13. Which of the following would be the most likely to undergo an E1 elimination reaction with...
Fantastic news! We've Found the answer you've been seeking!
Question:
13. Which of the following would be the most likely to undergo an E1 elimination reaction with a very strong base, such as the (CH3)3CO ion?

24. Identify the compound in each of the following pairs that react at the faster rate in an SN1
reaction:
(a) Isopropyl bromide or isobutyl bromide
(b) Cyclopentyl iodide or 1-methylcyclopentyl iodide
(c) Cyclopentyl bromide or 1-bromo-2,2-dimethylpropane
(d) tert-Butyl chloride or tert-butyl iodide
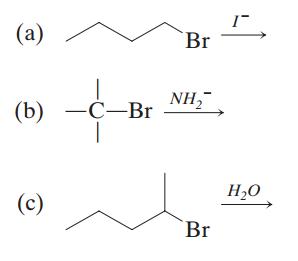
26. Predict whether the following reactions are more likely to undergo elimination or substitution.
Identify the mechanism of the dominant reaction (E1 versus E2; SN1 versus SN2).
Related Book For 

Auditing A Practical Approach with Data Analytics
ISBN: 978-1119401742
1st edition
Authors: Raymond N. Johnson, Laura Davis Wiley, Robyn Moroney, Fiona Campbell, Jane Hamilton
Posted Date:




