Consider the matrix 2 -3 -1 3 4 A = 1 -2 0 -1 1 -3...
Fantastic news! We've Found the answer you've been seeking!
Question:
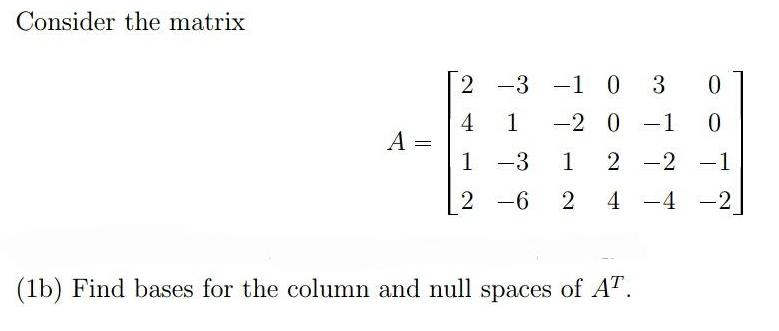
Transcribed Image Text:
Consider the matrix 2 -3 -1 3 4 A = 1 -2 0 -1 1 -3 1 2 -2 -1 2 -6 2 4 -4 -2 | (1b) Find bases for the column and null spaces of AT. Consider the matrix 2 -3 -1 3 4 A = 1 -2 0 -1 1 -3 1 2 -2 -1 2 -6 2 4 -4 -2 | (1b) Find bases for the column and null spaces of AT.
Expert Answer:
Posted Date:
Students also viewed these mathematics questions
-
Find bases for the column space of A and the null space of A T for the given exercise. Verify that every vector in col(A) is orthogonal to every vector in null(A T ). Exercise 8 Data from Exercise 8...
-
Consider the matrix 2 3 A = 1 8 (a) Show that the matrix A is invertible, and find A1 (b) Find elementary matrices, E1, E2, E3 and E, so that I, = E, E, E, E, A, where I, is the identity matrix of...
-
Below is the trial balance of Conex Inc. at December 31, 2021 Conex Inc. Trial Balance December 31, 2021 Account Cash Accounts Receivable Prepaid Insurance Supplies Equipment Accumulated...
-
Prove that the curves y 2 = 4x and x 2 = 4y divide the by of the squre bounded by x = 0, x = 4, y = 4 and y = 0 into three equal parts.
-
Consider two 2-curves with degrees of freedom 12 and 20, respectively. Which one more closely resembles a normal curve? Explain your answer.
-
Bigstate's highway department must select projects to be built during the coming year. Eleven projects have passed the preliminary screening (see Table 47-1). Five projects have mutually exclusive...
-
A material processing oven has a first cost of $\$ 16,999$ with a life of 10 years and a salvage value of $\$ 2,500$. The corporate MARR is $12 \%$. If the system provides about $\$ 2,750$ annually,...
-
Four years ago Omega Technology, Inc., acquired a machine to use in its computer chip manufacturing operations at a cost of $35,000,000. The firm expected the machine to have a seven-year useful life...
-
Entries and Schedules for Unfinished Jobs and Completed Jobs Hildreth Company uses job order costing. The following data summarize the operations related to production for April, the first month of...
-
Chris and Sue are 50 percent shareholders in the BackBone personal service corporation. BackBone provides chiropractic services in four small towns: Troy, Union, Vista, and Willow. Chris is the main...
-
Hilda gives Irving a house worth $300,000 in which Hilda has a basis of $420,000. The house has been owned for 5 years. a) If Irving rents the house out what will his basis be for depreciation? b)...
-
The design of RISC-V provides for 32 general-purpose registers and 32 floating-point registers. If registers are good, are more registers better? List and discuss as many trade-offs as you can that...
-
Explain the divide-and-conquer strategy to get close to a bug in a debugger.
-
Write a program that prints a Christmas tree: /\ "1 11 1 " " Remember to use escape sequences.
-
Compute the effective CPI for RISC-V using Figure A.29 and the table above. Average the instruction frequencies of bzip and hmmer to obtain the instruction mix. You may assume that all other...
-
Write a program that reads a number between 1,000 and 999,999 from the user and prints it with a comma separating the thousands. Here is a sample dialog; the user input is in color: Please enter an...
-
Question 9 of 40 Fatima is a project manager. She lists the project tasks and places them in the correct sequence. To build the project schedule, what is her next step? Add contingency buffers to the...
-
Why can wastewater treatment requirements in Hawaii be less stringent than those in most locations on the U.S. mainland?
-
Figure P29.8 is an energylevel diagram for a simple atom. What wavelengths appear in the atom's (a) emission spectrum and (b) absorption spectrum? An electron with \(2.0 \mathrm{eV}\) of \(n=3...
-
In this hypothetical atom, what is the photon energy photon of the longest wavelength photons emitted by atoms in the 5p state? A. 1.0 eV B. 2.0eV C. 3.0 eV D. 4.0eV Energy (eV) S 5- 4 3- 2- 5s -5p...
-
What are the \(n\) and \(l\) values of the following states of a hydrogen atom: (a) \(4 d\), (b) \(5 f\), (c) \(6 s\) ?

Study smarter with the SolutionInn App


