12. (Hyperbolic angle measure) Consider the upper-half plane model for the hyperbolic plane H, where the...
Fantastic news! We've Found the answer you've been seeking!
Question:
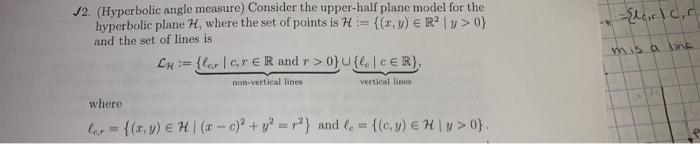

Transcribed Image Text:
12. (Hyperbolic angle measure) Consider the upper-half plane model for the hyperbolic plane H, where the set of points is H:= {(x, y) = R²|y>0} and the set of lines is LH:= {ler | cr ER and r>0}U{le | CER}, non-vertical lines. vertical lines where ler = {(x, y) = H | (x-c)² + y2 = r²} and f = {(cy) € Hy>0}. Elcrc, c mis a line Find the critical number ro = || r(P, ) for the line and the external point P, where l = lo, lo = {(x, y) = H | x=0}, and P = (1, 1). Use the following steps: (a) Suppose m is the line containing P such that ml. Show m = lo.v₂. (b) Show that m and intersect at the point (0, √2), call this point D. 12. (Hyperbolic angle measure) Consider the upper-half plane model for the hyperbolic plane H, where the set of points is H:= {(x, y) = R²|y>0} and the set of lines is LH:= {ler | cr ER and r>0}U{le | CER}, non-vertical lines. vertical lines where ler = {(x, y) = H | (x-c)² + y2 = r²} and f = {(cy) € Hy>0}. Elcrc, c mis a line Find the critical number ro = || r(P, ) for the line and the external point P, where l = lo, lo = {(x, y) = H | x=0}, and P = (1, 1). Use the following steps: (a) Suppose m is the line containing P such that ml. Show m = lo.v₂. (b) Show that m and intersect at the point (0, √2), call this point D.
Expert Answer:
Answer rating: 100% (QA)
a To locate the line that is perpendicular to line and contains point P 2 0 Since ll oxyR 2 x0 we mu... View the full answer

Related Book For 

Income Tax Fundamentals 2013
ISBN: 9781285586618
31st Edition
Authors: Gerald E. Whittenburg, Martha Altus Buller, Steven L Gill
Posted Date:
Students also viewed these mathematics questions
-
derive a shwarz-christoffel transformation mapping the upper half plane onto the triangle X, =-1 X, = 1 w, = 0 w, = 1 z-plane w-plane
-
Consider the upper half of the astroid described by x 2/3 + y 2/3 = a 2/3 , where a > 0 and |x| a. Find the area of the surface generated when this curve is revolved about the x-axis. Use symmetry....
-
Suppose that points x and y are fixed in R" and set a) Prove that if for (a, b) R2, then the system is solved by And b) Prove that if a0 and b0 are given by part a), then the straight line whose...
-
Information sent electronically over the Internet affords little privacy. One of the reasons that cryptography is included in this text is because its importance is growing due to the increasing need...
-
The following hypotheses are given. H0: .70 H1: > .70 A sample of 100 observations revealed that = .75. At the .05 significance level, can the null hypothesis be rejected? a. State the decision...
-
In Sec. 1410, Eq. (a) is given for Ks based on the procedure in Ex. 142. Derive this equation. A speed-reducer has 20 full-depth teeth, and the single-reduction spur-gear gearset has 22 and 60 teeth....
-
Consider the calculation of an external rate of return (ERR). The positive cash flows in the cash flow profile are moved forward to \(t=n\) using what value of \(i\) in the \((F \mid P i, n-t)\)...
-
Friendleys Miniature Golf and Driving Range Inc. was opened on March 1 by Dean Barley. These selected events and transactions occurred during March. Mar. 1 Stockholders invested $50,000 cash in the...
-
1-2. Draw graphs of the following functions using transformations: 1. y = 2x-1 x-2' 2. y log2x+1|- 3.
-
Sandra works in British Columbia and is paid on a semi-monthly basis. She has a claim code of 1 on both her TD1 and TD1BC. She has the following types of earnings, benefits & deductions. She will...
-
The rise of fluent writers in English during the post-war period up to the present shows how flexible, imaginative, and talented Filipinos are. Rizal's influence is eminent among the social realist...
-
Kevin borrows $7100 on January 1 for one year interest is calculated annually. He does not have to make any monthly payments but he hast to repay the entire 1 interest on December 31 of the same year...
-
Suppose F() is the Fourier transform of f(x). If fx|f(x)|dx \(x)|dx 5 |F()|d S\F()|d Ex www exist, then ExE 1/1. and Ep
-
18 LT 7 +16% +100 first complete the square to write the denominator in the form (x+p) + q To solve the indefinite integral = dx
-
Directions: Practice your banking skills by completing the following exercise. Don't forget to record all of your checks in the check register. 1. On October 3rd of this year, your received a...
-
TransLink has announced the Canada Line will be extended to Steveston. The train will take one of two routes No. 3 Road or Garden City. You have an opportunity to purchase some property on each of...
-
Four members from a 17 -person committee are to be selected randomly to serve as chairperson, vice-chairperson, secretary, and treasurer. The first person selected is the chairperson; the second, the...
-
The maximum pressure that can be developed for a certain fluid power cylinder is 15.0 MPa. Compute the required diameter for the piston if the cylinder must exert a force of 30 kN.
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Phil and Linda are 25-year-old newlyweds and file a joint tax return. Linda is covered by a retirement plan at work, but Phil is not. a. Assuming Phil's wages were $27,000 and Linda's wages were...
-
Quince Corporation has taxable income of $450,000 for its 2012 calendar tax year. Calculate the corporation's income tax liability for 2012 before tax credits. $_________
-
A blender does 5000 J of work on the food in its bowl. During the time the blender runs, 2000 J of heat is transferred from the warm food to the cooler environment. What is the change in the thermal...
-
Which system contains more atoms: 5 mol of helium (A = 4) or 1 mol of neon (A = 20)? A. Helium B. Neon C. They have the same number of atoms.
-
A sample of ideal gas is in a sealed container. The temperature of the gas and the volume of the container are both increased. What other properties of the gas necessarily change? (More than one...

Study smarter with the SolutionInn App


