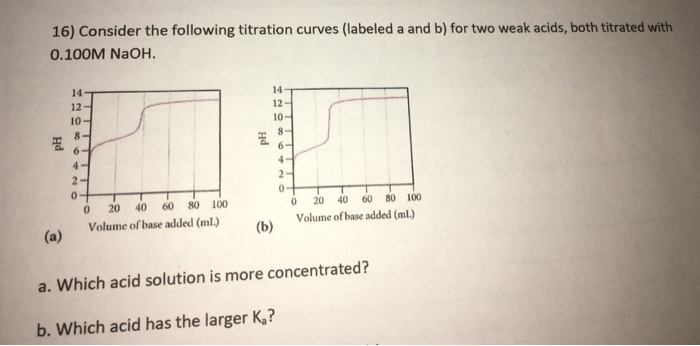
16) Consider the following titration curves (labeled a and b) for two weak acids, both titrated...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
16) Consider the following titration curves (labeled a and b) for two weak acids, both titrated with 0.100M NaOH. Hd 14 12 10- 8. 6- 4- 2- 0 20 40 60 80 100 Volume of base added (ml.) 0 Hd (b) 14- 12- 10- 8 6- 4 2- 0- 20 40 60 80 100 Volume of base added (mL.) 0 a. Which acid solution is more concentrated? b. Which acid has the larger K₂? 16) Consider the following titration curves (labeled a and b) for two weak acids, both titrated with 0.100M NaOH. Hd 14 12 10- 8. 6- 4- 2- 0 20 40 60 80 100 Volume of base added (ml.) 0 Hd (b) 14- 12- 10- 8 6- 4 2- 0- 20 40 60 80 100 Volume of base added (mL.) 0 a. Which acid solution is more concentrated? b. Which acid has the larger K₂?
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Consider the following supply and demand schedule for steel: Price per tonne ($) 20 40 60 80 100 120 140 160 180 Q demanded (million tonnes) 200 180 160 140 120 100 80 60 40 Q supplied (million...
-
We have vector x = [20 40 60 80 100 120 140 160 180 200] and we want to multiply all the elements of the vector x e.g. (20 x 40 x 60 . x 200) and store the result in temp. You can use it for the loop.
-
Find the equivalent resistance between points A and B for the group of resistors shown in FIGURE 21-40. wwww 35 0 82 0 45 0
-
The comparative balance sheet of Beets Industries, Inc. at December 31, 2013 and 2012, is as follows: An examination of the income statement and the accounting records revealed the following...
-
Gasper Company has transactions with companies in many countries. It purchases components from companies in Korea and several European countries and sells its products throughout the world. The CEO...
-
For Example 17.4 in the text, dealing with a single-station assembly system, suppose that the sequence of assembly elements were to be accomplished on a seven-station assembly system with synchronous...
-
Find the inverse Laplace transform in two different ways: (i) Use tables. (ii) Use the Bromwich Integral. a. \(F(s)=\frac{1}{s^{3}(s+4)^{2}}\). b. \(F(s)=\frac{1}{s^{2}-4 s-5}\). c....
-
The Colson Company issued $300,000 of 10% bonds on January 1, 2011. The bonds are due January 1, 2016, with interest payable each July 1 and January 1. The bonds are issued at face value. Prepare...
-
slove plsslove for yellow boxs On January 1: 2024, the Mountain Company agreed to purchase a building by making six payments. The lirst three are to be \( \$ 38,000 \) each, and will be paid on...
-
TV Trends to Watch in 2021 In 2021, expect larger and cheaper 4K TVs, a new level of voice control, interaction with other smart products, and mini Led backlights to improve color contrast and...
-
Internet UDP (User Datagram Protocol) packets conform to the format given below. The source and destination port numbers are 16 bit numbers; the Length field is also a 16 bit number that contains the...
-
You approve budget allocations for projects. Marie, who is very competent in developing budgets, has come to you. You: a. Review the budget, make revisions, and explain them in a supportive way. Deal...
-
In what phase of an audit do you evaluate internal controls? Why do you evaluate internal controls during this phase?
-
As a time-and-motion expert, you have been called in regards to a complaint about the standard time it takes to perform a job. As you analyze the entire job, you realize one element should take...
-
Briefly describe two types of evidence collection procedures you might use to obtain an understanding of internal controls.
-
Compare and contrast the drivers and the role of the MRP and the DRP systems.
-
Case Study 15.1: Organizational Change at Amazon Amazon has reinvented itself again and again and thrived on organizational change. Since the company went online in 1995, the e-commerce giant has...
-
A sample statistic will not change from sample to sample. Determine whether the statement is true or false. If it is false, rewrite it as a true statement.
-
Draw short segments of the polymers obtained from the following monomers: a.CH2=CHF b. CH2=CHCO2H c. d. e. HOCH2sCOH NCOHOCH CH OH OCN
-
Give the major product obtained from reaction of each of the following with excess HCl: a. CH3CH2C==CH b. CH3CH2C===CCH2CH3 c. CH3CH2C==CCH2CH2CH3
-
The mechanism for hydroxide-ion-promoted ester hydrolysis is shown in Section 17.12. What catalytic role does hydroxide ion play in this mechanism?
-
For \(a>0\), find the Fourier transform, \(\hat{f}(k)\), of \(f(x)=e^{-a|x|}\).
-
For the case that a function has multiple roots, \(f\left(x_{i} ight)=0, i=1,2, \ldots\), it can be shown that \[\delta(f(x))=\sum_{i=1}^{n} \frac{\delta\left(x-x_{i}...
-
Find a Fourier series representation of the Dirac delta function, \(\delta(x)\), on \([-L, L]\).

Study smarter with the SolutionInn App



