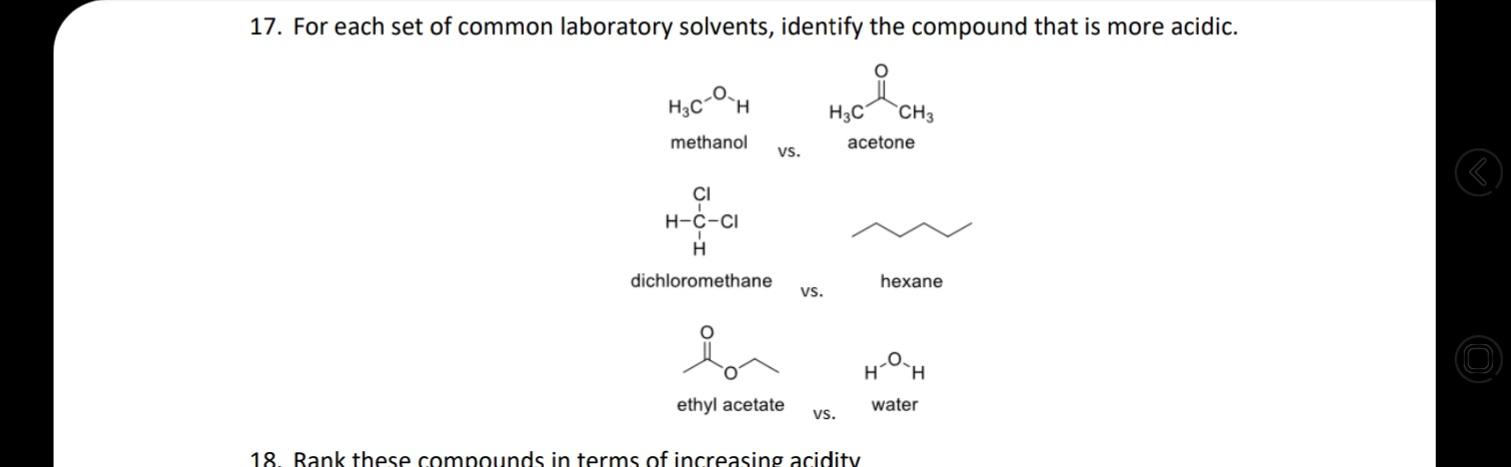
18. Rank these compounds in terms of increasing acidity HNO, H,SO4 HF H,CO3 . NH3 CH4...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
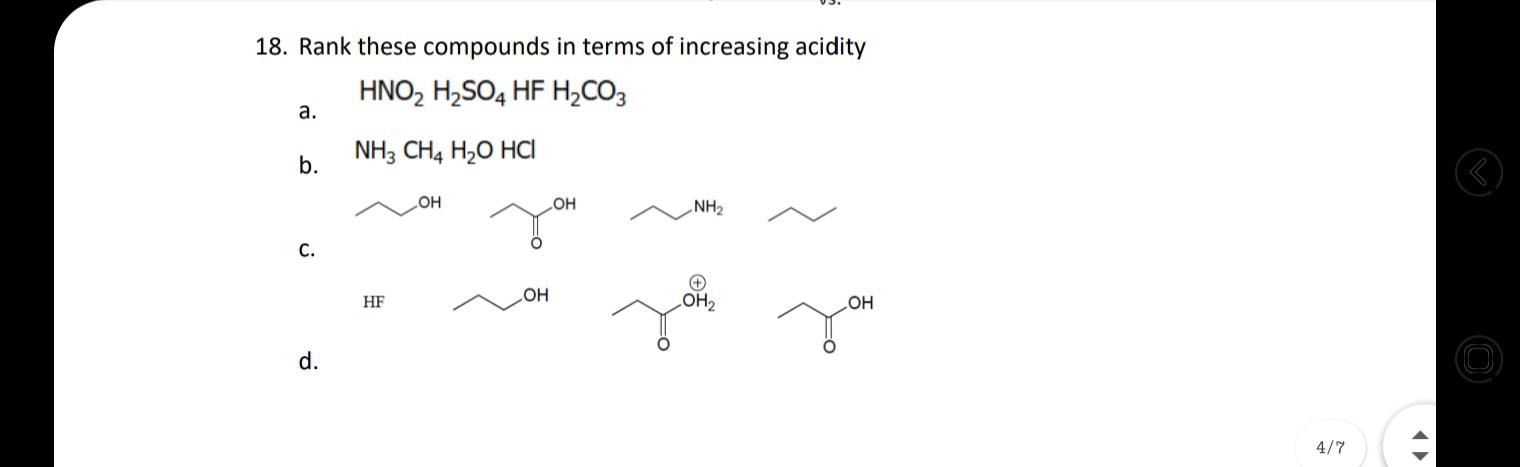
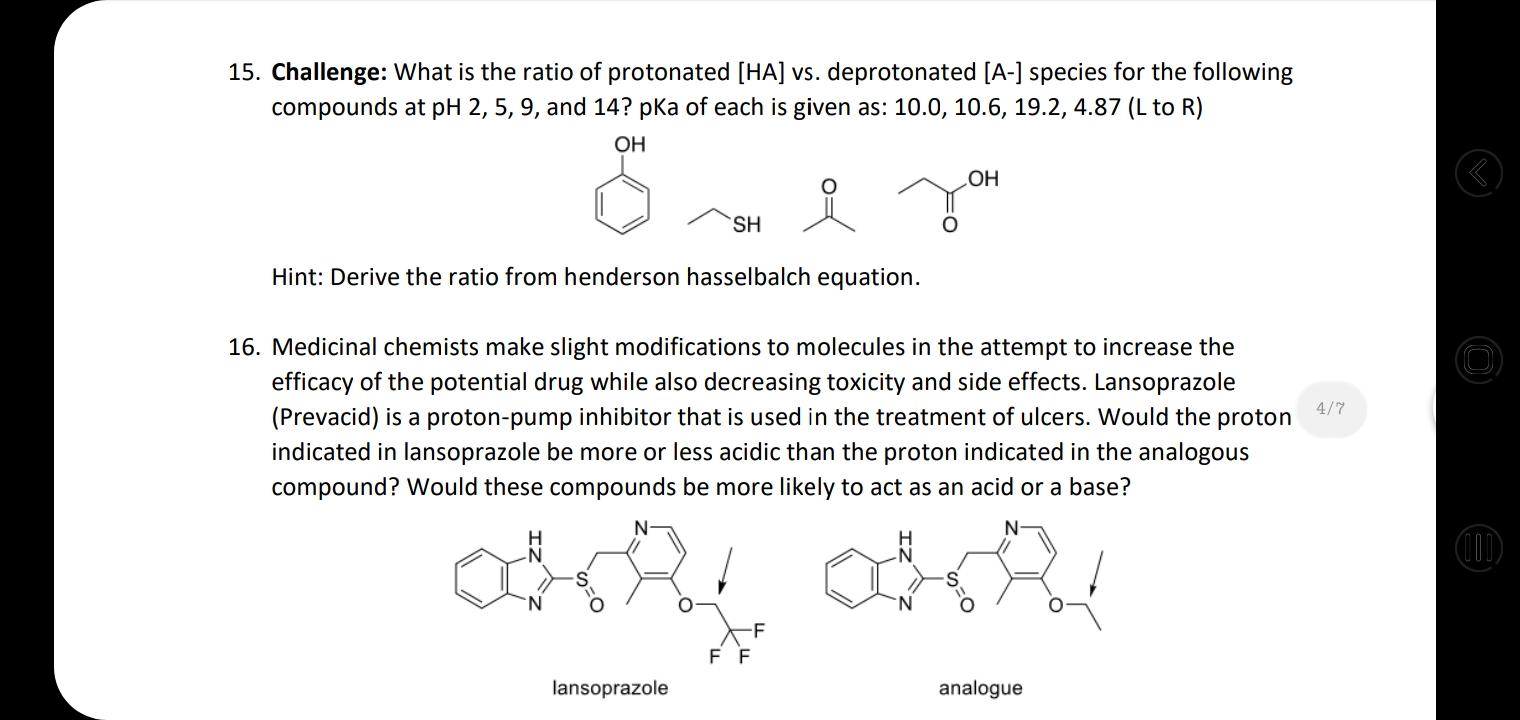
18. Rank these compounds in terms of increasing acidity HNO, H,SO4 HF H,CO3 а. NH3 CH4 H2O HCI b. OH NH2 HO С. HF LOH2 d. 4/7 17. For each set of common laboratory solvents, identify the compound that is more acidic. H3C `CH3 methanol acetone vs. CI H-C-CI H dichloromethane hexane vs. H ethyl acetate water vs. 18. Rank these compounds in terms of increasing acidity 15. Challenge: What is the ratio of protonated [HA] vs. deprotonated [A-] species for the following compounds at pH 2, 5, 9, and 14? pka of each is given as: 10.0, 10.6, 19.2, 4.87 (L to R) OH HO SH Hint: Derive the ratio from henderson hasselbalch equation. 16. Medicinal chemists make slight modifications to molecules in the attempt to increase the | O efficacy of the potential drug while also decreasing toxicity and side effects. Lansoprazole 4/7 (Prevacid) is a proton-pump inhibitor that is used in the treatment of ulcers. Would the proton indicated in lansoprazole be more or less acidic than the proton indicated in the analogous compound? Would these compounds be more likely to act as an acid or a base? (1) -F F F lansoprazole analogue 18. Rank these compounds in terms of increasing acidity HNO, H,SO4 HF H,CO3 а. NH3 CH4 H2O HCI b. OH NH2 HO С. HF LOH2 d. 4/7 17. For each set of common laboratory solvents, identify the compound that is more acidic. H3C `CH3 methanol acetone vs. CI H-C-CI H dichloromethane hexane vs. H ethyl acetate water vs. 18. Rank these compounds in terms of increasing acidity 15. Challenge: What is the ratio of protonated [HA] vs. deprotonated [A-] species for the following compounds at pH 2, 5, 9, and 14? pka of each is given as: 10.0, 10.6, 19.2, 4.87 (L to R) OH HO SH Hint: Derive the ratio from henderson hasselbalch equation. 16. Medicinal chemists make slight modifications to molecules in the attempt to increase the | O efficacy of the potential drug while also decreasing toxicity and side effects. Lansoprazole 4/7 (Prevacid) is a proton-pump inhibitor that is used in the treatment of ulcers. Would the proton indicated in lansoprazole be more or less acidic than the proton indicated in the analogous compound? Would these compounds be more likely to act as an acid or a base? (1) -F F F lansoprazole analogue
Expert Answer:

Related Book For 

Conceptual Physical Science
ISBN: 978-0134060491
6th edition
Authors: Paul G. Hewitt, John A. Suchocki, Leslie A. Hewitt
Posted Date:
Students also viewed these chemistry questions
-
For eight attributes rank-ordered in terms of increasing importance, the weighting of the sixth attribute is closest to: (a) 0.17 (b) 0.14 (c) 0.08 (d) 0.03
-
Rank the three carbocations shown in terms of increasing stability: a. b.
-
Rank the indicated bonds in terms of increasing bond length: . 1 =C .
-
A bank reconciliation takes time and must balance. An employee was struggling in balancing the bank reconciliation. Her supervisor told her to plug (make an unsupported entry for) the difference,...
-
Identify what is being oxidized and reduced in this redox equation by assigning oxidation numbers to the atoms. 2K + MgCl2 ( 2KCl + Mg
-
Sylvia has worked as a waitress at an upscale restaurant for several years. A new general manager was hired six months ago and has acted inappropriately with Sylvia. Sylvia sued and won...
-
Consider the hemodialysis device in Example 24.1. Examine the effect on the rate of urea removal of changing the hemodialyzer geometry, the blood and dialysate flow rates to the dialyzer, the rate of...
-
On December 31, 2016, Vail Company owned the following assets: Vail computes depreciation and amortization expense to the nearest whole year. During 2017, Vail engaged in the following transactions:...
-
Yellow Sticker Company's variable expenses are 40% of sales. The company has monthly fixed expenses of $15,000 and sells each unit for $0.50. The monthly target operating income is $7,500. a. What is...
-
EYE SPY sells sophisticated video surveillance equipment. EYE SPY sells the equipment and computer integration services together. It does not sell these separately. The equipment cannot operate...
-
A PC engineer wires electrical designing and software engineering to foster new innovation. PC engineers configuration, assemble, and keep up with equipment in present day PCs. These designers center...
-
Is it lawful to use force to resist an arrest? Under what circumstances?
-
What is the castle doctrine? How is it relevant in the self-defense context?
-
Compare and contrast treason, sedition, and sabotage.
-
How does failure of proof differ from justification and excuse defenses?
-
Define criminal syndicalism. How is it similar to other offenses against the state?
-
The following table shows the production function for a firm. Number of workers Average productivity 1 6 2 8 3 19 4 9.25 19.30 9.32 5 6 Which worker represents the first decline in marginal...
-
Carlton Stokes owns and operates a car-detailing business named SuperShine & Detailing. For $150, Carltons business will hand wash and wax customers cars, vacuum the interior, and thoroughly clean...
-
What isotope is commonly used for dating sediments or organic material from the late Pleistocene?
-
What is a dipole?
-
What two minerals make up most of the sand in the world?
-
The equity section of Atrio Ltd. showed the following: share premium 6,101, share capitalordinary 925, share capitalpreference 58, retained earnings 7,420, and treasury shares 2,828. (All amounts are...
-
The following equity accounts are in the ledger of Eudaley Group at December 31, 2025. Instructions Prepare the equity section of the statement of financial position at December 31, 2025. Share...
-
Travis Mordica asks, Since share dividends dont change anything, why declare them? What is your answer to Travis?

Study smarter with the SolutionInn App