Dipole moment of -X is 1.6D. What is the dipole moment of the following compound: Dipole...
Fantastic news! We've Found the answer you've been seeking!
Question:
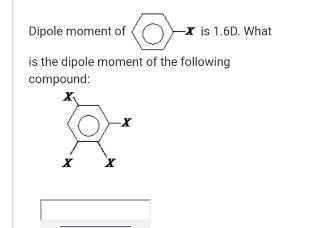
Transcribed Image Text:
Dipole moment of -X is 1.6D. What is the dipole moment of the following compound: Dipole moment of -X is 1.6D. What is the dipole moment of the following compound:
Expert Answer:
Answer rating: 100% (QA)
Dipole moment of the compound 16 D O dipole moment giver by these groups ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The ammonia molecule (NH3) has a dipole moment of 5.0 X 10-30 C m. Ammonia molecules in the gas phase are placed in a uniform electric field E with magnitude 1.6 X 106 N/c. (a) What is the change in...
-
The following compound has two isomers: ClCH==CHCl One isomer has a dipole moment of 0 D, and the other has a dipole moment of 2.95 D. Propose structures for the two isomers that are consistent with...
-
The dipole moment of the water molecule (H20) is 6.17 X 10-30 C. m. Consider a water molecule located at the origin whose dipole moment p points in the + x-direction. A chlorine ion (C1-), of charge...
-
What are the advantages and disadvantages of Qantass international cooperative alliances? The Qantas Group maintained its strong position in the Australian domestic market in 2016/17. Through a dual...
-
A rigid bar AB of length L = 66 in. is hinged to a support at A and supported by two vertical wires attached at points C and D (see figure). Both wires have the same cross-sectional area (A = 0.0272...
-
Consider the difference equation yn = -3yn-1 + 8, y0 = 1. (a) Generate y1, y2, y3 from the difference equation. (b) Solve the difference equation. (c) Use the solution in part (b) to obtain y4?
-
If the probability is 0.90 that a new machine will produce 40 or more chairs, find the probabilities that among 16 such machines (a) 12 will produce 40 or more chairs; (b) at least 10 will produce 40...
-
Assume that Whee, Cheatham, and Howe is an auditing firm that has found that its summer interns are subject to a 90 percent learning curve for one of its important tasks, proofreading financial...
-
Presented below are selected ledger accounts of Concord Corporation as of December 31, 2025. Cash Administrative expenses $44,000 88,000 Selling expenses Net sales 70,400 475,200 Cost of goods sold...
-
Eleven-Eleven, Inc. creates guided meditation programs for individuals. On November 30, 20Y9, the balances of selected accounts of Eleven-Eleven, Inc. are as follows: Prepare a classified balance...
-
We need to conduct the Industry Analysis for Barriers to Entry on the industries of Pakistan. 1. You need to work on at least 5 Industries of Pakistan. Choose any of the 5 industries of Pakistan of...
-
Royal Lawncare Company produces and sells two packaged products Weed ban and Green grow. Revenue and cost information relating to the products follow: Product Weed ban Green grow Selling price per...
-
In 1929, U.S. GDP was, adjusted for inflation, $1,056.6 billion. The following year it was, also adjusted for inflation, $966.7 billion. What was the growth rate of U.S. in 1930? (Do not type the...
-
The parents stay for a while and play with their child and the other children and seem happy to leave. They say goodbye and their child cries initially when they leave and needs comforting. You sit...
-
What role do telomeres play in maintaining chromosome stability, and how do they relate to cellular aging and cancer development? Explain
-
Selected comparative financial statements of Korbin Company follow. Sales KORBIN COMPANY Comparative Income Statements For Years Ended December 31 2021 2020 $ 460,845 $ 353,045 Cost of goods sold...
-
Use the given trigonometric ratio, which is based on actual side length measures, to sketch a right triangle and solve the triangle. 1 sin (A) Solution: Sketch:
-
How do network effects help Facebook fend off smaller social-networking rivals? Could an online retailer doing half as much business compete on an equal footing with Amazon in terms of costs? Explain.
-
How can the following compounds be prepared, starting with a carbonyl compound with one fewer carbon atoms than the desired product? a. HOCH 2 CH 2 NH 2 b. CH3CHOH
-
For each of the following compounds, indicate which is the stronger acid: a. CH3OCH2CH2OH or CH3CH2CH2CH2OH b. c. CH3OCH2CH2CH2OH or CH3CH2OCH2CH2OH d. CH CH CH,NH or CHaCH2CH2OH CH3CCH2OH or...
-
Give the systematic name for each of the following structures: a. b. c. d. CH C CCH2CHCH3 CHCH,CH3 CH CH3C CCH2CCH3 CH CHCH-C CCHCH CI CH3
-
a. For the allowed energies of a particle in a box to be large, should the box be very big or very small? Explain. b. Which is likely to have larger values for the allowed energies: an atom in a...
-
The molecules in the rods and cones in the eye are tuned to absorb photons of particular energies. The retinal molecule, like many molecules, is a long chain. Electrons can freely move along one...
-
What was the approximate activity of the plutonium source at the start of the mission? A. \(2 \times 10^{21} \mathrm{~Bq}\) B. \(2 \times 10^{19} \mathrm{~Bq}\) C. \(2 \times 10^{17} \mathrm{~Bq}\)...

Study smarter with the SolutionInn App


