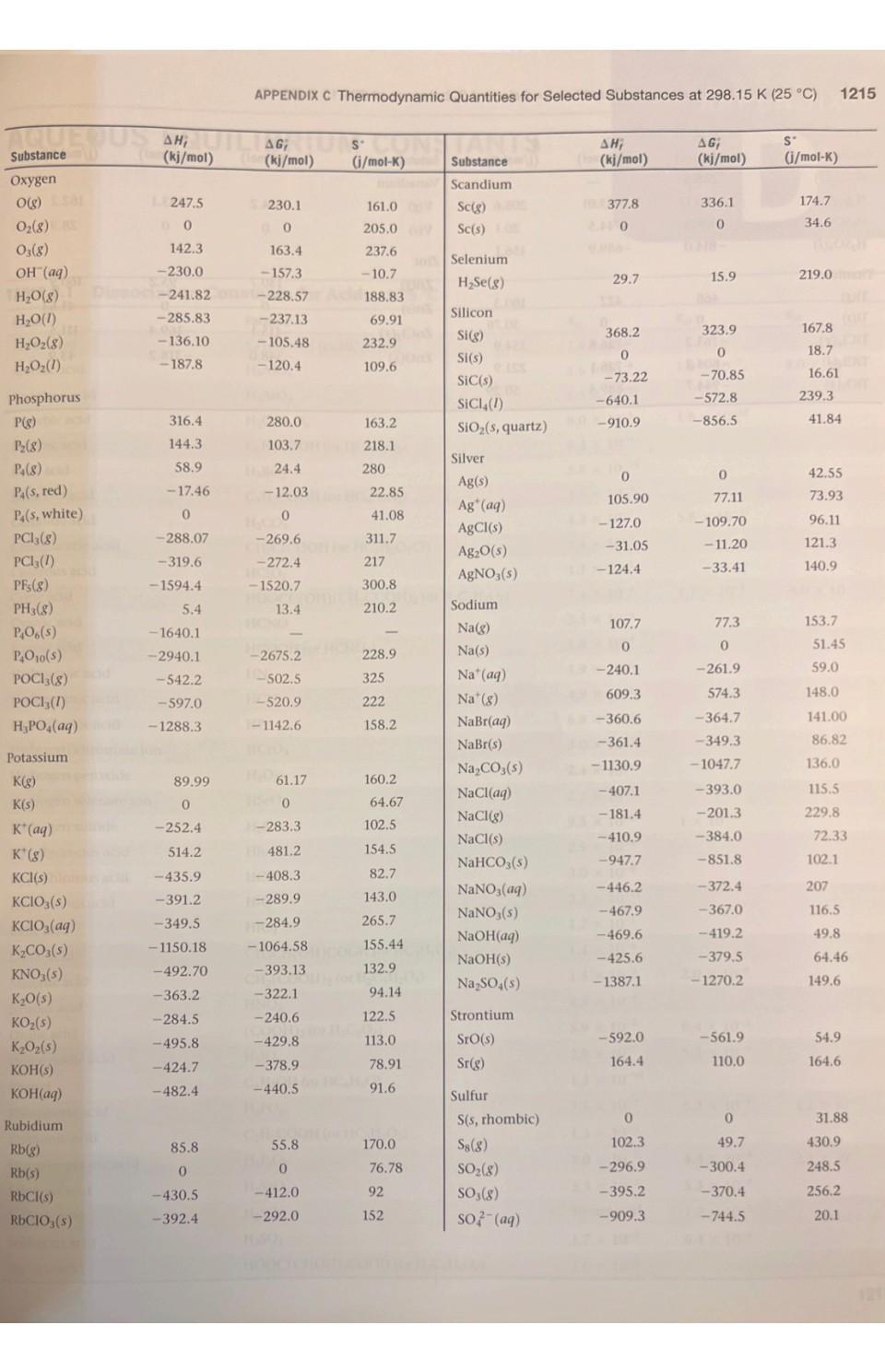
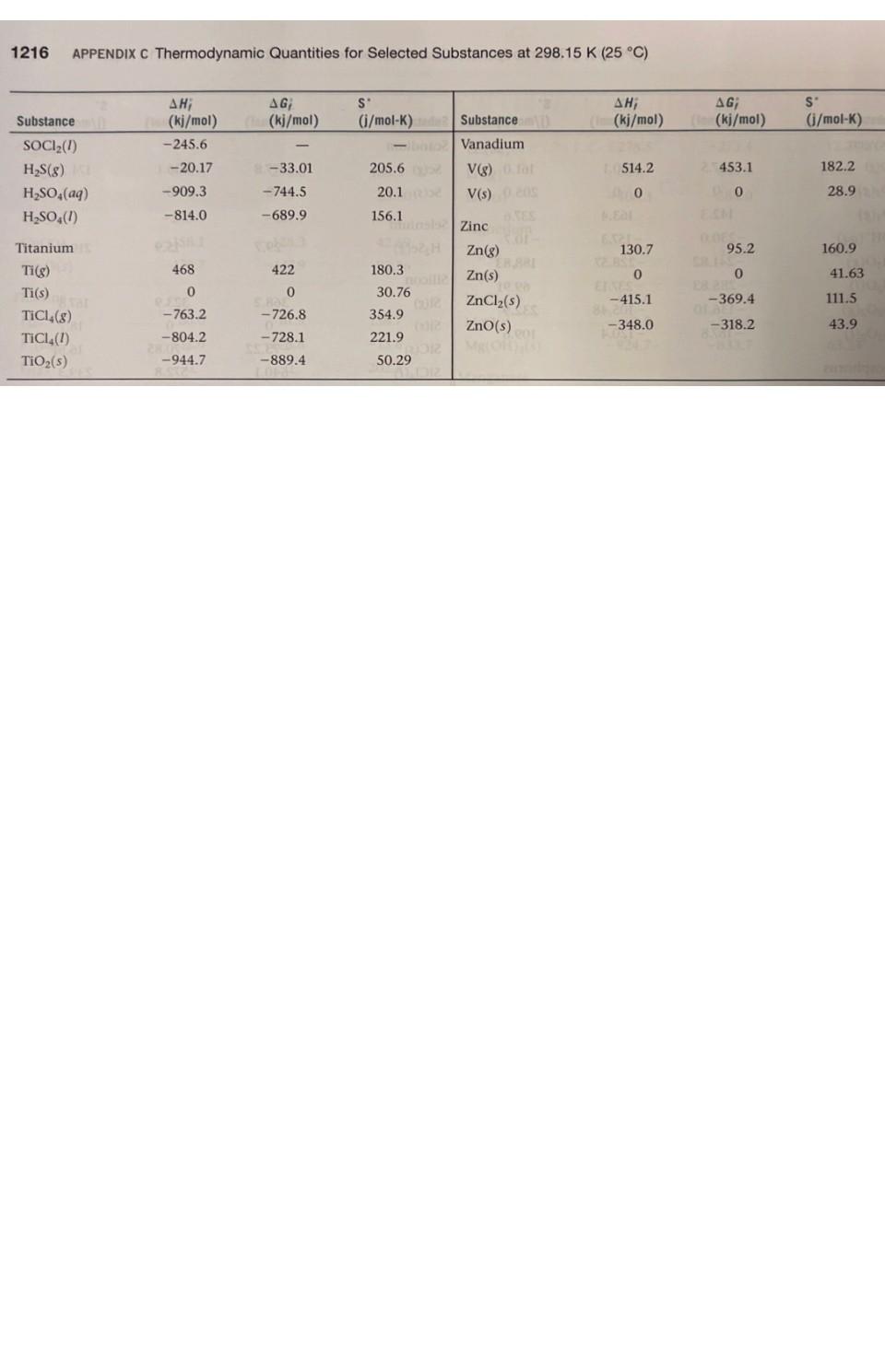
1. Using data in Appendix C, calculate AH, AS', and AG at 298K for each of...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
1. Using data in Appendix C, calculate AH, AS', and AG at 298K for each of the following reactions. Indicate whether the reaction is spontaneous at 298K under standard conditions. (a) Ti (s) + 2 Cl₂(g) → TiCl4 (1) (b) HCI (aq) + NaOH (aq) → NaCl (aq) + H₂O (1) (c) CF4 (g) + 2 H₂ (g) → C(s) + 4 HF (g) (d) Na₂CO3 (s) + 2 HNO3(aq) → 2 NaNO3(aq) + CO₂ (g) + H₂O (1) THERMODYNAMIC QUANTITIES FOR SELECTED SUBSTANCES AT 298.15 K (25 °C) Substance Aluminum Al(s) AICI3(s) Al₂O3(s) Barium Ba(s) BaCO3(s) BaO(s) Beryllium Be(s) BeO(s) Be (OH)2 (s) Bromine Br(g) Br (aq) Br₂(8) Br₂(1) HBr(g) Calcium Ca(g) Ca(s) CaCO3(s, calcite) CaCl₂(s) CaF₂(s) CaO(s) Ca(OH)2(s) CaSO4(s) Carbon C(g) C(s, diamond) C(s, graphite) CC14(8) CC1₂ (1) CF4(8) CH₂(8) AH; (kj/mol) 0 -705.6 -1669.8 0 -1216.3 -553.5 0 -608.4 -905.8 111.8 - 120.9 30.71 -36.23 179.3 0 -1207.1 -795.8 -1219.6 -635.5 -986.2 -1434.0 9718.4 1.88 0 -106.7 -139.3 -679.9 -74.8 AG; (kj/mol) 0 -630.0 -1576.5 0 - 1137.6 -525.1 0 -579.1 -817.9 82.38 - 102.8 3.14 0 -53.22 145.5 0 -1128.76 -748.1 -1167.3 -604.17 -898.5 -1321.8 672.9 2.84 0 -64.0 -68.6 -635.1 -50.8 S (j/mol-K) 28.32 109.3 51.00 63.2 112.1 70.42 9.44 13.77 50.21 174.9 80.71 6H 245.3 152.3 198.49 154.8 41.4 92.88 104.6 68.87 39.75 83.4 106.7 158.0 309.4 214.4 262.3 186.3 2.43 5.69 Will Substance C₂H₂(8) C₂H4(8) C₂H6(8) C3H8(8) C4H10(8) C4H10(1) C6H6(8) C6H6(1) CH₂OH(g) CH₂OH(1) C₂H5OH (8) C₂H₂OH(1) C6H12O6(S) CO(g) CO₂(8) CH3COOH(1) Cesium08 Cs(g) Cs(1) Cs(s) ELLIPS CsCl(s) Chlorine CI(g) HW CI (aq) Cl₂(8) 11.8 HCl(aq) HCI(g) Chromium Cr(g) Cr(s) E Cr₂O3(s) Cobalt Co(g) Co(s) B AH; (kj/mol) 226.77 52.30 -84.68 -103.85 -124.73 -147.6 82.9 49.0 -201.2 -238.6 al-235.1 -277.7 -1273.02 -110.5 06-393.5 -487.0 76.50 2.09 1200 -442.8 121.7 SHOO 1-167.2 -92.30 397.5 22000 -1139.7 APPENDIX C 439 0 AG; (kj/mol) 209.2 68.11 -32.89 -23.47 -15.71 -15.0 129.7 124.5 -161.9 -166.23 08-168.5 1-174.76 25-910.4 -137.2 40-394.4 -392.4 8-167.2-131.2 49.53 08 0.03 eat200 -414.4 105.7 150 2-131.2 -95.27 D 352.6 LABO -1058.1 EXIDE 393 0 S' (j/mol-K) 200.8 219.4 229.5 269.9 310.0 231.0 269.2 172.8 237.6 126.8 282.7 160.7 212.1 197.9 213.6 159.8 175.6 92.07 85.15 101.2 165.2 56.5 222.96 56.5 186.69 174.2 23.6 81.2 179 28.4 121 1214 APPENDIX C Thermodynamic Quantities for Selected Substances at 298.15 K (25 °C) Substance Copper Cu(g) Cu(s) CuCl₂(s) CuO(s) Cu₂O(s) Fluorine F(g) F (aq) F₂(8) HF(g) Hydrogen H(g) H*(aq) H*(8) H₂(8) ES lodine 051 I(g) 58 (8) 00 12(8) LESS 1₂(s) e HI(g) E Iron Fe(g) Fe(s) 21 Fe²+ (aq) Fe³+ (aq) FeCl₂(s) FeCl3(s) FeO(s) zal Fe₂O3(s) Fe3O4(s) FeS₂(s) Lead Pb(s) PbBr₂(S) PbCO3(s) 2 Pb(NO3)2(aq) Pb(NO3)2(S) PbO(s) Lithium Li(g) Li(s) AH; (kj/mol) 338.4 0 -205.9 -156.1 -170.7 80.0 -332.6 0 -268.61 217.94 0 1536.2 0 28106.60 8-55.19 A0162.25 STEL O 25.94 415.5 EERO 20.0-87.86 (-47.69 -341.8 -400 -271.9 -822.16 -1117.1 1-171.5 0 -277.4 -699.1 -421.3 -451.9 -217.3 159.3 0 (fo AG; PROSPERT (kj/mol) (i/mol-K) 298.6 0 -161.7 -128.3 -147.9 61.9 -278.8 0 88-270.70 203.26 0 1517.0 8.1000 Le 70.16 1.-51.57 50. 19.37 20110 ERE 1.30 369.8 02.81 0 POL-84.93 (-10.54 8-302.3 -334 -255.2 -740.98 -1014.2 -160.1 0 2-260.7 -625.5 -246.9 -187.9 126.6 0 166.3 33.30 108.1 42.59 92.36 158.7 -13.8 202.7 173.51 114.60 0 108.9 130.58 1000 HD 180.66 111.3 HO 260.57 H 116.737 206.30 OID 180.5) 27.15 113.4 293.3 117.9 142.3 del 60.75 89.96 146.45 52.92 68.85 161 ( 131.0 303.300 68.70 Hid 138.8 DH 29.09 STUIT WAUTO; DI MAGYOOSHEHT Substance (kj/mol) (kj/mol) (i/mol-K) LI (aq)MATC-278.5-273.4 12.20 Li* (g) LICI(s) Magnesium Mg(g) Mg(s) MgCl₂(s) MgO(s) Mg(OH)₂(s) Manganese Mn(g) Mn(s) MnO(s) MnO₂ (s) MnO4 (aq) Mercury Hg(g) Hg(1) HgCl₂(s) Hg₂Cl₂ (s) Nickel Ni(g) Ni(s) NiCl₂(s) NIO(s) Nitrogen N(g) N₂ (8) NH3(aq) NH3(g) NH, (aq) N₂H₂(8) NH₂CN(S) NH₂Cl(s) NH₂NO3(s) NO(g) NO₂(8) N₂O(g) N₂O4(8) NOCI(g) HNO3(aq) HNO3 (8) 686725) 648.51.833,00 TA -408.3 147.1 0 -641.6 -601.8 -924.7 280.7 0 -385.2 -519.6 -541.4 60.83 0 -230.1 -264.9 429.7 0 -305.3 -239.7 472.7 0 -80.29 -46.19 -132.5 42 Eta 95.40 0.4 -314.4 8.150.0 -365.6 90.37 33.84 81.6 9.66 52.6 -206.6 -134.3 (lo 112.5 0 -592.1 -569.6 -833.7 238.5 0 -362.9 -464.8 -447.2 31.76 0 -184.0 -210.5 384.5 0 -259.0 -211.7 455.5 0 -26.50 -16.66 -79.31 159.4 -203.0 -184.0 86.71 51.84 103.59 98.28 66.3 -110.5 -73.94 148.6 32.51 89.6 26.8 63.24 173.6 32.0 59.7 53.14 191.2 174.89 77.40 144.5 192.5 182.1 29.9 97.65 37.99 153.3 191.50 111.3 192.5 113.4 238.5 94.6 151 210.620 240.45 220.0 304.3 264 146 266.4 Substance Oxygen O(g) 0₂(8) 03(8) OH (aq) H₂O(g) H₂O(1) H₂O₂(8) H₂O₂(1) Phosphorus P(g) P₂(8) P4(8) P₁(s, red) P,(s, white) PC13 (8) PC13 (1) PFs (8) PH3(g) P406(S) PO10(S) POCI3 (8) POCI (1) H₂PO₂(aq) Potassium K(g) K(s) K* (aq) K*(8) KCI(s) KCIO3(s) KCIO3(aq) K₂CO3(s) KNO3(s) K₂O(s) KO₂ (s) K₂O₂ (s) KOH(s) KOH(aq) Rubidium Rb(g) Rb(s) RbCl(s) RbCIO3(s) JUS AHUI (kj/mol) 1247.5 0 142.3 -230.0 -285.83 -136.10 -187.8 316.4 144.3 58.9 -17.46 0 -241.82-228.57 -288.07 -319.6 -1594.4 5.4 -1640.1 -2940.1 -542.2 -597.0 -1288.3 89.99 0 -252.4 514.2 -435.9 -391.2 -349.5 -1150.18 -492.70 -363.2 -284.5 -495.8 -424.7 -482.4 APPENDIX C Thermodynamic Quantities for Selected Substances at 298.15 K (25 °C) 85.8 0 -430.5 -392.4 AG; SU (kj/mol) (i/mol-K) 230.1 0 163.4 -157.3 -237.13 -105.48 -120.4 280.0 103.7 24.4 -12.03 0 -269.6 -272.4 -1520.7 13.4 61.17 0 -283.3 481.2 161.0 205.0 237.6 -10.7 H-408.3 -289.9 -284.9 -1064.58 -393.13 -322.1 -240.6 -429.8 -378.9. -440.5 Conical 188.83 69.91 232.9 109.6 55.8 0 -412.0 -292.0 163.2 218.1 280 -2675.2 228.9 11-502.5 325 H-520.9 222 1-1142.6 158.2 22.85 41.08 311.7 217 300.8 210.2 160.2 64.67 102.5 154.5 82.7 143.0 265.7 155.44 132.9 94.14 122.5 113.0 78.91 91.6 CHETICALO 170.0 76.78 92 152 Substance Scandium Sc(g) Sc(s) and Selenium H₂Se(g) Silicon Si(g) Si(s) SIC(s) SIC14(1) SiO₂ (s, quartz) Silver Ag(s) Ag* (aq) AgCl(s) Ag₂O(s) AgNO3(s) Sodium Na(g) Na(s) Na* (aq) Na' (g) NaBr(aq) NaBr(s) Na₂CO3(s) NaCl(aq) NaCl(g) NaCl(s) NaHCO3(s) NaNO3(aq) NaNO3(s) NaOH(aq) NaOH(s) Na₂SO4(s) Strontium Sro(s) Sr(g) Sulfur S(s, rhombic) S8(8) SO₂(8) SO3(8) SO2 (aq) ΔΗ, (kj/mol) 377.8 200 29.7 368.2 0 -73.22 -640.1 1-910.9 0 105.90 -127.0 -31.05 -124.4 107.7 0 -240.1 609.3 -360.6 -361.4 -1130.9 -407.1 -181.4 -410.9 -947.7 -446.2 -467.9 -469.6 -425.6 -1387.1 -592.0 164.4 0 102.3 -296.9 -395.2 -909.3 AG; (kj/mol) 336.1 0 15.9 323.9 0 -70.85 -572.8 -856.5 0 77.11 -109.70 -11.20 -33.41 77.3 0 -261.9 574.3 -364.7 -349.3 -1047.7 -393.0 -201.3 -384.0 -851.8 -372.4 -367.0 -419.2 -379.5 -1270.2 -561.9 110.0 0 49.7 -300.4 -370.4 -744.5 S' (i/mol-K) 174.7 34.6 219.0 167.8 18.7 16.61 239.3 41.84 42.55 73.93 96.11 121.3 140.9 153.7 51.45 59.0 148.0 141.00 86.82 136.0 115.5 229.8 1215 72.33 102.1 207 116.5 49.8 64.46 149.6 54.9 164.6 31.88 430.9 248.5 256.2 20.1 1216 APPENDIX C Thermodynamic Quantities for Selected Substances at 298.15 K (25 °C) Substance SOCI₂(1) H₂S(8) H₂SO4(aq) H₂SO4(1) Titanium Ti(g) Ti(s) TiCl (8) TICI (1) TiO₂ (s) AH; AG (kj/mol) Cha(kj/mol) -245.6 -20.17 -909.3 -814.0 468 0 -763.2 -804.2 -944.7 - -33.01 -744.5 -689.9 422 0 -726.8 -728.1 -889.4 S (i/mol-K)de -bto 205.6 20.1 156.1 180.3 insis nooill2 30.76 354.9 221.9 50.29 ALDIZ Substance Vanadium V(g) 0.fat V(s) 0.205 Zinc Zn(g) Zn(s) ZnCl₂(s) ZnO(s) 10.08 201 AH; (m(kj/mol) 1514.2 0 130.7 0 -415.1 -348.0 X2 BST AG; ((kj/mol) 25453.1 00 0.065 95.2 0 -369.4 -318.2 S' (j/mol-K) 182.2 28.9 160.9 41.63 111.5 43.9 1. Using data in Appendix C, calculate AH, AS', and AG at 298K for each of the following reactions. Indicate whether the reaction is spontaneous at 298K under standard conditions. (a) Ti (s) + 2 Cl₂(g) → TiCl4 (1) (b) HCI (aq) + NaOH (aq) → NaCl (aq) + H₂O (1) (c) CF4 (g) + 2 H₂ (g) → C(s) + 4 HF (g) (d) Na₂CO3 (s) + 2 HNO3(aq) → 2 NaNO3(aq) + CO₂ (g) + H₂O (1) 1. Using data in Appendix C, calculate AH, AS', and AG at 298K for each of the following reactions. Indicate whether the reaction is spontaneous at 298K under standard conditions. (a) Ti (s) + 2 Cl₂(g) → TiCl4 (1) (b) HCI (aq) + NaOH (aq) → NaCl (aq) + H₂O (1) (c) CF4 (g) + 2 H₂ (g) → C(s) + 4 HF (g) (d) Na₂CO3 (s) + 2 HNO3(aq) → 2 NaNO3(aq) + CO₂ (g) + H₂O (1) THERMODYNAMIC QUANTITIES FOR SELECTED SUBSTANCES AT 298.15 K (25 °C) Substance Aluminum Al(s) AICI3(s) Al₂O3(s) Barium Ba(s) BaCO3(s) BaO(s) Beryllium Be(s) BeO(s) Be (OH)2 (s) Bromine Br(g) Br (aq) Br₂(8) Br₂(1) HBr(g) Calcium Ca(g) Ca(s) CaCO3(s, calcite) CaCl₂(s) CaF₂(s) CaO(s) Ca(OH)2(s) CaSO4(s) Carbon C(g) C(s, diamond) C(s, graphite) CC14(8) CC1₂ (1) CF4(8) CH₂(8) AH; (kj/mol) 0 -705.6 -1669.8 0 -1216.3 -553.5 0 -608.4 -905.8 111.8 - 120.9 30.71 -36.23 179.3 0 -1207.1 -795.8 -1219.6 -635.5 -986.2 -1434.0 9718.4 1.88 0 -106.7 -139.3 -679.9 -74.8 AG; (kj/mol) 0 -630.0 -1576.5 0 - 1137.6 -525.1 0 -579.1 -817.9 82.38 - 102.8 3.14 0 -53.22 145.5 0 -1128.76 -748.1 -1167.3 -604.17 -898.5 -1321.8 672.9 2.84 0 -64.0 -68.6 -635.1 -50.8 S (j/mol-K) 28.32 109.3 51.00 63.2 112.1 70.42 9.44 13.77 50.21 174.9 80.71 6H 245.3 152.3 198.49 154.8 41.4 92.88 104.6 68.87 39.75 83.4 106.7 158.0 309.4 214.4 262.3 186.3 2.43 5.69 Will Substance C₂H₂(8) C₂H4(8) C₂H6(8) C3H8(8) C4H10(8) C4H10(1) C6H6(8) C6H6(1) CH₂OH(g) CH₂OH(1) C₂H5OH (8) C₂H₂OH(1) C6H12O6(S) CO(g) CO₂(8) CH3COOH(1) Cesium08 Cs(g) Cs(1) Cs(s) ELLIPS CsCl(s) Chlorine CI(g) HW CI (aq) Cl₂(8) 11.8 HCl(aq) HCI(g) Chromium Cr(g) Cr(s) E Cr₂O3(s) Cobalt Co(g) Co(s) B AH; (kj/mol) 226.77 52.30 -84.68 -103.85 -124.73 -147.6 82.9 49.0 -201.2 -238.6 al-235.1 -277.7 -1273.02 -110.5 06-393.5 -487.0 76.50 2.09 1200 -442.8 121.7 SHOO 1-167.2 -92.30 397.5 22000 -1139.7 APPENDIX C 439 0 AG; (kj/mol) 209.2 68.11 -32.89 -23.47 -15.71 -15.0 129.7 124.5 -161.9 -166.23 08-168.5 1-174.76 25-910.4 -137.2 40-394.4 -392.4 8-167.2-131.2 49.53 08 0.03 eat200 -414.4 105.7 150 2-131.2 -95.27 D 352.6 LABO -1058.1 EXIDE 393 0 S' (j/mol-K) 200.8 219.4 229.5 269.9 310.0 231.0 269.2 172.8 237.6 126.8 282.7 160.7 212.1 197.9 213.6 159.8 175.6 92.07 85.15 101.2 165.2 56.5 222.96 56.5 186.69 174.2 23.6 81.2 179 28.4 121 THERMODYNAMIC QUANTITIES FOR SELECTED SUBSTANCES AT 298.15 K (25 °C) Substance Aluminum Al(s) AICI3(s) Al₂O3(s) Barium Ba(s) BaCO3(s) BaO(s) Beryllium Be(s) BeO(s) Be (OH)2 (s) Bromine Br(g) Br (aq) Br₂(8) Br₂(1) HBr(g) Calcium Ca(g) Ca(s) CaCO3(s, calcite) CaCl₂(s) CaF₂(s) CaO(s) Ca(OH)2(s) CaSO4(s) Carbon C(g) C(s, diamond) C(s, graphite) CC14(8) CC1₂ (1) CF4(8) CH₂(8) AH; (kj/mol) 0 -705.6 -1669.8 0 -1216.3 -553.5 0 -608.4 -905.8 111.8 - 120.9 30.71 -36.23 179.3 0 -1207.1 -795.8 -1219.6 -635.5 -986.2 -1434.0 9718.4 1.88 0 -106.7 -139.3 -679.9 -74.8 AG; (kj/mol) 0 -630.0 -1576.5 0 - 1137.6 -525.1 0 -579.1 -817.9 82.38 - 102.8 3.14 0 -53.22 145.5 0 -1128.76 -748.1 -1167.3 -604.17 -898.5 -1321.8 672.9 2.84 0 -64.0 -68.6 -635.1 -50.8 S (j/mol-K) 28.32 109.3 51.00 63.2 112.1 70.42 9.44 13.77 50.21 174.9 80.71 6H 245.3 152.3 198.49 154.8 41.4 92.88 104.6 68.87 39.75 83.4 106.7 158.0 309.4 214.4 262.3 186.3 2.43 5.69 Will Substance C₂H₂(8) C₂H4(8) C₂H6(8) C3H8(8) C4H10(8) C4H10(1) C6H6(8) C6H6(1) CH₂OH(g) CH₂OH(1) C₂H5OH (8) C₂H₂OH(1) C6H12O6(S) CO(g) CO₂(8) CH3COOH(1) Cesium08 Cs(g) Cs(1) Cs(s) ELLIPS CsCl(s) Chlorine CI(g) HW CI (aq) Cl₂(8) 11.8 HCl(aq) HCI(g) Chromium Cr(g) Cr(s) E Cr₂O3(s) Cobalt Co(g) Co(s) B AH; (kj/mol) 226.77 52.30 -84.68 -103.85 -124.73 -147.6 82.9 49.0 -201.2 -238.6 al-235.1 -277.7 -1273.02 -110.5 06-393.5 -487.0 76.50 2.09 1200 -442.8 121.7 SHOO 1-167.2 -92.30 397.5 22000 -1139.7 APPENDIX C 439 0 AG; (kj/mol) 209.2 68.11 -32.89 -23.47 -15.71 -15.0 129.7 124.5 -161.9 -166.23 08-168.5 1-174.76 25-910.4 -137.2 40-394.4 -392.4 8-167.2-131.2 49.53 08 0.03 eat200 -414.4 105.7 150 2-131.2 -95.27 D 352.6 LABO -1058.1 EXIDE 393 0 S' (j/mol-K) 200.8 219.4 229.5 269.9 310.0 231.0 269.2 172.8 237.6 126.8 282.7 160.7 212.1 197.9 213.6 159.8 175.6 92.07 85.15 101.2 165.2 56.5 222.96 56.5 186.69 174.2 23.6 81.2 179 28.4 121 1214 APPENDIX C Thermodynamic Quantities for Selected Substances at 298.15 K (25 °C) Substance Copper Cu(g) Cu(s) CuCl₂(s) CuO(s) Cu₂O(s) Fluorine F(g) F (aq) F₂(8) HF(g) Hydrogen H(g) H*(aq) H*(8) H₂(8) ES lodine 051 I(g) 58 (8) 00 12(8) LESS 1₂(s) e HI(g) E Iron Fe(g) Fe(s) 21 Fe²+ (aq) Fe³+ (aq) FeCl₂(s) FeCl3(s) FeO(s) zal Fe₂O3(s) Fe3O4(s) FeS₂(s) Lead Pb(s) PbBr₂(S) PbCO3(s) 2 Pb(NO3)2(aq) Pb(NO3)2(S) PbO(s) Lithium Li(g) Li(s) AH; (kj/mol) 338.4 0 -205.9 -156.1 -170.7 80.0 -332.6 0 -268.61 217.94 0 1536.2 0 28106.60 8-55.19 A0162.25 STEL O 25.94 415.5 EERO 20.0-87.86 (-47.69 -341.8 -400 -271.9 -822.16 -1117.1 1-171.5 0 -277.4 -699.1 -421.3 -451.9 -217.3 159.3 0 (fo AG; PROSPERT (kj/mol) (i/mol-K) 298.6 0 -161.7 -128.3 -147.9 61.9 -278.8 0 88-270.70 203.26 0 1517.0 8.1000 Le 70.16 1.-51.57 50. 19.37 20110 ERE 1.30 369.8 02.81 0 POL-84.93 (-10.54 8-302.3 -334 -255.2 -740.98 -1014.2 -160.1 0 2-260.7 -625.5 -246.9 -187.9 126.6 0 166.3 33.30 108.1 42.59 92.36 158.7 -13.8 202.7 173.51 114.60 0 108.9 130.58 1000 HD 180.66 111.3 HO 260.57 H 116.737 206.30 OID 180.5) 27.15 113.4 293.3 117.9 142.3 del 60.75 89.96 146.45 52.92 68.85 161 ( 131.0 303.300 68.70 Hid 138.8 DH 29.09 STUIT WAUTO; DI MAGYOOSHEHT Substance (kj/mol) (kj/mol) (i/mol-K) LI (aq)MATC-278.5-273.4 12.20 Li* (g) LICI(s) Magnesium Mg(g) Mg(s) MgCl₂(s) MgO(s) Mg(OH)₂(s) Manganese Mn(g) Mn(s) MnO(s) MnO₂ (s) MnO4 (aq) Mercury Hg(g) Hg(1) HgCl₂(s) Hg₂Cl₂ (s) Nickel Ni(g) Ni(s) NiCl₂(s) NIO(s) Nitrogen N(g) N₂ (8) NH3(aq) NH3(g) NH, (aq) N₂H₂(8) NH₂CN(S) NH₂Cl(s) NH₂NO3(s) NO(g) NO₂(8) N₂O(g) N₂O4(8) NOCI(g) HNO3(aq) HNO3 (8) 686725) 648.51.833,00 TA -408.3 147.1 0 -641.6 -601.8 -924.7 280.7 0 -385.2 -519.6 -541.4 60.83 0 -230.1 -264.9 429.7 0 -305.3 -239.7 472.7 0 -80.29 -46.19 -132.5 42 Eta 95.40 0.4 -314.4 8.150.0 -365.6 90.37 33.84 81.6 9.66 52.6 -206.6 -134.3 (lo 112.5 0 -592.1 -569.6 -833.7 238.5 0 -362.9 -464.8 -447.2 31.76 0 -184.0 -210.5 384.5 0 -259.0 -211.7 455.5 0 -26.50 -16.66 -79.31 159.4 -203.0 -184.0 86.71 51.84 103.59 98.28 66.3 -110.5 -73.94 148.6 32.51 89.6 26.8 63.24 173.6 32.0 59.7 53.14 191.2 174.89 77.40 144.5 192.5 182.1 29.9 97.65 37.99 153.3 191.50 111.3 192.5 113.4 238.5 94.6 151 210.620 240.45 220.0 304.3 264 146 266.4 1214 APPENDIX C Thermodynamic Quantities for Selected Substances at 298.15 K (25 °C) Substance Copper Cu(g) Cu(s) CuCl₂(s) CuO(s) Cu₂O(s) Fluorine F(g) F (aq) F₂(8) HF(g) Hydrogen H(g) H*(aq) H*(8) H₂(8) ES lodine 051 I(g) 58 (8) 00 12(8) LESS 1₂(s) e HI(g) E Iron Fe(g) Fe(s) 21 Fe²+ (aq) Fe³+ (aq) FeCl₂(s) FeCl3(s) FeO(s) zal Fe₂O3(s) Fe3O4(s) FeS₂(s) Lead Pb(s) PbBr₂(S) PbCO3(s) 2 Pb(NO3)2(aq) Pb(NO3)2(S) PbO(s) Lithium Li(g) Li(s) AH; (kj/mol) 338.4 0 -205.9 -156.1 -170.7 80.0 -332.6 0 -268.61 217.94 0 1536.2 0 28106.60 8-55.19 A0162.25 STEL O 25.94 415.5 EERO 20.0-87.86 (-47.69 -341.8 -400 -271.9 -822.16 -1117.1 1-171.5 0 -277.4 -699.1 -421.3 -451.9 -217.3 159.3 0 (fo AG; PROSPERT (kj/mol) (i/mol-K) 298.6 0 -161.7 -128.3 -147.9 61.9 -278.8 0 88-270.70 203.26 0 1517.0 8.1000 Le 70.16 1.-51.57 50. 19.37 20110 ERE 1.30 369.8 02.81 0 POL-84.93 (-10.54 8-302.3 -334 -255.2 -740.98 -1014.2 -160.1 0 2-260.7 -625.5 -246.9 -187.9 126.6 0 166.3 33.30 108.1 42.59 92.36 158.7 -13.8 202.7 173.51 114.60 0 108.9 130.58 1000 HD 180.66 111.3 HO 260.57 H 116.737 206.30 OID 180.5) 27.15 113.4 293.3 117.9 142.3 del 60.75 89.96 146.45 52.92 68.85 161 ( 131.0 303.300 68.70 Hid 138.8 DH 29.09 STUIT WAUTO; DI MAGYOOSHEHT Substance (kj/mol) (kj/mol) (i/mol-K) LI (aq)MATC-278.5-273.4 12.20 Li* (g) LICI(s) Magnesium Mg(g) Mg(s) MgCl₂(s) MgO(s) Mg(OH)₂(s) Manganese Mn(g) Mn(s) MnO(s) MnO₂ (s) MnO4 (aq) Mercury Hg(g) Hg(1) HgCl₂(s) Hg₂Cl₂ (s) Nickel Ni(g) Ni(s) NiCl₂(s) NIO(s) Nitrogen N(g) N₂ (8) NH3(aq) NH3(g) NH, (aq) N₂H₂(8) NH₂CN(S) NH₂Cl(s) NH₂NO3(s) NO(g) NO₂(8) N₂O(g) N₂O4(8) NOCI(g) HNO3(aq) HNO3 (8) 686725) 648.51.833,00 TA -408.3 147.1 0 -641.6 -601.8 -924.7 280.7 0 -385.2 -519.6 -541.4 60.83 0 -230.1 -264.9 429.7 0 -305.3 -239.7 472.7 0 -80.29 -46.19 -132.5 42 Eta 95.40 0.4 -314.4 8.150.0 -365.6 90.37 33.84 81.6 9.66 52.6 -206.6 -134.3 (lo 112.5 0 -592.1 -569.6 -833.7 238.5 0 -362.9 -464.8 -447.2 31.76 0 -184.0 -210.5 384.5 0 -259.0 -211.7 455.5 0 -26.50 -16.66 -79.31 159.4 -203.0 -184.0 86.71 51.84 103.59 98.28 66.3 -110.5 -73.94 148.6 32.51 89.6 26.8 63.24 173.6 32.0 59.7 53.14 191.2 174.89 77.40 144.5 192.5 182.1 29.9 97.65 37.99 153.3 191.50 111.3 192.5 113.4 238.5 94.6 151 210.620 240.45 220.0 304.3 264 146 266.4 Substance Oxygen O(g) 0₂(8) 03(8) OH (aq) H₂O(g) H₂O(1) H₂O₂(8) H₂O₂(1) Phosphorus P(g) P₂(8) P4(8) P₁(s, red) P,(s, white) PC13 (8) PC13 (1) PFs (8) PH3(g) P406(S) PO10(S) POCI3 (8) POCI (1) H₂PO₂(aq) Potassium K(g) K(s) K* (aq) K*(8) KCI(s) KCIO3(s) KCIO3(aq) K₂CO3(s) KNO3(s) K₂O(s) KO₂ (s) K₂O₂ (s) KOH(s) KOH(aq) Rubidium Rb(g) Rb(s) RbCl(s) RbCIO3(s) JUS AHUI (kj/mol) 1247.5 0 142.3 -230.0 -285.83 -136.10 -187.8 316.4 144.3 58.9 -17.46 0 -241.82-228.57 -288.07 -319.6 -1594.4 5.4 -1640.1 -2940.1 -542.2 -597.0 -1288.3 89.99 0 -252.4 514.2 -435.9 -391.2 -349.5 -1150.18 -492.70 -363.2 -284.5 -495.8 -424.7 -482.4 APPENDIX C Thermodynamic Quantities for Selected Substances at 298.15 K (25 °C) 85.8 0 -430.5 -392.4 AG; SU (kj/mol) (i/mol-K) 230.1 0 163.4 -157.3 -237.13 -105.48 -120.4 280.0 103.7 24.4 -12.03 0 -269.6 -272.4 -1520.7 13.4 61.17 0 -283.3 481.2 161.0 205.0 237.6 -10.7 H-408.3 -289.9 -284.9 -1064.58 -393.13 -322.1 -240.6 -429.8 -378.9. -440.5 Conical 188.83 69.91 232.9 109.6 55.8 0 -412.0 -292.0 163.2 218.1 280 -2675.2 228.9 11-502.5 325 H-520.9 222 1-1142.6 158.2 22.85 41.08 311.7 217 300.8 210.2 160.2 64.67 102.5 154.5 82.7 143.0 265.7 155.44 132.9 94.14 122.5 113.0 78.91 91.6 CHETICALO 170.0 76.78 92 152 Substance Scandium Sc(g) Sc(s) and Selenium H₂Se(g) Silicon Si(g) Si(s) SIC(s) SIC14(1) SiO₂ (s, quartz) Silver Ag(s) Ag* (aq) AgCl(s) Ag₂O(s) AgNO3(s) Sodium Na(g) Na(s) Na* (aq) Na' (g) NaBr(aq) NaBr(s) Na₂CO3(s) NaCl(aq) NaCl(g) NaCl(s) NaHCO3(s) NaNO3(aq) NaNO3(s) NaOH(aq) NaOH(s) Na₂SO4(s) Strontium Sro(s) Sr(g) Sulfur S(s, rhombic) S8(8) SO₂(8) SO3(8) SO2 (aq) ΔΗ, (kj/mol) 377.8 200 29.7 368.2 0 -73.22 -640.1 1-910.9 0 105.90 -127.0 -31.05 -124.4 107.7 0 -240.1 609.3 -360.6 -361.4 -1130.9 -407.1 -181.4 -410.9 -947.7 -446.2 -467.9 -469.6 -425.6 -1387.1 -592.0 164.4 0 102.3 -296.9 -395.2 -909.3 AG; (kj/mol) 336.1 0 15.9 323.9 0 -70.85 -572.8 -856.5 0 77.11 -109.70 -11.20 -33.41 77.3 0 -261.9 574.3 -364.7 -349.3 -1047.7 -393.0 -201.3 -384.0 -851.8 -372.4 -367.0 -419.2 -379.5 -1270.2 -561.9 110.0 0 49.7 -300.4 -370.4 -744.5 S' (i/mol-K) 174.7 34.6 219.0 167.8 18.7 16.61 239.3 41.84 42.55 73.93 96.11 121.3 140.9 153.7 51.45 59.0 148.0 141.00 86.82 136.0 115.5 229.8 1215 72.33 102.1 207 116.5 49.8 64.46 149.6 54.9 164.6 31.88 430.9 248.5 256.2 20.1 Substance Oxygen O(g) 0₂(8) 03(8) OH (aq) H₂O(g) H₂O(1) H₂O₂(8) H₂O₂(1) Phosphorus P(g) P₂(8) P4(8) P₁(s, red) P,(s, white) PC13 (8) PC13 (1) PFs (8) PH3(g) P406(S) PO10(S) POCI3 (8) POCI (1) H₂PO₂(aq) Potassium K(g) K(s) K* (aq) K*(8) KCI(s) KCIO3(s) KCIO3(aq) K₂CO3(s) KNO3(s) K₂O(s) KO₂ (s) K₂O₂ (s) KOH(s) KOH(aq) Rubidium Rb(g) Rb(s) RbCl(s) RbCIO3(s) JUS AHUI (kj/mol) 1247.5 0 142.3 -230.0 -285.83 -136.10 -187.8 316.4 144.3 58.9 -17.46 0 -241.82-228.57 -288.07 -319.6 -1594.4 5.4 -1640.1 -2940.1 -542.2 -597.0 -1288.3 89.99 0 -252.4 514.2 -435.9 -391.2 -349.5 -1150.18 -492.70 -363.2 -284.5 -495.8 -424.7 -482.4 APPENDIX C Thermodynamic Quantities for Selected Substances at 298.15 K (25 °C) 85.8 0 -430.5 -392.4 AG; SU (kj/mol) (i/mol-K) 230.1 0 163.4 -157.3 -237.13 -105.48 -120.4 280.0 103.7 24.4 -12.03 0 -269.6 -272.4 -1520.7 13.4 61.17 0 -283.3 481.2 161.0 205.0 237.6 -10.7 H-408.3 -289.9 -284.9 -1064.58 -393.13 -322.1 -240.6 -429.8 -378.9. -440.5 Conical 188.83 69.91 232.9 109.6 55.8 0 -412.0 -292.0 163.2 218.1 280 -2675.2 228.9 11-502.5 325 H-520.9 222 1-1142.6 158.2 22.85 41.08 311.7 217 300.8 210.2 160.2 64.67 102.5 154.5 82.7 143.0 265.7 155.44 132.9 94.14 122.5 113.0 78.91 91.6 CHETICALO 170.0 76.78 92 152 Substance Scandium Sc(g) Sc(s) and Selenium H₂Se(g) Silicon Si(g) Si(s) SIC(s) SIC14(1) SiO₂ (s, quartz) Silver Ag(s) Ag* (aq) AgCl(s) Ag₂O(s) AgNO3(s) Sodium Na(g) Na(s) Na* (aq) Na' (g) NaBr(aq) NaBr(s) Na₂CO3(s) NaCl(aq) NaCl(g) NaCl(s) NaHCO3(s) NaNO3(aq) NaNO3(s) NaOH(aq) NaOH(s) Na₂SO4(s) Strontium Sro(s) Sr(g) Sulfur S(s, rhombic) S8(8) SO₂(8) SO3(8) SO2 (aq) ΔΗ, (kj/mol) 377.8 200 29.7 368.2 0 -73.22 -640.1 1-910.9 0 105.90 -127.0 -31.05 -124.4 107.7 0 -240.1 609.3 -360.6 -361.4 -1130.9 -407.1 -181.4 -410.9 -947.7 -446.2 -467.9 -469.6 -425.6 -1387.1 -592.0 164.4 0 102.3 -296.9 -395.2 -909.3 AG; (kj/mol) 336.1 0 15.9 323.9 0 -70.85 -572.8 -856.5 0 77.11 -109.70 -11.20 -33.41 77.3 0 -261.9 574.3 -364.7 -349.3 -1047.7 -393.0 -201.3 -384.0 -851.8 -372.4 -367.0 -419.2 -379.5 -1270.2 -561.9 110.0 0 49.7 -300.4 -370.4 -744.5 S' (i/mol-K) 174.7 34.6 219.0 167.8 18.7 16.61 239.3 41.84 42.55 73.93 96.11 121.3 140.9 153.7 51.45 59.0 148.0 141.00 86.82 136.0 115.5 229.8 1215 72.33 102.1 207 116.5 49.8 64.46 149.6 54.9 164.6 31.88 430.9 248.5 256.2 20.1 1216 APPENDIX C Thermodynamic Quantities for Selected Substances at 298.15 K (25 °C) Substance SOCI₂(1) H₂S(8) H₂SO4(aq) H₂SO4(1) Titanium Ti(g) Ti(s) TiCl (8) TICI (1) TiO₂ (s) AH; AG (kj/mol) Cha(kj/mol) -245.6 -20.17 -909.3 -814.0 468 0 -763.2 -804.2 -944.7 - -33.01 -744.5 -689.9 422 0 -726.8 -728.1 -889.4 S (i/mol-K)de -bto 205.6 20.1 156.1 180.3 insis nooill2 30.76 354.9 221.9 50.29 ALDIZ Substance Vanadium V(g) 0.fat V(s) 0.205 Zinc Zn(g) Zn(s) ZnCl₂(s) ZnO(s) 10.08 201 AH; (m(kj/mol) 1514.2 0 130.7 0 -415.1 -348.0 X2 BST AG; ((kj/mol) 25453.1 00 0.065 95.2 0 -369.4 -318.2 S' (j/mol-K) 182.2 28.9 160.9 41.63 111.5 43.9 1216 APPENDIX C Thermodynamic Quantities for Selected Substances at 298.15 K (25 °C) Substance SOCI₂(1) H₂S(8) H₂SO4(aq) H₂SO4(1) Titanium Ti(g) Ti(s) TiCl (8) TICI (1) TiO₂ (s) AH; AG (kj/mol) Cha(kj/mol) -245.6 -20.17 -909.3 -814.0 468 0 -763.2 -804.2 -944.7 - -33.01 -744.5 -689.9 422 0 -726.8 -728.1 -889.4 S (i/mol-K)de -bto 205.6 20.1 156.1 180.3 insis nooill2 30.76 354.9 221.9 50.29 ALDIZ Substance Vanadium V(g) 0.fat V(s) 0.205 Zinc Zn(g) Zn(s) ZnCl₂(s) ZnO(s) 10.08 201 AH; (m(kj/mol) 1514.2 0 130.7 0 -415.1 -348.0 X2 BST AG; ((kj/mol) 25453.1 00 0.065 95.2 0 -369.4 -318.2 S' (j/mol-K) 182.2 28.9 160.9 41.63 111.5 43.9
Expert Answer:
Answer rating: 100% (QA)
Sure we can use the data provided in Appendix C to calculate the standard enthalpy change H the standard entropy change S and the standard Gibbs free ... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemical engineering questions
-
Using data from Appendix C, calculate the change in Gibbs free energy for each of the following reactions. In each case indicate whether the reaction is spontaneous at 298 K under standard...
-
For each of the following reactions, indicate whether the reaction would work well, poorly, or not at all. Formulate alternative products, if appropriate. (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) (k)...
-
import java.util.ArrayList; import java.util.Map; import java.util.Set; import java.util.TreeMap; /* * * This class contains a map of string keys, representing professors, to an array list of...
-
Thalassines Kataskeves, S.A., of Greece makes marine equipment. The company has been experiencing losses on its bilge pump product line for several years. The most recent quarterly contribution...
-
Solve Problem 35 by the big M method. Data from Problem 35 Solve by the dual problem method: Minimize subject to C = 3x + 2x 2x + x = 20 2x1 + x = 9 X + X = 6 X1, X2 = 0
-
Explain with a neat sketch fast and loose pulley.
-
In the face of stable (or declining) enrollments and increasing costs, many colleges and universities, both public and private, find themselves in progressively tighter financial dilemmas that...
-
A 7400 kg rocket blasts off vertically from the launch pad with a constant upward acceleration of 2.20 m/s2 and feels no appreciable air resistance. When it has reached a height of 600 m , its...
-
The following salaried employees of Mountain Stone Brewery in Fort Collins, Colorado, are paid semimonthly. Some employees have union dues or garnishments deducted from their pay. Required: Calculate...
-
5. Hens and Turtles Inc. issues a $1 million, 7% mortgage note payable on January 1, 2020. The note will be paid in annual blended payments of principal and interest of $140,000 at the end of each...
-
Discuss the scenario below, as to whether a riskless opportunity exists in the FX market and how to capitalise the opportunity. Suppose that you can borrow 21 million today. Assume that the current...
-
Factor. x +15x+36
-
Divide. (x3-4x2+5x-2)+(x-1) (x3-4x+5x-2)=(x-1)= | + X-1 (Simplify your answers. Do not factor.)
-
Evaluate the following. 3x4-4-204
-
(c) Consider the interconnected system presented in Figure Q2.3 and the information provided in Table Q2.1 and Table Q2.2. Area 1 Area 2 Generator G G Load L L G1 G ( G L L2 D Base 1000 (MVA) 750...
-
True OR False When the auditors compare a sample of shipping documents to related sales invoices, they are trying to locate Items that have been billed but not shipped. All allowances to customers...
-
In the busy port of Chennai, India, the number of containers loaded onto ships during a 15-week period is as follows: 1. Develop a linear trend equation to forecast container loadings. 2. Using the...
-
The following processes were all discussed in Chapter 18, "Chemistry of the Environment." Estimate whether the entropy of the system increases or decreases during each process: (a) Photodissociation...
-
We saw in Chapter 6 that the probability of finding an electron in three-dimensional space depends on what orbital it is in. Look back at Figures 6.19 and 6.22, which show the radial probability...
-
(a) Precipitation of the group 4 cations of Figure 17.23 requires a basic medium. Why is this so? (b) What is the most significant difference between the sulfides precipitated in group 2 and those...
-
Compare and contrast licensure and credentialing.
-
Compare and contrast the different inventory management techniques and discuss how each technique might play a role within different health care organizations.
-
Distinguish between core privileges and specific privileges in physician credentialing.

Study smarter with the SolutionInn App


