15. Challenge: What is the ratio of protonated [HA] vs. deprotonated [A-] species for the following...
Fantastic news! We've Found the answer you've been seeking!
Question:
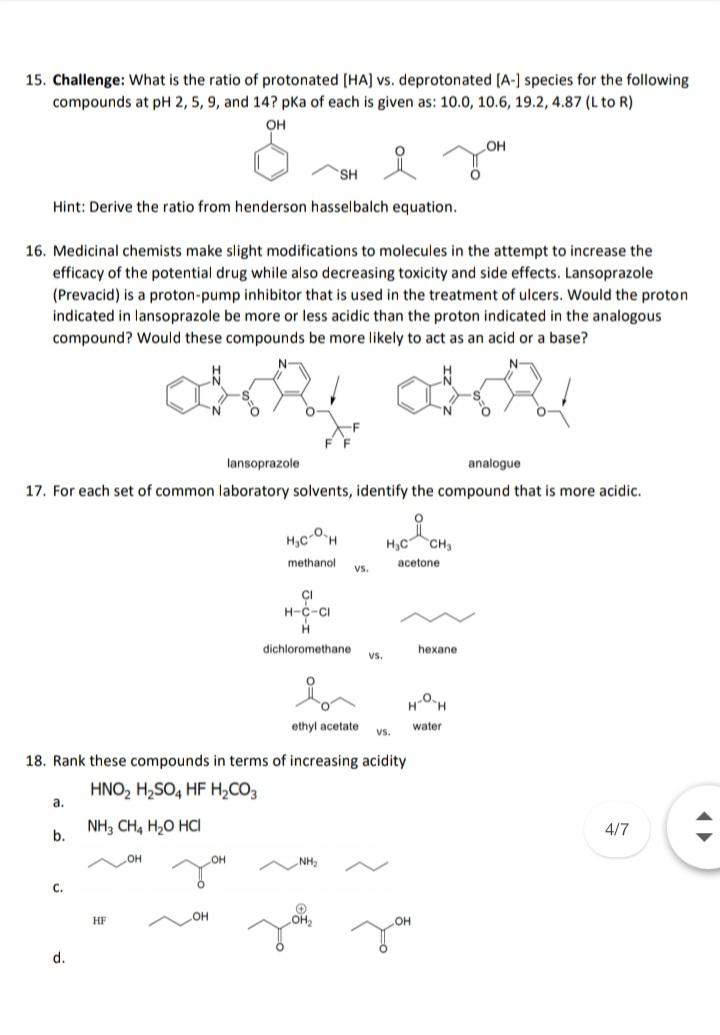
Transcribed Image Text:
15. Challenge: What is the ratio of protonated [HA] vs. deprotonated [A-] species for the following compounds at pH 2, 5, 9, and 14? pKa of each is given as: 10.0, 10.6, 19.2, 4.87 (L to R) OH OH SH Hint: Derive the ratio from henderson hasselbalch equation. 16. Medicinal chemists make slight modifications to molecules in the attempt to increase the efficacy of the potential drug while also decreasing toxicity and side effects. Lansoprazole (Prevacid) is a proton-pump inhibitor that is used in the treatment of ulcers. Would the proton indicated in lansoprazole be more or less acidic than the proton indicated in the analogous compound? Would these compounds be more likely to act as an acid or a base? N- lansoprazole analogue 17. For each set of common laboratory solvents, identify the compound that is more acidic. of H,C CH, methanol acetone Vs. CI H-C-CI dichloromethane hexane vs. ethyl acetate water v. 18. Rank these compounds in terms of increasing acidity HNO, H,SO, HF H,CO3 a. NH, CH, H,O HCI 4/7 b. NH, с. HF он d. 15. Challenge: What is the ratio of protonated [HA] vs. deprotonated [A-] species for the following compounds at pH 2, 5, 9, and 14? pKa of each is given as: 10.0, 10.6, 19.2, 4.87 (L to R) OH OH SH Hint: Derive the ratio from henderson hasselbalch equation. 16. Medicinal chemists make slight modifications to molecules in the attempt to increase the efficacy of the potential drug while also decreasing toxicity and side effects. Lansoprazole (Prevacid) is a proton-pump inhibitor that is used in the treatment of ulcers. Would the proton indicated in lansoprazole be more or less acidic than the proton indicated in the analogous compound? Would these compounds be more likely to act as an acid or a base? N- lansoprazole analogue 17. For each set of common laboratory solvents, identify the compound that is more acidic. of H,C CH, methanol acetone Vs. CI H-C-CI dichloromethane hexane vs. ethyl acetate water v. 18. Rank these compounds in terms of increasing acidity HNO, H,SO, HF H,CO3 a. NH, CH, H,O HCI 4/7 b. NH, с. HF он d.
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
In Sample Problem what is the ratio of the amplitude of the damped oscillations to the initial amplitude at the end of 20 cycles?
-
What is the ratio of the sunlight intensity reaching Pluto compared with the sunlight intensity reaching Earth? (On average, Pluto is 39 times as far from the Sun as is Earth.)
-
What is the ratio of the sunlight intensity reaching Mercury compared with the sunlight intensity reaching Earth? (On average, Mercury's distance from the Sun is 0.39 that of Earth's.)
-
In 2012, the average credit score for loans that were purchased through Fannie Mae was 766. A random sample of 35 mortgages were selected, and it was found that the average credit score was 771with a...
-
Describe the steps you will take to recruit and select (a) the engineers, (b) the clerical/secretarial workers, and (c) the MBAs.
-
It can be shown that a solution of the system below is x 1 = 3, x 2 = 2, and x 3 = -1. Use this fact and the theory from this section to explain why another solution is x 1 = 30, x 2 = 20, and x 3 =...
-
What are at least five strategies followed by smart shoppers.
-
You want to park your bicycle in a bicycle parking area where bike racks are aligned in a row. There are already N bikes parked there (each bike is attached to exactly one rack, but a rack can have...
-
REI sells snowboards. Assume the following information relates to REI's purchases of snowboards during September. During the same month, 107 snowboards were sold. REI uses a periodic inventory system...
-
In this exercise you will be assuming the role of an Account Manager working within our Personal Care Appliances category during Amazon Black Friday. You act as a general manager responsible for...
-
In 500 words or more narrate an experience that you have had with reading and writing. How might you compare this experience to one of the experiences that we read about in this weeks unit? What did...
-
Let's assume that you buy a $500,000 house. You decide to make 25% down payment and finance the rest. You get 15-year mortgage at 3.5% APR and you will make 15 annual payments to pay off the loan....
-
a) The interest rate for Mary's loan is 12=i12=5.8%. Calculate the effective monthly rate. Give your answer as a percentage to 4 decimal places. b) Calculate the effective fortnightly rate. Give your...
-
BC Ltd manufactures a single product; the standard costs per unit being variable manufacturing $9.50, fixed manufacturing $6. Selling and administrative costs are $2 per unit sold. The selling price...
-
Jansen Corp. uses machine hours as a base for its factory overhead rate. The following information is available for the year 2030: Budgeted Machine hours 273,000 hr. Factory Overhead Rate P5.00/mhr....
-
Classify the following items as per their respective category with regards to value chain, quality cost, activity based costing and theory of constraints. The items given are mismatched and needs to...
-
Write a recursive Racket function "update-if" that takes two functions, f and g, and a list xs as parameters and evaluates to a list. f will be a function that takes one parameter and checks if the...
-
Could a set of three vectors in span all of? Explain. What about n vectors in when n is less than m? R4
-
Find the energy of the photon required to excite a hydrogen atom from the n = 1 state to the n = 4 state.
-
Suppose 72.3 J of heat are added to a 101-g piece of aluminum at 20.5 C. What is the final temperature of the aluminum?
-
A Boeing KC-135A airplane has a wingspan of 39.9 m and flies at constant altitude in a northerly direction with a speed of 850 km/h. If the vertical component of the Earth's magnetic field is 5.0 ...
-
Discuss ways in which securities issued by state and local governments are similar to corporate bonds.
-
What are reasons investors buy bonds?
-
You have decided to diversify your investment portfolio. To research, compare a type of corporate bond with a type of government security.

Study smarter with the SolutionInn App


