100 percent transmittance 80 60 40 20 0 2.6 2.8 3 wavelength, micrometers 3.5 4 4.5...
Fantastic news! We've Found the answer you've been seeking!
Question:
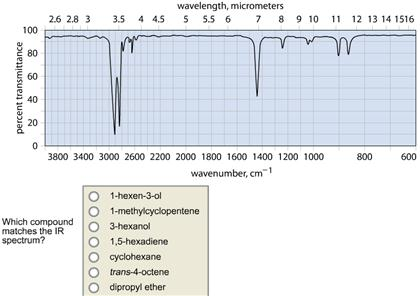
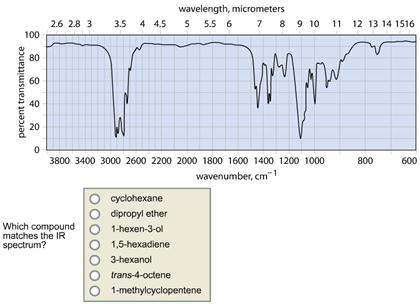
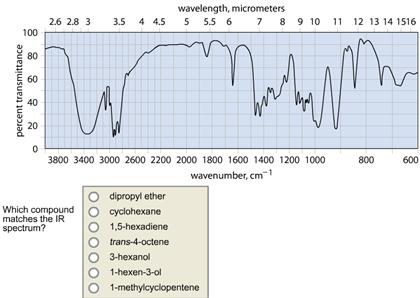
Transcribed Image Text:
100 percent transmittance 80 60 40 20 0 2.6 2.8 3 wavelength, micrometers 3.5 4 4.5 5 5.5 6 7 8 9 10 11 12 13 14 1516 3800 3400 3000 2600 2200 2000 1800 1600 1400 1200 1000 wavenumber, cm¹ Which compound matches the IR spectrum? 1-hexen-3-ol 1-methylcyclopentene 3-hexanol 1,5-hexadiene cyclohexane trans-4-octene dipropyl ether 800 600 percent transmittance 100 80 60 40 20 0 wavelength, micrometers 2.6 2.8 3 3.5 4 4.5 5 5.5 6 7 8 9 10 3800 3400 3000 2600 2200 2000 1800 1600 1400 1200 1000 wavenumber, cm1 Which compound matches the IR spectrum? cyclohexane dipropyl ether 1-hexen-3-ol 1,5-hexadiene 3-hexanol trans-4-octene. 1-methylcyclopentene 11 12 13 14 1516 Wh 800 600 100 percent transmittance 80 60 40 20 0 2.6 2.8 3 3.5 4 4.5 Which compound matches the IR spectrum? wavelength, micrometers 5 5.5 6 7 8 9 10 dipropyl ether cyclohexane 1,5-hexadiene trans-4-octene 3-hexanol 1-hexen-3-ol M 3800 3400 3000 2600 2200 2000 1800 1600 1400 1200 1000 wavenumber, cm1 1-methylcyclopentene vr Mal 11 12 13 14 1516 h 800 600 100 percent transmittance 80 60 40 20 0 2.6 2.8 3 wavelength, micrometers 3.5 4 4.5 5 5.5 6 7 8 9 10 11 12 13 14 1516 3800 3400 3000 2600 2200 2000 1800 1600 1400 1200 1000 wavenumber, cm¹ Which compound matches the IR spectrum? 1-hexen-3-ol 1-methylcyclopentene 3-hexanol 1,5-hexadiene cyclohexane trans-4-octene dipropyl ether 800 600 percent transmittance 100 80 60 40 20 0 wavelength, micrometers 2.6 2.8 3 3.5 4 4.5 5 5.5 6 7 8 9 10 3800 3400 3000 2600 2200 2000 1800 1600 1400 1200 1000 wavenumber, cm1 Which compound matches the IR spectrum? cyclohexane dipropyl ether 1-hexen-3-ol 1,5-hexadiene 3-hexanol trans-4-octene. 1-methylcyclopentene 11 12 13 14 1516 Wh 800 600 100 percent transmittance 80 60 40 20 0 2.6 2.8 3 3.5 4 4.5 Which compound matches the IR spectrum? wavelength, micrometers 5 5.5 6 7 8 9 10 dipropyl ether cyclohexane 1,5-hexadiene trans-4-octene 3-hexanol 1-hexen-3-ol M 3800 3400 3000 2600 2200 2000 1800 1600 1400 1200 1000 wavenumber, cm1 1-methylcyclopentene vr Mal 11 12 13 14 1516 h 800 600
Expert Answer:
Answer rating: 100% (QA)
The detailed answer for the above question is provided below In graph 1 we ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The cost principle states that if no cash is involved in a transaction the cash-equivalent value must be used. True False
-
3 4 5 6 7 8 9 10 11 12 13 A 14 Cost of the Asset 15 Life of the Asset in Years 16 Book Value of the Asset after 5 years 17 Depreciable Basis 18 Yearly depreciation 19 After tax Salvage Value in year...
-
3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 Mo's Motor Corp. issued $ 380,000 of 7%, 5-year bonds on April 1, 2021. Interest is payable semi-annually on April 1 and October 1. On April 1, 2023,...
-
Happy Colors manufactures crayons in a three-step process: mixing, molding, and packaging. The Mixing Department combines the direct materials of paraffin wax and pigments. The heated mixture is...
-
EZ, Inc., manufactures two products that require both machine processing and labor operations. Although there is unlimited demand for both products, EZ could devote all its capacities to a single...
-
What information should be provided in the caption to a pleading?
-
Explain why the FBI would need forensic accountants. What other government agencies might need forensic accounting skills?
-
The following partial work sheet is taken from Kevin's Gift Shop for the year ended December 31, 20--. The ending merchandise inventory is $50,000. 1. Complete the Adjustments columns for the...
-
Current Attempt in Progress Sheridan Company produces corn chips. The cost of one batch is below: Direct materials $16 Direct labor 11 Variable 9 overhead Fixed overhead 14 An outside supplier has...
-
The balances in the general ledger accounts of Thunder Graphics are shown below. Instructions On the forms provided in your working papers: 1 . Open the accounts in the general ledger with their...
-
H.R. 3731: Immigrants in Nursing and Allied Health Act of 2023 Be sure to address the following: Determine the legislative intent of the bill you have reviewed. Identify the proponents/opponents of...
-
The management of Wengel Corporation is considering dropping product B90D. Data from the company's accounting system appear below: Sales Variable expenses Fixed manufacturing expenses Fixed selling...
-
Steinberg Company had the following direct materials costs for the manufacturing of product T in March: Actual purchase price per pound of direct materials $ 9.00 Standard direct materials allowed...
-
Financial data for Beaker Company for last year appear below: Beaker Company Statements of Financial Position Beginning Balance Ending Balance Assets: Cash Accounts receivable Inventory Plant and...
-
A chemical plant produces sodium bisulfate in 100 kg bags. Demand for this product is 28 tonnes per day. The capacity for producing this product is 60 tonnes per day. Setup cost is $420, and holding...
-
Capes Corporation is a wholesaler of industrial goods. Data regarding the store's operations follow: Sales are budgeted at $300,000 for November, $310,000 for December, and $290,000 for January....
-
An investment project provides cash inflows of $1046 per year for nine years. If the initial cost is $4,200, what is the project payback period? What if the initial cost is $5,400? What if it is...
-
The May 2014 revenue and cost information for Houston Outfitters, Inc. follow: Sales Revenue (at standard).............. $ 540,000 Cost of Goods Sold (at standard) ..........341,000 Direct Materials...
-
When p-aminophenol reacts with one molar equivalent of acetic anhydride, a compound acetaminophen (A, C8H9NO2) is formed that dissolves in dilute NaOH. When A is treated with one equivalent of NaOH...
-
Assuming you knew the properties of the compound obtained in Problem 24.23, including its optical rotation, show how you could use periodate oxidation to distinguish methyl -D- galactopyranoside from...
-
Compound A, C6H12O2, was found to be optically active, and it was slowly oxidized to an optically active carboxylic acid B, C6H12O3, by +Ag(NH3)2. Oxidation of A by anhydrous CrO3 gave an optically...
-
12. ROLE REVERSAL: Write a multiple-choice question that raises the issue of an instrument's negotiability.
-
Question: Winona owns a tropical fish store. To buy a spectacular new tank, she borrows $25,000 from her sister, Pauline, and signs an agreement giving Pauline a security interest in the tank....
-
Consider the set of vectors \((-1,1,1),(1,-1,1),(1,1,-1)\). a. Use the Gram-Schmidt process to find an orthonormal basis for \(R^{3}\) using this set in the given order. b. What do you get if you do...

Study smarter with the SolutionInn App


