17. Which of the following is a secondary alkyl halide? A) (CH)CHCHCI 18. Which of the...
Fantastic news! We've Found the answer you've been seeking!
Question:
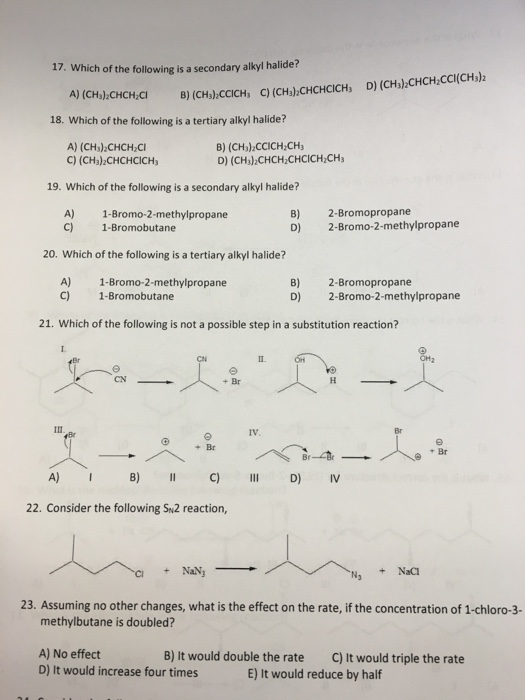
Transcribed Image Text:
17. Which of the following is a secondary alkyl halide? A) (CH₂)₂CHCH₂CI 18. Which of the following is a tertiary alkyl halide? A) (CH₂)₂CHCH₂Cl B) (CH₂)2CCICH₂CH₂ C) (CH₂)2CHCHCICH, D) 19. Which of the following is a secondary alkyl halide? 1-Bromo-2-methylpropane B) (CH₂):CCICH, C) (CH₂):CHCHCICH, D) (CH₂)2CHCH₂CCI(CH3)2 III A) C) 1-Bromobutane 20. Which of the following is a tertiary alkyl halide? A) B) 1-Bromo-2-methylpropane 1-Bromobutane C) D) 21. Which of the following is not a possible step in a substitution reaction? A) I e + Br B) II C) 22. Consider the following SN2 reaction, + NaN3 (CH₂)2CHCH₂CHCICH₂CH₂ + Br A) No effect D) It would increase four times II B) D) IV. 2-Bromopropane 2-Bromo-2-methylpropane OH 2-Bromopropane 2-Bromo-2-methylpropane H III D) IV + NaCl OH₂ Br 23. Assuming no other changes, what is the effect on the rate, if the concentration of 1-chloro-3- methylbutane is doubled? B) It would double the rate C) It would triple the rate E) It would reduce by half 17. Which of the following is a secondary alkyl halide? A) (CH₂)₂CHCH₂CI 18. Which of the following is a tertiary alkyl halide? A) (CH₂)₂CHCH₂Cl B) (CH₂)2CCICH₂CH₂ C) (CH₂)2CHCHCICH, D) 19. Which of the following is a secondary alkyl halide? 1-Bromo-2-methylpropane B) (CH₂):CCICH, C) (CH₂):CHCHCICH, D) (CH₂)2CHCH₂CCI(CH3)2 III A) C) 1-Bromobutane 20. Which of the following is a tertiary alkyl halide? A) B) 1-Bromo-2-methylpropane 1-Bromobutane C) D) 21. Which of the following is not a possible step in a substitution reaction? A) I e + Br B) II C) 22. Consider the following SN2 reaction, + NaN3 (CH₂)2CHCH₂CHCICH₂CH₂ + Br A) No effect D) It would increase four times II B) D) IV. 2-Bromopropane 2-Bromo-2-methylpropane OH 2-Bromopropane 2-Bromo-2-methylpropane H III D) IV + NaCl OH₂ Br 23. Assuming no other changes, what is the effect on the rate, if the concentration of 1-chloro-3- methylbutane is doubled? B) It would double the rate C) It would triple the rate E) It would reduce by half
Expert Answer:
Answer rating: 100% (QA)
The detailed answer for the above question is provided below ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Which of the following is a reason to obtain professional certification? a. Certification provides credibility that an individual is technically competent. b. Certification often is a necessary...
-
Which of the following is a specific audit procedure that would be completed in response to a particular fraud risk in an account balance or class of transactions? a. Exercising more professional...
-
Which of the following is a preventive control? a. Reconciliation of a bank account. b. Recalculation of a sample of payroll entries by internal auditors. c. Separation of duties between the payroll...
-
Repeat Prob. 37 using singularity functions exclusively (including reactions). Repeat Prob. 37, For the beam shown, find the reactions at the supports and plot the shear-force and bending-moment...
-
Differentiate between general and application controls. Give two examples of each.
-
In problem, solve each equation. log 5 x = 3
-
The adjusted trial balances of CAS, Inc., at March 31,2010, and March 31, 2011, include these amounts: Requirement 1. Compute the amount of supplies expense, salary expense, and service revenue CAS,...
-
GreenBeam Ltd. claims that its compact fluorescent bulbs average no more than 3.50 mg of mercury. A sample of 25 bulbs shows a mean of 3.59 mg of mercury. (a) Write the hypotheses for a right-tailed...
-
Cash Payback Period for a Service Company Jane's Clothing Inc. is evaluating two capital investment proposals for a retail outlet, each requiring an investment of $150,000 and each with an eight-year...
-
Required - Please submit the following two documents: 1. Using Excel, submit the following: i. ii. Calculate Rashmika's minimum net income for tax purposes in accordance with the ordering provisions...
-
Selected transactions of Duffy Inc. are listed below. 1. Common shares are sold for cash. 2. Bonds payable are issucd for cash at a discount. 3. Interest on a short-term note receivable is collected....
-
Air at 20 0 C and 80 KPa enters the diffuser of a jet engine steadily increases with a velocity of 140 m/s. The inlet area of the diffuser is 0.5 m 2 . What is the mass flowrate of the air? (note; R...
-
Find the effects of the "F1" force lifting the "G" load and the "F2" force occurring at "A" in the hydraulic system of a ship. (Bir gemiye ait hidrolik sistemde "G" ykn kaldran "Fi" kuvveti ve "A"da...
-
Hsu Company reported the following on its income statement: Line Item Description Amount Income before income tax expense $303,333 Income tax expense (91,000) Net income $212,333 What is the...
-
Sofia has an opportunity to invest $18,000 she inherited from her great aunt. She wants to know how long it would take to double her money. The average investment return over the past decade was 8%...
-
A corporation issues $273000, 12%, 5-year bonds on January 1, 2020, for $261600. Interest is paid annually on January 1. If the Corporation uses the straight-line method of amortization of bond...
-
Topic of "Flawless Consulting" stresses that the Engagement & Implementation portion of the Consulting Process is critical. However, it also emphasizes that implementing the Consultants...
-
Classify each of the following activities as proper or prohibited under the various consumer statutes you have studied. a. Calling a hospital room to talk to a debtor who is a patient there. b....
-
Chloramphenicol is a broad-spectrum antibiotic that is particularly useful against typhoid fever. What is the configuration of each asymmetric carbon in chloramphenicol? HO NHCCHCh NO, chloramphenicol
-
How could the following compounds be synthesized from an alkene? a. b. c. d. OCH2CH3 CH3CHCH2CH3 OCH2CH3 CH CH3CCH,CH3 OCH
-
Label each set of chemically equivalent protons, using a for the set that will be at the lowest frequency (farthest upfield) in the 1H NMR spectrum, b for the next, etc. Indicate the multiplicity of...
-
Why is communications management so important during project execution? What is the difference between formal and informal communications? Why are nonverbal communications so important?
-
What is involved in managing quality, and how does it affect project execution? What are tools and techniques used in performing quality assurance? How did Kristins team use one of these tools to...
-
Find an example of a large project that took more than a year to complete. Describe some of the planning work completed for the project as part of project integration and scope management. Summarize...

Study smarter with the SolutionInn App


