14) Which of the following compounds is optically active: a. trans-1-bromo-3-chlorocyclobutane b. cis-1,4-dimethylcyclohexane c. cis-1,3-dimethylcyclohexane d....
Fantastic news! We've Found the answer you've been seeking!
Question:


Transcribed Image Text:
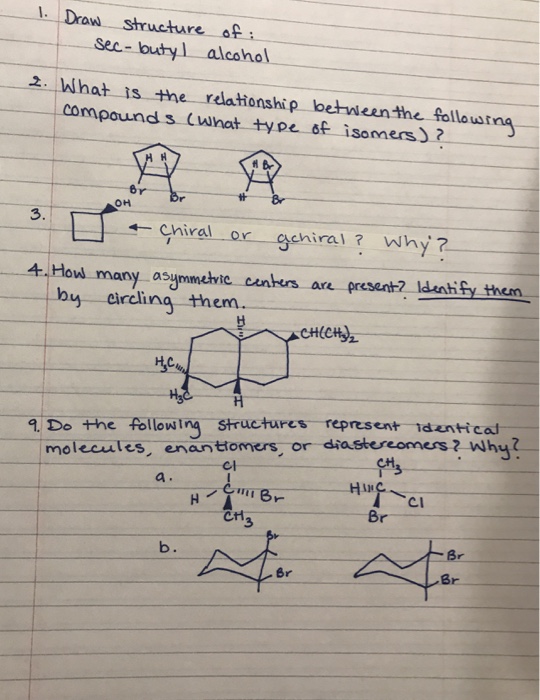
14) Which of the following compounds is optically active: a. trans-1-bromo-3-chlorocyclobutane b. cis-1,4-dimethylcyclohexane c. cis-1,3-dimethylcyclohexane d. trans-1,3-dimethylcyclohexane CHS MC CH3 CHE CH3 CH3 Br 6) Which of the following compounds (choose only one) is never chiral? (4 points) 2,3-dibromobutane; 1,3-dibromobutane; 1,2-dichlorobutane; 1,4-dibromobutane; and 1-bromo-2-chlorobutane. 1. Draw structure of: Sec-butyl alcohol 2. What is the relationship between the following compounds (what type of isomers)? 3. HH Br OH Chiral or gchiral? Why? 4. How many asymmetric centers are present? Identify them by circling them. CH(CH₂)₂ a. Br H A H₂C H₂C 9. Do the following structures represent identical molecules, enantiomers, or diastereomers? Why? CI CH3 I C Br A CH3 b. Br H- Br HINC Br CI Br Br 14) Which of the following compounds is optically active: a. trans-1-bromo-3-chlorocyclobutane b. cis-1,4-dimethylcyclohexane c. cis-1,3-dimethylcyclohexane d. trans-1,3-dimethylcyclohexane CHS MC CH3 CHE CH3 CH3 Br 6) Which of the following compounds (choose only one) is never chiral? (4 points) 2,3-dibromobutane; 1,3-dibromobutane; 1,2-dichlorobutane; 1,4-dibromobutane; and 1-bromo-2-chlorobutane. 1. Draw structure of: Sec-butyl alcohol 2. What is the relationship between the following compounds (what type of isomers)? 3. HH Br OH Chiral or gchiral? Why? 4. How many asymmetric centers are present? Identify them by circling them. CH(CH₂)₂ a. Br H A H₂C H₂C 9. Do the following structures represent identical molecules, enantiomers, or diastereomers? Why? CI CH3 I C Br A CH3 b. Br H- Br HINC Br CI Br Br
Expert Answer:
Answer rating: 100% (QA)
14 D option is correct Explanation Only compound D does not have a p... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Which of the following compounds is easier to decarboxylate? NCOH
-
Which of the following compounds is more easily decarboxylated? CH2CH2 or
-
Which of the following compounds is more likely to exhibit activity as a tranquilizer? CH3 CH CH O or CH C
-
The largest government expenditure for Japan & the US is... a) military / defense b) transfer payments c) education / healthcare d) infrastructure
-
In building supply chain competencies, what are the trade-offs that must be considered?
-
Critique the following research done in a service industry as to the extent to which it meets the hallmarks of scientific investigation discussed in this chapter. The Mobile Phone Company the Mobile...
-
Explain how deductions and tax credits affect the amount of income tax you pay.
-
As president of Youngs of California, a large clothing chain, you have just received a letter from a major stockholder. The stockholder asks about the companys dividend policy. In fact, the...
-
Textra produces parts for a machine manufacturer. Parts go through two departments, Molding and Trimming. The company budgets overhead cost of $262,500 in the Molding department and $222,500 in the...
-
On January 1. Ruiz Company issued bonds as follows: Face Value: Number of Years: Stated Interest Rate: Interest payments per year 500,000 15 7% Required: 1) Calculate the bond selling price given the...
-
How would you use the content sharing capabilities of LinkedIn for personal branding? How to use then?
-
What does the high cost of replacing an international manager suggest regarding staffing philosophy?
-
The makes of cars sold by a particular car dealer In Exercises 1524, determine whether the data described are qualitative or quantitative.
-
A pollster obtains a list of all the residential addresses in a certain town, and uses a computer random number generator to choose 150 of them. The pollster visits each of the 150 households and...
-
How easy or difficult do you think it is to handle the equity issue in international compensation?
-
The director of the recreation center at a large university wants to sample 100 students to ask them whether they would support an increase in their recreation fees in order to expand the hours that...
-
Question 1 : Is ethical decision making always rational and logical? Why? Why not? What approaches are often taken when educating pre-service teachers about ethics? How do you now understand the...
-
On 1 July 2021, Croydon Ltd leased ten excavators for five years from Machines4U Ltd. The excavators are expected to have an economic life of 6 years, after which time they will have an expected...
-
Determine the partial negative charge on the oxygen atom in a C=O bond. The bond length is 1.22 * and the bond dipole moment is 2.30 D.
-
What m zvalue is most likely for the base peak in the mass spectrum of 3-methylpentane?
-
What would be the results of the experiment in Problem 39 if radioactive AMP were added to the incubation mixture instead of radioactive pyrophosphate?
-
Simplify the irrational number \(\sqrt{550}\) and express in lowest terms. Identify the rational and irrational parts.
-
Identify which of the following numbers are irrational. 1. \(\sqrt{225}\) 2. \(3 \sqrt{5}\) 3. \(\sqrt{80}\) 4. \(20-3 \pi\)
-
Determine which of the following are perfect squares. 1. 36 2. 27 3. \(\frac{9}{49}\) 4. \(\frac{12}{221}\)

Study smarter with the SolutionInn App


