A sample Mg metal reacts with excess aqueous hydrochloric acid to produce 257.3 mL of dry...
Fantastic news! We've Found the answer you've been seeking!
Question:
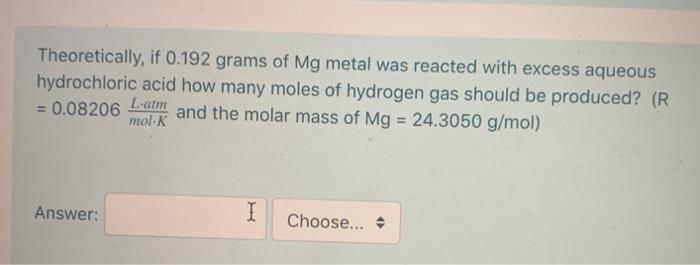
Transcribed Image Text:
A sample Mg metal reacts with excess aqueous hydrochloric acid to produce 257.3 mL of dry hydrogen at a pressure of 1.00 atm and a temperature of 27.1 °C. How many moles of hydrogen gas are produced? (R = 0.08206 Lam :) mol-K Answer: Theoretically, if 0.192 grams of Mg metal was reacted with excess aqueous hydrochloric acid how many moles of hydrogen gas should be produced? (R = 0.08206 and the molar mass of Mg = 24.3050 g/mol) L-atm mol-K Answer: I Choose... A sample Mg metal reacts with excess aqueous hydrochloric acid to produce 257.3 mL of dry hydrogen at a pressure of 1.00 atm and a temperature of 27.1 °C. How many moles of hydrogen gas are produced? (R = 0.08206 Lam :) mol-K Answer: Theoretically, if 0.192 grams of Mg metal was reacted with excess aqueous hydrochloric acid how many moles of hydrogen gas should be produced? (R = 0.08206 and the molar mass of Mg = 24.3050 g/mol) L-atm mol-K Answer: I Choose...
Expert Answer:
Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these accounting questions
-
A series of dialkyl ethers was allowed to react with excess hydrogen bromide, with the following results. Identify the ether in each case. (a) Another ether gave only benzyl bromide. (b) A third...
-
(a) If excess C is allowed to react with 25.0 g of CO2 in a 3.00-L vessel at 1000 K, how many grams of CO are produced? (b) How many grams of C are consumed? (c) If a smaller vessel is used for the...
-
Hydrogen peroxide in aqueous solution will decompose to produce oxygen gas and water. Use the graph to the right to: Reaction Progress 0.35 0.3 a) calculate the average reaction rate for the...
-
The following linear systems Ax = b have x as the actual solution and x as an approximate solution. Compute ||x x|| and ||Ax b|| a. 1/2 x1 + 1/3 x2 = 1/63, 1/3 x1 + 1/4 x2 = 1/168, x = ( 1/7 ,1/6)t...
-
How do the terms accountability, authority, and responsibility differ in meaning?
-
A ball moves in a straight line (the x-axis). The graph in Fig. 2.33 shows this ball's velocity as a function of time. (a) What are the ball's average speed and average velocity during the first 3.0...
-
The 2014-T6 aluminum column has the cross section shown. If the column is pinned at both ends and subjected to an axial force \(P=100 \mathrm{kN}\), determine the maximum length of the column. 15 mm]...
-
City Cab Company identified 10 primary pickup and drop locations for cab riders in New York City. In an effort to minimize travel time and improve customer service and the utilization of the companys...
-
Find Find lim x - sin x limx In x +0x Evaluate fre'dx
-
Calculate the NPV of the proposed overhaul of the Vital Spark, with and without the new engine and control system. To do the calculation, you will have to prepare a spreadsheet table showing all...
-
Camilla uses emalto communicate with clents and often sendsiout marketing imaterial to her existing clients and prospects Which of the following statements about the legislation thatgoverns...
-
What do we call a company that is created and jointly owned by two or more independent entities to achieve a common business objective?
-
In what production technique is inventory kept to a minimum and inputs arrive to the production process exactly when they are needed?
-
Home-country expatriates manage operations abroad in ethnocentric staffing, and the best qualified candidate from anywhere manages operations in geocentric staffing. What are some key reasons...
-
Why might households and firms in a foreign country prefer to use U.S. dollars rather than their own countrys currency in making transactions? What advantages or disadvantages do foreign governments...
-
Which of the following individuals must file a tax return in 2017? a. Carolyn is single and age 66. She receives $2,000 of interest income, $3,000 of dividend income, and $6,000 in Social Security...
-
Answer to the following: Discuss how a company's internal environment might affect the development of the corporate strategy. Include product life cycle, personnel, and organizational structure in...
-
Consider a game of poker being played with a standard 52-card deck (four suits, each of which has 13 different denominations of cards). At a certain point in the game, six cards have been exposed. Of...
-
A number of salts containing the tetrahedral polyatomic anion, BF4- , are ionic liquids, whereas salts containing the somewhat larger tetrahedral ion SO42- do not form ionic liquids? Explain this...
-
A sample of 0.2140 g of an unknown monoprotic acid was dissolved in 25.0 mL of water and titrated with 0.0950 M NaOH. The acid required 27.4 mL of base to reach the equivalence point. (a) What is the...
-
(a) What is the frequency of radiation that has a wavelength of 10m, about the size of a bacterium? (b) What is the wavelength of radiation that has a frequency of 5.50 1014s-1? (c) Would the...
-
Brooks Development Corporation (BDC) faces the following capital budgeting decision. Six real estate projects are available for investment. The net present value and expenditures required for each...
-
Morgan Inc. is planning the purchase of one of the component parts it needs for its finished product. The anticipated demands for the component for the next 12 periods are shown in the following...
-
John White is the program scheduling manager for the television channel CCFO. John would like to plan the schedule of television shows for next Wednesday evening. The table below lists nine shows...

Study smarter with the SolutionInn App




