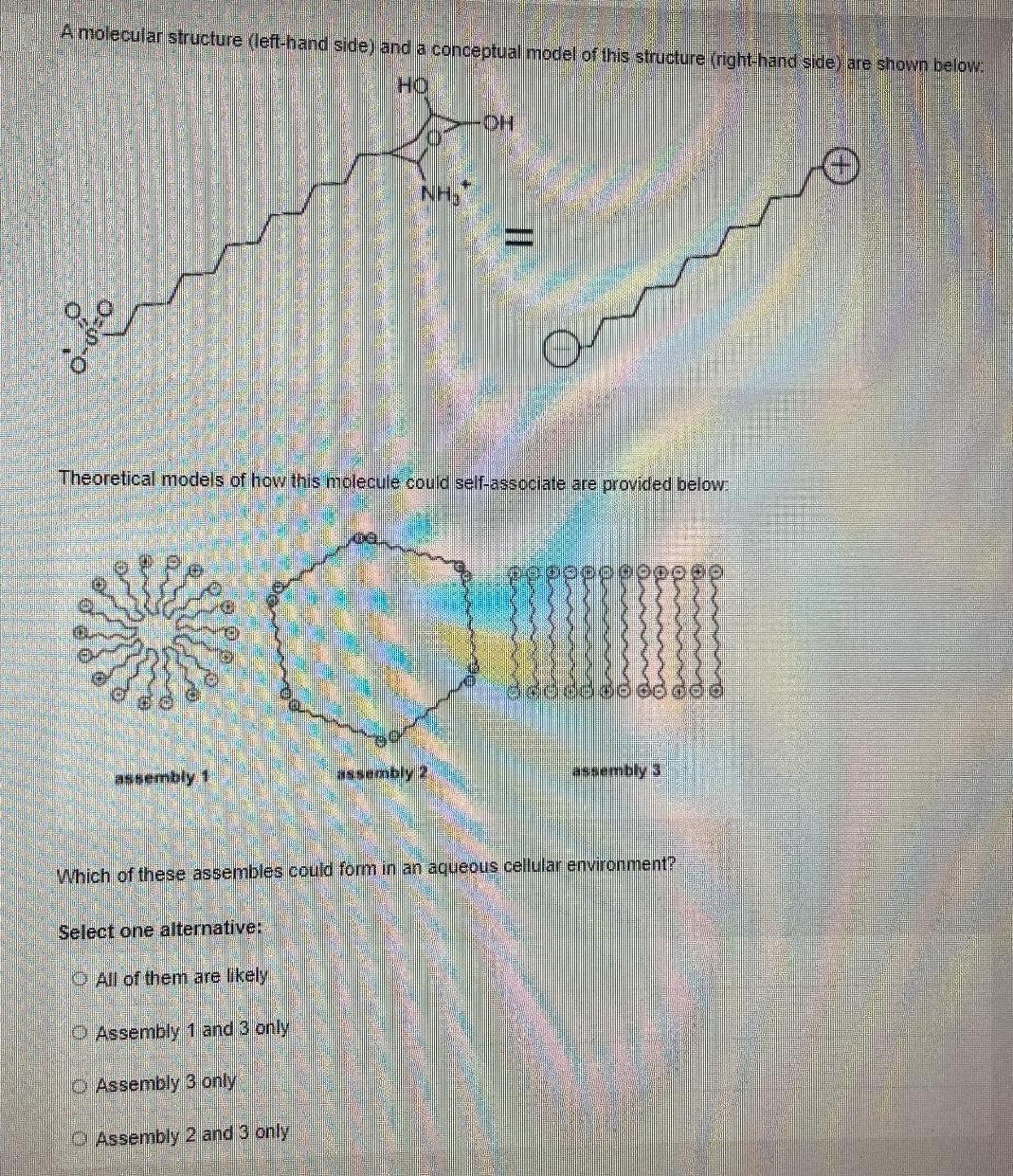
A molecular structure (left-hand side) and a conceptual model of this structure (righthand side) are shown...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
A molecular structure (left-hand side) and a conceptual model of this structure (righthand side) are shown below HO NH3 Theoretical models of how this molecule could self-associate are provided below: assembly 1 assembly 2 assembly 3 Which of these assembles could form in an aqueous cellular environment? Select one alternative: O All of them are likely O Assembly 1 and 3 only O Assembly 3 only O Assembly 2 and 3 only A molecular structure (left-hand side) and a conceptual model of this structure (righthand side) are shown below HO NH3 Theoretical models of how this molecule could self-associate are provided below: assembly 1 assembly 2 assembly 3 Which of these assembles could form in an aqueous cellular environment? Select one alternative: O All of them are likely O Assembly 1 and 3 only O Assembly 3 only O Assembly 2 and 3 only
Expert Answer:
Answer rating: 100% (QA)
ONLY Assembly 1 3 are possible Because only chargedpositive and negative end of the ... View the full answer

Posted Date:
Students also viewed these biology questions
-
Below on the left side are models of two atoms, one from a metal, the other from a nonmetal. On the right side are corresponding monatomic ions of those atoms. Decide which of these ions is the...
-
Below are shown three C4H8C12 isomers on the left and three sets of 1H NMR data that one would expect on application of the simple N + 1 rule on the right. Match the structures to the proper spectral...
-
Molecular orbitals are most commonly delocalized throughout the molecule and exhibit distinct bonding or anti-bonding character. Loss of an electron from a specific molecular orbital from excitation...
-
In Table 4. 4, Professor Kip Viscusi estimates that the cost per life saved by current government risk-reducing programs ranges from $100,000 for unvented space heaters to $72 billion for a proposed...
-
You are the assistant to the information security director. Your director is interested in hiring a new group of IT staff professionals. Required a. Write a report to your director explaining the...
-
The tropical year, the time from vernal equinox to the next vernal equinox, is the basis for our calendar. It contains 365.242199 days. Find the number of seconds in a tropical year.
-
Camille Stibbe is general manager of Alden Corporation. She suggests that the budgeted income statement and the cash budget seem to show the same information. Therefore, she recommends that one of...
-
When people make estimates, they are influenced by anchors to their estimates. A study was conducted in which students were asked to estimate the number of calories in a cheeseburger. One group was...
-
Review the annual report for a firm that operates in an industry other than the firms analyzed in the Group Project. The company you select must be different from those selected by your classmates...
-
A beam ABCD with a vertical arm CE is supported as a simple beam at A and D (see figure). A cable passes over a small pulley that is attached to the arm at E. One end of the cable is attached to the...
-
Paul and Mary are twins. They are 29 years old. Their father, Ned, is retiring and is leaving the farm to them. They will assume control before the next planting season. The father operated 9,000...
-
In an \(L C\) circuit, there is always some energy dissipation as electrical energy is converted to thermal energy. (a) What property of the circuit is responsible for electrical energy being...
-
1. You are a new administrator at Jonestown Medical Center. You receive a telephone call from the nurse manager of the emergency room. Dr. Smith, an emergency room physician who is an employee of...
-
A feed stream containing a mixture of 40% n-butane, 30% n-pentane, and 30% n-hexane fl ows into a fl ash unit. The fl ash temperature is 290 K and the fl ash pressure is 0.6 bar. What is the ratio of...
-
A circuit consists of an AC source, for which \(\mathscr{E}_{\max }=\) \(18 \mathrm{~V}\), in series with a \(1200-\Omega\) resistor and a capacitor. If the amplitude of the potential difference...
-
Darshan's management is pondering whether to lease the machine for \($3,000\) per year or whether to buy it outright. The company MARR is 12%. Problem data is gathered below. What is your...
-
Benefits acknowledgment the board (BRM) upgrades typical venture the executives methods through an emphasis on results (benefits) of an undertaking instead of items or results and afterward...
-
Repeat the previous problem, but close the positions on September 20. Use the spreadsheet to find the profits for the possible stock prices on September 20. Generate a graph and use it to identify...
-
Modify Table 10-1 web site for to indicate whether each control listed there is preventive, detective, or corrective in nature. 10.2Table 10-1 suggests that restricting physical access to inventory...
-
The Quality Building Supplies Company operates six wholesale outlets that sell roofing materials, electrical and plumbing supplies, lumber, and other building materials to general contractors in a...
-
The following documents are used in the revenue cycle; Customer order Sales order Sales invoice Credit memo Picking ticket Packing slip Bill of lading Monthly statement Remittance advice ...

Study smarter with the SolutionInn App

