1ONA(s) + 2KNO, (s) K,0(3) + 5Na,0(s) +N, (g) A common laboratory-scale reaction can also...
Fantastic news! We've Found the answer you've been seeking!
Question:
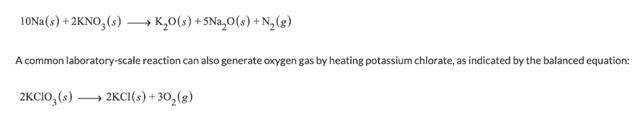
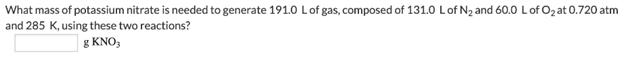

Transcribed Image Text:
1ONA(s) + 2KNO, (s) – K,0(3) + 5Na,0(s) +N, (g) A common laboratory-scale reaction can also generate oxygen gas by heating potassium chlorate, as indicated by the balanced equation: 2KCIO,() → 2KCI(s) + 30,(g) What mass of potassium nitrate is needed to generate 191.0 Lof gas, composed of 131.0 Lof N2 and 60.0 Lof O2 at 0.720 atm and 285 K, using these two reactions? g KNO, What mass of potassium chlorate is needed to generate 191.0 L of gas, composed of 131.0 Lof N2 and 60.0 Lof O2 at 0.720 atm and 285 K, using these two reactions? 8 KCIO, 1ONA(s) + 2KNO, (s) – K,0(3) + 5Na,0(s) +N, (g) A common laboratory-scale reaction can also generate oxygen gas by heating potassium chlorate, as indicated by the balanced equation: 2KCIO,() → 2KCI(s) + 30,(g) What mass of potassium nitrate is needed to generate 191.0 Lof gas, composed of 131.0 Lof N2 and 60.0 Lof O2 at 0.720 atm and 285 K, using these two reactions? g KNO, What mass of potassium chlorate is needed to generate 191.0 L of gas, composed of 131.0 Lof N2 and 60.0 Lof O2 at 0.720 atm and 285 K, using these two reactions? 8 KCIO,
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these accounting questions
-
Oxygen can be prepared by heating potassium chlorate, KClO3, with manganese dioxide as a catalyst. The reaction is How many moles of O2 would be obtained from 1.300 g KClO3? If this amount of O2 were...
-
The reaction N2 + O2 2NO occurs in internal combustion engines. Determine the equilibrium mole fraction of NO when the pressure is 101 kPa and the temperature is 1800 K.
-
A quantity of N2 gas originally held at 5.25 atm pressure in a 1.00-L container at 26oC is transferred to a 12.5-L container at 20oC .A quantity of O2 gas originally at 5.25 atm and 26oC in a 5.00-L...
-
For rigid-body motion, the strains will vanish. Under these conditions, integrate the straindisplacement relations (7.6.1) to show that the most general form of a rigid-body motion displacement field...
-
Multiple Choice Questions: 1. Which of the following issues is not dealt with in The United Nations Convention against Transnational Organized Crime and its Protocols? a. Money laundering. b....
-
Locate the centroid x of the area. 4 m y 4 m X
-
Charlie and Larry both face the same budget line for consumption and leisure. At every possible consumption-leisure bundle on the budget line, Charlie always requires marginally more leisure than...
-
This problem is a continuation of P5-35. Mortar Corporation acquired 80 percent ownership of Granite Company on January 1, 20X7, for $173,000. At that date, the fair value of the noncontrolling...
-
A person who proves that at the time of the commission of the offense, that the person as a result of a severe mental disease or defect did not know the wrongfulness of his actions is?
-
A beam ABCD with a vertical arm CE is supported as a simple beam at A and D (see figure). A cable passes over a small pulley that is attached to the arm at E. One end of the cable is attached to the...
-
Set up (but do not simplify) a line integral which gives the work done by the force field F = (x, ye*) in moving an object on the curve r(t) = (+1, t) for Osts 1. A. OB. 1 O C. = S(( + 1), tel + 1)....
-
Draw an AVL tree whose elements are the letters AZ. Label each node with its balance factor. Draw a second non-AVL tree whose elements are from AZ. Label each node with its balance factor and mark...
-
The radix sort algorithm uses an array of queues. Would an array of stacks work just as well?
-
Finish designing the algorithms for the explicit set representation.
-
We said that HeapSort is inherently unstable. Explain why.
-
Add the elements 7 and 8 to the balanced AVL tree in Figure 10.2e. Is the tree still balanced? If not, identify the unbalanced node and which rotation operation should be applied. Redraw the tree,...
-
A rectangular airstrip measures 32.30 m by 210 m, with the width measured more accurately than the length. Find the area, taking into account significant figures. (Select all that apply.) 6.783 x 103...
-
True & False The basis of an asset must be reduced by the depreciation allowable, 2. Adjusted gross income (AGI) is the basis for a number of phase-outs of deductions. 3. A change to adjusted gross...
-
There are 2.619 1022 atoms in 1.000 g of sodium. Assume that sodium atoms are spheres of radius 1.86 and that they are lined up side by side. How many miles in length is the line of sodium atoms?
-
A solution is 0.35 M Sr(OH)2. What are the concentrations of H3O+ and OH in this solution?
-
Two compounds have the same composition: 85.62% C and 14.38% H. a. Obtain the empirical formula corresponding to this composition. b. One of the compounds has a molecular mass of 28.03 amu; the...
-
Describe which characteristics of HR metrics and workforce analytics are likely to result in greater return on investment and organizational impact.
-
Why are information security and privacy important considerations in the design, development, and maintenance of an HRIS?
-
List and discuss the major information security and privacy threats to organizations.

Study smarter with the SolutionInn App



