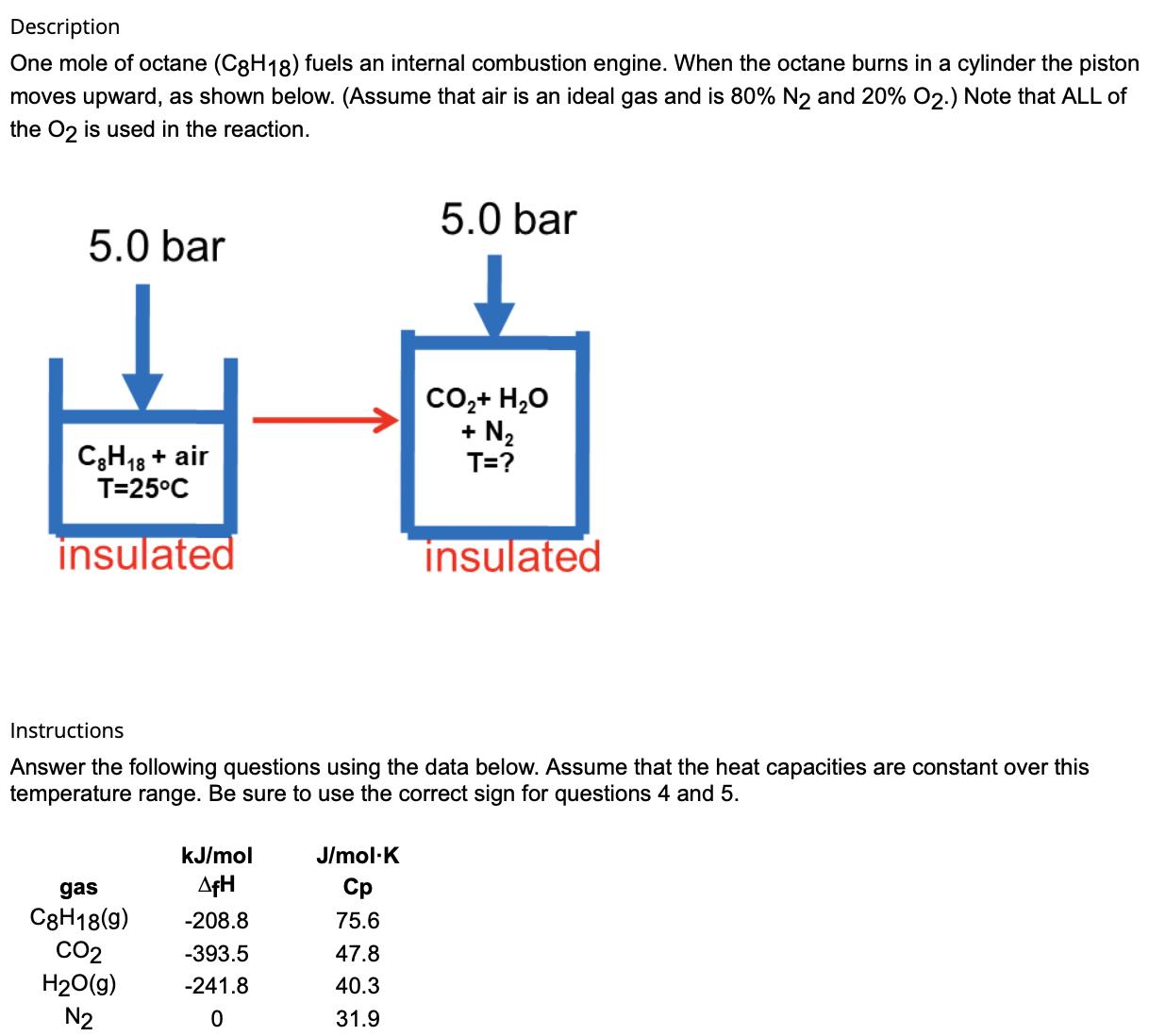
1. What is the change in enthalpy (H) for the contents of the cylinder? (kJ) 2. How...
Fantastic news! We've Found the answer you've been seeking!
Question:
1. What is the change in enthalpy (∆H) for the contents of the cylinder? (kJ)
2. How much work is performed by the gas? (kJ) (Hint: The answer is a negative number.)
3. What is the change in internal energy (∆U) for the gas in the cylinder? (kJ)
Related Book For 

Posted Date:




