8. One pound of an ideal gas undergoes an isentropic process from 95.3 psig and a...
Fantastic news! We've Found the answer you've been seeking!
Question:
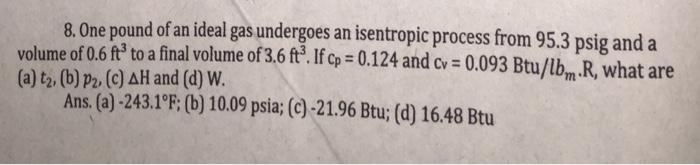
Transcribed Image Text:
8. One pound of an ideal gas undergoes an isentropic process from 95.3 psig and a volume of 0.6 ft³ to a final volume of 3.6 ft°. If cp = 0.124 and cv = 0.093 Btu/lb, R, what are (a) t2, (b) P2. (c) AH and (d) W. Ans. (a)-243.1 F; (b) 10.09 psia; (c) -21.96 Btu; (d) 16.48 Btu %3D %3D 8. One pound of an ideal gas undergoes an isentropic process from 95.3 psig and a volume of 0.6 ft³ to a final volume of 3.6 ft°. If cp = 0.124 and cv = 0.093 Btu/lb, R, what are (a) t2, (b) P2. (c) AH and (d) W. Ans. (a)-243.1 F; (b) 10.09 psia; (c) -21.96 Btu; (d) 16.48 Btu %3D %3D
Expert Answer:
Answer rating: 100% (QA)
One pound of an ideal gas undergoes an isenbropic proc... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
One mole of an ideal gas undergoes an isothermal reversible expansion at 25oC. During this process, the system absorbs 855 J of heat from the surroundings. When this gas is compressed to the original...
-
One mole of an ideal gas undergoes an isothermal compression at 0 oC, and 7.5 x 103 J of work is done in compressing the gas. (a) Will the entropy of the gas (1) increase, (2) remain the same, or (3)...
-
A system consisting of n mol of an ideal gas undergoes two reversible processes. It starts with pressure Pi and volume Vi, expands isothermally, and then contracts adiabatically to reach a final...
-
A physical pendulum of mass m = 3 . 3 6 kg is comprised of an odd shape that has a centre - of - mass a distance of d = 0 . 5 5 5 m from the pivot point. The pendulum is displaced from equilibrium to...
-
What are the general rules related to the amortization of intangible assets ? Read the basis for conclusions in SFAS No. 142, particularly paragraphs B49 through B53. What were the amortization rules...
-
The Editions Sorbonne is wholly owned by the university. It performs the bulk of its work for other university departments, which pay as though the Editions Sorbonne were an outside business...
-
Lisa Sheraton, accountant for Chase Home Finance, was injured in a boating accident. Another employee prepared the accompanying income statement for the year ended December 31, 2008. The individual...
-
Boulder Mines paid $425,000 for the right to extract ore from a 250,000-ton mineral deposit. In addition to the purchase price, Boulder Mines also paid a $110 filing fee, a $2,000 license fee to the...
-
Solve each of the following three problems, all of which involve borrowing money from a bank with an APR of 6.5% compounded annually. Look carefully at how the problems differ from one another, in...
-
Three employees in the maintenance department are responsible for repairing the video games at Pinball Wizard, a video arcade. A maintenance worker can fix one video game machine every B hours on...
-
If you were a hedge fund manager, which style would you employ and why? Describe this style and how it works. Why would you utilize it and under what macroeconomic conditions would this style work...
-
cial decision. 2 4. You as the manager of Vertex Company have been asked by your president to purchase a machine for your production department. A new machine can be purchased for Tk. 5,50,000 with...
-
Randall UPS is a supplier of a variety of services for small businesses. Commercial printer, scanning, and copier equipment is being considered to replace a five - year - old machine. After the...
-
41) Assume that you purchase a 10-year Rs 1,000 par value bond, with a 12% coupon, and a yield of 9 Immediately after you purchase the bond, yields fall to 8% and remain at that level to maturity...
-
On July 1, 2023 Bramble Ltd. purchased 6% bonds having a maturity value of $70,000 for $67,594. The bonds provide the bondholders with a 7% yield. The bonds mature four years later, on July 1, 2027,...
-
Calculate the following ratios for Walmart and a competitor for two years. Present your calculations in a table. Liquidity ratios: 1. Working Capital = Current Assets - Current Liabilities 2. Current...
-
My observations of the budget prepared for Bruin Sales and Service are that it appears to be well-structured and comprehensive. The budget takes into consideration various factors such as seasonality...
-
A container holds 2.0 mol of gas. The total average kinetic energy of the gas molecules in the container is equal to the kinetic energy of an 8.0 10-3-kg bullet with a speed of 770 m/s. What is the...
-
A mixture contains only NaCl and Fe(NO 3 ) 3 . A 0.456- g sample of the mixture is dissolved in water, and an excess of NaOH is added, producing a precipitate of Fe(OH) 3 . The precipitate is...
-
Will a crystalline solid or an amorphous solid give a simpler X-ray diffraction pattern? Why?
-
In Exercise 112 in Chapter 5, the pressure of CO2 in a bottle of sparkling wine was calculated assuming that the CO2 was insoluble in water. This was an incorrect assumption. Redo this problem by...
-
Through the implementation of efficient and effective management, New Belgium Brewing (NBB) has grown from operating out of the founder's basement to having two state-of-the-art facilities and over...
-
Define management and indicate what its principal purposes are and why managers are essential to organizations.
-
What are the resources of the organization? How are these used in management decision making?

Study smarter with the SolutionInn App


