When alkaline phosphatase is added to substrate in borate buffer (pH 9.5) and allowed to sit...
Fantastic news! We've Found the answer you've been seeking!
Question:
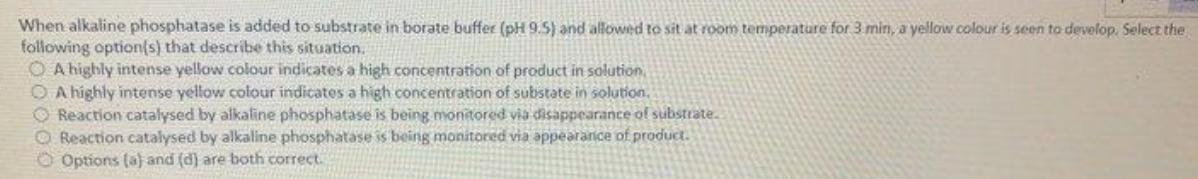
Transcribed Image Text:
When alkaline phosphatase is added to substrate in borate buffer (pH 9.5) and allowed to sit at room temperature for 3 min, a yellow colour is seen to develop. Select the following option(s) that describe this situation. O A highly intense yellow colour indicates a high concentration of product in solution O A highly intense yellow colour indicates a high concentration of substate in solution. O Reaction catalysed by alkaline phosphatase is being monitored via disappearance of substrate. O Reaction catalysed by alkaline phosphatase is being monitored via appearanice of product. O Options (a) and (d) are both correct. When alkaline phosphatase is added to substrate in borate buffer (pH 9.5) and allowed to sit at room temperature for 3 min, a yellow colour is seen to develop. Select the following option(s) that describe this situation. O A highly intense yellow colour indicates a high concentration of product in solution O A highly intense yellow colour indicates a high concentration of substate in solution. O Reaction catalysed by alkaline phosphatase is being monitored via disappearance of substrate. O Reaction catalysed by alkaline phosphatase is being monitored via appearanice of product. O Options (a) and (d) are both correct.
Expert Answer:
Answer rating: 100% (QA)
In the reaction the ALP catalyzes the hydrolysis of the colorless organic phosphate ester substrate pNitrophenylphosphate to the yellow colored produc... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
When alkaline phosphatase is added to substrate in borate buffer (pH 9.5) and allowed to sit at room temperature for 3 min, a yellow colour is seen to develop. Select the following option(s) that...
-
Calculate the Fermi Energy & Conductivity at room temperature for germanium containing 5x10^6 aresenic atoms/cm^3. Use the mobility of the electrons in the host materials. The textbook answers...
-
Calculate dp/dT at room temperature for(a) Copper and(b) Silicon, using data from Table41-1. CH H - CH H H* H. Testosterone
-
Data for Video Phones, Inc., are provided in P114A. Required: Prepare the statement of cash flows for Video Phones, Inc., using the direct method. Disclose any noncash transactions in an accompanying...
-
Chris has an income of $90 to allocate between Goods A and B. Initially, the price of A is $3 and the price of B is $4. a. Draw Chriss budget line, indicating its slope if units of A are measured on...
-
The Air Transport Association of America publishes figures on the busiest airports in the United States. The following frequency distribution has been constructed from these figures for a recent...
-
How many forms of main() are there?
-
On January 1, Zabel Corporation purchased a 25% equity in Helbert Corporation for $180,000. At December 31, Helbert declared and paid a $60,000 cash dividend and reported net income of $200,000....
-
In its simplest form, profit is: Fixed costs - Depreciation - Interest = Profit Sales/Turnover - Variable costs/Used goods = Profit (Actual retail price - cost)/ Actual retail price = Profit (Price -...
-
Consider the following scenario in the hypothetical country of Zootopia: The government budget deficit is $80 billion, total domestic savings are $1.2 trillion, and total domestic investment in...
-
Average Joe's Eller classmate Richie Rich brags to Joe that he has earned an annual return of 60% investing in dogecoin, polka dot, and other cryptocurrencies. If Joe saves $750 per month and somehow...
-
What is the power required to accelerate a 10 tons (metric tons) truck from rest to 36 kmh1 in 10 s, on a road level?
-
In Civil Litigation in Kenya what is a Third party proceeding ?
-
The angle between the lockers/wall/elevated surface and the floor is assumed to be a perfect 90 degrees. Explain why?
-
A rock is thrown at a 60.0 angle above the horizontal across level ground. It is thrown from a height of 2.00 m above the ground with a speed of 20.0 m/s and experiences no appreciable air...
-
After completing the process of recording the purchase of supplies, you noticed that two account balances are incorrect in your trial balance related to that transaction. What accounts (by type) are...
-
hey can ypls answerhttps://chegg-study.app.link/?%24randomized_bundle_token=1178706551087942599&feature=share_qna&campaign=share_qna&type=0&duration=0&source=android&data=eyIk 1 answer
-
Using Gauss-Jordan elimination, invert this matrix ONLY 0 0 0 0 1
-
For each of the ternary systems shown in figure, indicate whether: (a) Simple, countercurrent extraction, or (b) Countercurrent extraction with extract reflux, or (c) Countercurrent extraction with...
-
Naphtali and Sandholm group the N(2C + 1) equations by stage. Instead, group the equations by type (i.e., enthalpy balances, component balances, and equilibrium relations). Using a three-component,...
-
A stripper operating at 50 psia with three equilibrium stages is used to strip 1,000 kmol/h of liquid at 250F having the following molar composition: 0.03% C1, 0.22% C2, 1.82% C3, 4.47% nC4, 8.59%...
-
How many structural isomers are there for hydrocarbons that have the molecular formula C 4 H 10 ? (a) none (b) one (c) two (d) three
-
Which contains more hydrogen atomsa five-carbon saturated hydrocarbon molecule or a five-carbon unsaturated hydrocarbon molecule? (a) The unsaturated hydrocarbon has more hydrogen atoms. (b) The...
-
Explain why caprylic acid, CH 3 (CH 2 ) 6 COOH, dissolves in a 5, aqueous solution of sodium hydroxide but caprylaldehyde, CH 3 (CH 2 ) 6 CHO, does not dissolve. (a) With two oxygens, the caprylic...

Study smarter with the SolutionInn App


