1. Write a balanced equation for the reaction of the active ingredient in Tums with excess...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
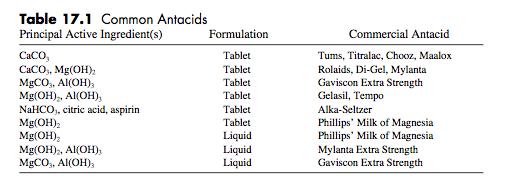
1. Write a balanced equation for the reaction of the active ingredient in Tums with excess acid. See Table 17.1. 2. Identify the two most common anions present in antacids. 3. a. How much time should be allowed for the titrant io drain from the buret wall before a reading is made? 16 b. Experimental Procedure, Part B.2. Bromophenol blue is the indicator used in delecting the endpoint for the antacid analysis in this experiment. What is the expected color change at the endpoint? 4. a. How many moles of stomach acid would be neutralized by one tablet of Tums Ultra 1000 that contains 1000 mg of calcium carbonate? CacO, (ag) +2 H,0(ag) - Ca"(ag) + CO(g) + 3 H,O) b. Assuming the volume of the stomach to be 1.0 L what will be the pH change of the stomach acid resulting from the ingestion of one Tums ultra 1000 tablet that contains 1000 mg of cal- cium carbonate. TUMS Table 17.1 Common Antacids Principal Active Ingredient(s) Formulation Commercial Antacid CaCO, CaCO, Mg(OH); MgCO,, Al(OH), Mg(OH),, Al(OH), NaHCO, citric acid, aspirin Mg(OH): Mg(OH), Mg(OH)2, Al(OH), MGCO, Al(OH), Tablet Tums, Titralac, Chooz, Maalox Rolaids, Di-Gel, Mylanta Gaviscon Extra Strength Gelasil, Tempo Tablet Tablet Tablet Alka-Seltzer Tablet Tablet Phillips' Milk of Magnesia Phillips' Milk of Magnesia Mylanta Extra Strength Gaviscon Extra Strength Liquid Liquid Liquid 1. Write a balanced equation for the reaction of the active ingredient in Tums with excess acid. See Table 17.1. 2. Identify the two most common anions present in antacids. 3. a. How much time should be allowed for the titrant io drain from the buret wall before a reading is made? 16 b. Experimental Procedure, Part B.2. Bromophenol blue is the indicator used in delecting the endpoint for the antacid analysis in this experiment. What is the expected color change at the endpoint? 4. a. How many moles of stomach acid would be neutralized by one tablet of Tums Ultra 1000 that contains 1000 mg of calcium carbonate? CacO, (ag) +2 H,0(ag) - Ca"(ag) + CO(g) + 3 H,O) b. Assuming the volume of the stomach to be 1.0 L what will be the pH change of the stomach acid resulting from the ingestion of one Tums ultra 1000 tablet that contains 1000 mg of cal- cium carbonate. TUMS Table 17.1 Common Antacids Principal Active Ingredient(s) Formulation Commercial Antacid CaCO, CaCO, Mg(OH); MgCO,, Al(OH), Mg(OH),, Al(OH), NaHCO, citric acid, aspirin Mg(OH): Mg(OH), Mg(OH)2, Al(OH), MGCO, Al(OH), Tablet Tums, Titralac, Chooz, Maalox Rolaids, Di-Gel, Mylanta Gaviscon Extra Strength Gelasil, Tempo Tablet Tablet Tablet Alka-Seltzer Tablet Tablet Phillips' Milk of Magnesia Phillips' Milk of Magnesia Mylanta Extra Strength Gaviscon Extra Strength Liquid Liquid Liquid
Expert Answer:

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemical engineering questions
-
Write a balanced equation for the reaction of each of the following compounds with water: (a) SO2(g) (b) Cl2O7(g) (c) Na2O2(s) (d) BaC2(s) (e) RbO2(s) (f) Mg3N2(s) (g) NaH(s).
-
Write a balanced equation for the reaction of -(1)-galactose (use either an acyclic or a cyclic structure, whichever seems more appropriate) with each of the following: a. Hydroxylamine (to form an...
-
Write down a balanced equation for the reaction of bromine with cyclohexene. Is the product of the reaction saturated or unsaturated? Explain your answer by describing what type of reaction is...
-
Consider the graph of a function g(x). g(x) 5 f 3 2 1 2 3 4 5 6 Find the point c at which the function has a jump discontinuity but is right-continuous. C = What value should be assigned to g(c) to...
-
For what compressor efficiency will the gas-turbine power plant in Problem 9-91E produce zero net work? Problem 9 - 91E A gas-turbine power plant operates on a simple Brayton cycle with air as the...
-
Explain how a company assesses materiality when attempting to report a true and fair view of its income.
-
0.1 Use the Standard Normal Table or technology to find the z-score that corresponds to the cumulative area or percentile. Table 4-Standard Normal Distribution Arca 0 z Z .09 .08 .07 .06 .05 .04 .03...
-
Why do you believe that SOX does not provide legal remedies for corporate whistleblowers who are punished by their employers after communicating with members of the news media?
-
Let the universal set be the set of all students at the university. Furthermore, let F denote the set of female students, M the set of all students enrolled in ECOCAL 1, C the set of students who are...
-
On November 1, 2005, Janet Morton and Kim Wong formed Pet Kingdom, Inc., to sell pets and pet supplies. Pertinent information regarding Pet Kingdom is summarized as follows: Pet Kingdom's business...
-
7 li a nonmagnetic material. H = 30 cos (2T x 10t- 6x) ay mA/m find: (a) the intrinsic impedance. (b) the Poynting vector. (c) the time-average power crossing the surface x = 1.0
-
In what circumstances can a person who is not the owner of goods pass a good title to them?
-
Give an example of strict interpretation of an exemption clause.
-
Explain the attitude of the courts toward exemption clauses.
-
When a buyer refuses to accept delivery of goods, explain the significance of supply and demand when determining the value of damages suffered.
-
What is the reasoning behind the requirement that an injured party mitigate its losses?
-
TipTop Flight School offers flying lessons at a small municipal airport. The school's owner and manager has been attempting to evaluate performance and control costs using a variance report that...
-
The water in tank A is at 270 F with quality of 10% and mass 1 lbm. It is connected to a piston/cylinder holding constant pressure of 40 psia initially with 1 lbm water at 700 F. The valve is opened,...
-
(a) How many coulombs are required to plate a layer of chromium metal 0.25 mm thick on an auto bumper with a total area of 0.32 m2 from a solution containing CrO42-? The density of chromium metal is...
-
Organic compounds containing C-O and C-1 bonds are more reactive than simple alkane hydrocarbons. Considering the comparative values of C - H, C - C, C--O and C -- 1 bond enthalpies (Table 8.4), why...
-
Identify each of the following elements as a metal, nonmetal, or metalloid: (a) Gallium, (b) Molybdenum, (c) Tellurium, (d) Arsenic, (e) Xenon, (f) Ruthenium.
-
What are the four main consumer rights?
-
Identify the four levels of a company's ethical environment.
-
What is meant by social responsibility, and why do companies exercise it?

Study smarter with the SolutionInn App


