(13%) Problem 8: Suppose the first-order maximum for 460 nm blue light being transmitted through a...
Fantastic news! We've Found the answer you've been seeking!
Question:
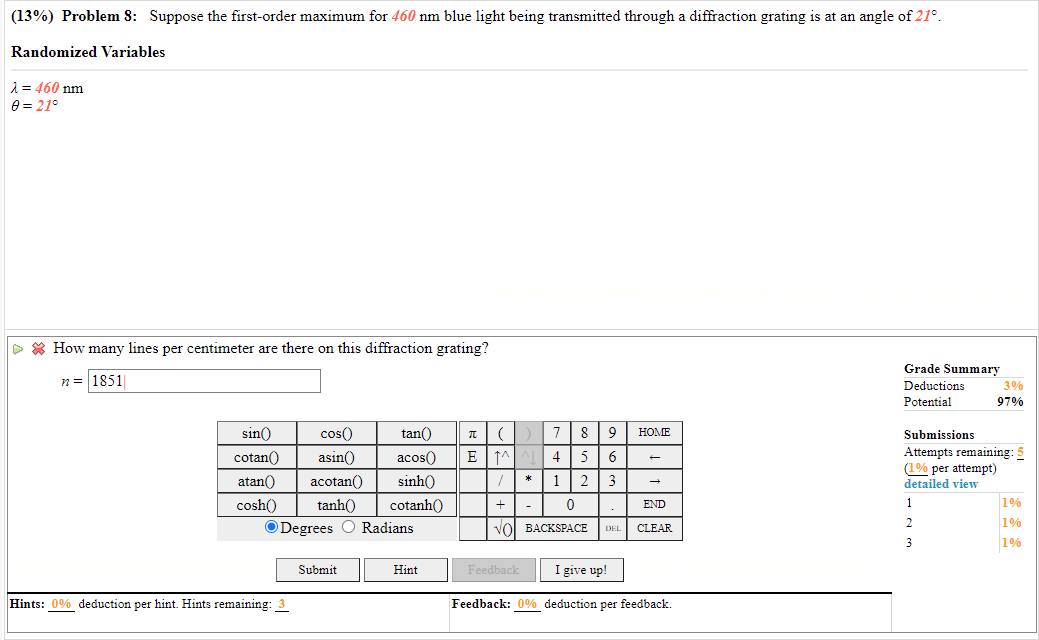
Transcribed Image Text:
(13%) Problem 8: Suppose the first-order maximum for 460 nm blue light being transmitted through a diffraction grating is at an angle of 21. Randomized Variables = 460 nm 0 = 21 How many lines per centimeter are there on this diffraction grating? n = 1851 sin() cos() 6 cotan asin() atan() acotan() tan() () 7 89 acos( EM^^ 4 5 sinh() 1 2 3 cosh() tanh() cotanh() ODegrees O Radians + 0 VO BACKSPACE Hints: 0% deduction per hint. Hints remaining: 3 Submit Hint * - . DEL Feedback I give up! HOME END CLEAR Feedback: 0% deduction per feedback. Grade Summary Deductions. Potential Submissions Attempts remaining: 5 (1% per attempt) detailed view 1 2 3% 97% 3 1% 1% 1% (13%) Problem 8: Suppose the first-order maximum for 460 nm blue light being transmitted through a diffraction grating is at an angle of 21. Randomized Variables = 460 nm 0 = 21 How many lines per centimeter are there on this diffraction grating? n = 1851 sin() cos() 6 cotan asin() atan() acotan() tan() () 7 89 acos( EM^^ 4 5 sinh() 1 2 3 cosh() tanh() cotanh() ODegrees O Radians + 0 VO BACKSPACE Hints: 0% deduction per hint. Hints remaining: 3 Submit Hint * - . DEL Feedback I give up! HOME END CLEAR Feedback: 0% deduction per feedback. Grade Summary Deductions. Potential Submissions Attempts remaining: 5 (1% per attempt) detailed view 1 2 3% 97% 3 1% 1% 1%
Expert Answer:
Answer rating: 100% (QA)
Answer Let a be the width of clear space and b be the width of a ... View the full answer

Related Book For 

Posted Date:
Students also viewed these physics questions
-
A diffraction grating is 1.50 cm wide and contains 2400 lines. When used with light of a certain wavelength, a third-order maximum is formed at an angle of 18.0. What is the wavelength (in nm)?
-
A force vector points at an angle of 52o above the +x axis. It has a y component of +290 newtons. Find (a) The magnitude and (b) The x component of the force vector?
-
An airfoil at an angle of attack α, as in Fig P3.150, provides lift by a Bernoulli effect, because the lower surface slows the flow (high pressure) and the upper surface speeds up the flow (low...
-
A construction company is considering taking on a new project. The total project cost is $5 million, and there is a 70% chance that the project will be completed on time and within budget, resulting...
-
Jessel Corporation bases its variable overhead performance report on the actual direct labor-hours of the period. Data concerning the most recent year that ended on December 31 are as follows:...
-
Leah is flying from Boston to Denver with a connection in Chicago. The probability her first flight leaves on time is 0.15. If the flight is on time, the probability that her luggage will make the...
-
For each of the following bond scenarios \(a\) through \(e\) described below, identify a bond type that applies. a. Harrison County Flood Control Improvement Bonds issued in the state of Texas. b....
-
Willingham Company has the following comparative balance sheet data. Additional information for 2014: 1. Net income was $25,000. 2. Sales on account were $410,000. Sales returns and allowances were...
-
I need help with this HW questions. Thank you!!. Common Stock, $6 par, 100,000 shares authorized, 50,000 shares Issued & 300,000 outstanding Paid-In Capital in excess of par 170,000 Retained Earni...
-
(a) A horizontal steel I-beam of cross-sectional area 0.041m2 is rigidly connected to two vertical steel girders. If the beam was installed when the temperature was 30oC, what stress is developed in...
-
Q3. Which of the following ions will give test of all the ions present in it: (a) Mohr's salt (b) K2[Fe(CN)6] (c) pdC142- (d) [Co(Cl)3(NH3)3]
-
(A) Draw Mohr's circle representing the state of plane stress for a spherical pressure vessel of radius r and wall thickness t, which enclosed a pressurized fluid of pressure, P. (B) Draw Mohr's...
-
The elementary step reactions for burning of a model compound is: k1 02-20. k2 0 +H20 20H k3 CO. +OH CO2+H k4 H +02 OH +0. Derive the rate expression for oxygen radicals (O.)
-
Calculate the half-cell potential of Ni in 0.1M NiCl2. The half-cell is represented as: NiNi2+, Cl- (0.1M). Cation Anion Ag+ +77.1 Br -102.8 Al3+ -483.1 cl -131.1 Au+ +410.8 F -274.8 Ca2+ +555.1...
-
The nurse is caring for a client with hypertension, gastroesophageal reflux, and osteoarthritis. While performing a bedside assessment, the nurse observes that the client is alert and oriented but is...
-
a) When the NO concentration is doubled, the rate of the reaction 2 NO(g) + O2(g) = 2 NO2 (g) increases by a factor of 4. When both the O2 and NO concentrations are doubled, the rate increases by a...
-
Take a few minutes to watch the video The Jar of Life . Big rocks are the important things in life. Illustrating the concept of a typically busy life with rocks and sand, Stephen Covey tells us that...
-
Annual dividends of ATTA Corp grew from $0.96 in 2005 to $1.76 in 2017. What was the annual growth rate?
-
The drawing shows a container that is partially filled with 2.0 grams of water. The temperature is maintained at a constant 20 °C. The space above the liquid contains air that is completely...
-
A circuit contains a resistor in series with a capacitor, the series combination being connected across the terminals of a battery, as in Figure 20.37a. The time constant for charging the capacitor...
-
The mass of a string is 5.0 10-3 kg, and it is stretched so that the tension in it is 180 N. A transverse wave traveling on this string has a frequency of 260 Hz and a wavelength of 0.60 m. What is...
-
Baerg & Vetzel, a public accounting firm, were the auditors of South-Western Development, Inc., a real estate company that owned several shopping centres in southwest ern Ontario. It was...
-
A partner in your public accounting firm has asked you to explain to a group of juniors the importance of engagement letters and the importance of following up unusual or unexplained items discovered...
-
Marino Rossi, a public accountant, audited the finan cial statements of Newfoundland Rugs Ltd. Cooke, the pres ident of Newfoundland Rugs, told Rossi that the company was planning a private placement...

Study smarter with the SolutionInn App


