2. A fuel analyzing 27% CO, 12% CO2, 2% H2, 5% CH4 and 54% N and...
Fantastic news! We've Found the answer you've been seeking!
Question:
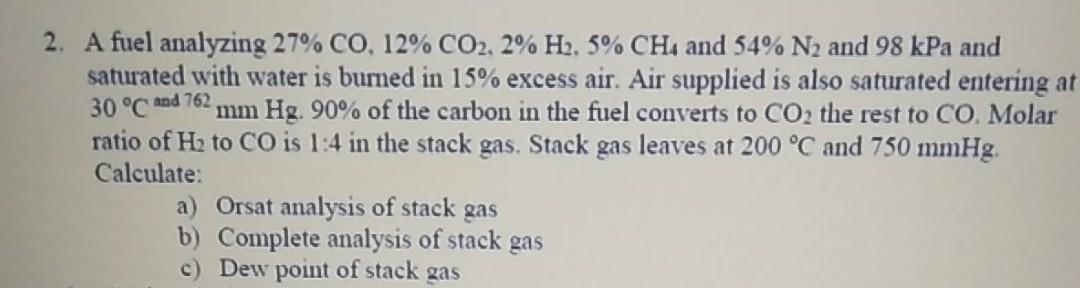
Transcribed Image Text:
2. A fuel analyzing 27% CO, 12% CO2, 2% H2, 5% CH4 and 54% N₂ and 98 kPa and saturated with water is burned in 15% excess air. Air supplied is also saturated entering at 30 °C and 762, mm Hg. 90% of the carbon in the fuel converts to CO2 the rest to CO. Molar ratio of H₂ to CO is 1:4 in the stack gas. Stack gas leaves at 200 °C and 750 mmHg. Calculate: a) Orsat analysis of stack gas b) Complete analysis of stack gas c) Dew point of stack gas 2. A fuel analyzing 27% CO, 12% CO2, 2% H2, 5% CH4 and 54% N₂ and 98 kPa and saturated with water is burned in 15% excess air. Air supplied is also saturated entering at 30 °C and 762, mm Hg. 90% of the carbon in the fuel converts to CO2 the rest to CO. Molar ratio of H₂ to CO is 1:4 in the stack gas. Stack gas leaves at 200 °C and 750 mmHg. Calculate: a) Orsat analysis of stack gas b) Complete analysis of stack gas c) Dew point of stack gas
Expert Answer:

Related Book For 

Income Tax Fundamentals 2013
ISBN: 9781285586618
31st Edition
Authors: Gerald E. Whittenburg, Martha Altus Buller, Steven L Gill
Posted Date:
Students also viewed these chemical engineering questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
1. A 10 m3 tank contains steam at 275C and 15.0 bar. The tank and its contents are cooled until the pressure drops to 1.8 bar. Some of the steam condenses in the process. a. How much heat was...
-
What is the common name for the following structure? CH3 CH3 -C-Br 1 CH3 Isobutyl bromide Osec-Butyl bromide O Neobutyl bromide O Isopropyl methyl bromide t-Butyl bromide
-
Using the Internet and whatever available sources, answer the following: What was the choice of Yelp? What is the current number of reviewers at Yelp? What are the number of counties they operate on,...
-
Briefly explain how the dividend decision is made. What corporate and market factors are important in deciding whether, and in what amount, to pay dividends?
-
As sales manager, you realize your salespeople need to be more efficient and effective in managing their time and territory. As you deliver the opening comments at an all-day seminar on time and...
-
From the data 1,4, 5, 3, 2, 5, 7, 3, 4, and 5, Poindexter created the following frequency table. What five things did he do wrong? f- 013589 f- 11223|-| 123457
-
Image transcription text 22 21.9615 0.6588 4.0606 0.3248 0.4648 35.1371 0./891 16 15.7412 0.4739 2.6706 0.3107 0.4507 35.1685 0.0258 10 9.9197 0.2982 1.4440 0.2890 0.4291 35.2908 0.0166 6 6.6168...
-
Make a list of assumptions that will be used as the basis for planning the wedding. And no, it is not acceptable to assume that Tony and Peggy Sue will just elope, no matter how tempting that may be!...
-
Give reason why Iron should be protected from moist air.
-
This information is for Java Growers for the year ended December 31, 2025 (amounts in thousands). Instructions Prepare the 2025 statement of cash flows for Java Growers. Identify cash flow...
-
What was the change in Globals book value of equity from 2021 to 2022 according to Table 2.1? Does this imply that the market price of Globals shares increased in 2022? Explain. TABLE 2.1 Liabilities...
-
The following quote on Tesla stock appeared on April 8, 2022, on Yahoo! Finance. If you wanted to buy Tesla, what price would you pay per share? How much would you receive per share if you wanted to...
-
Radiocarbon dating assumes that the abundance of \({ }^{14} \mathrm{C}\) in the environment has been constant. Suppose \({ }^{14} \mathrm{C}\) was less abundant 10,000 years ago than it is today....
-
Which of the following are valid PDFs? Justify your answer. a. \(f(x)=(.2)^{x}(.6)^{1-x} I_{\{0,1\}}(x)\) b. \(f(x)=(.3)(.7)^{x} I_{\{0,1,2, \ldots\}}\{x\}\) c. \(f(x)=.6 e^{-x / 4} I_{(0,...
-
Interpersonal Communication Approaches to Handling Conflict (CH 9) and Cultural Influences on Communication (CH 5) Paragraph 1-5: Describe & cite each of the Approaches to Handling Conflict (also...
-
An Atomic Energy Commission nuclear facility was established in Hanford, Washington, in 1943. Over the years, a significant amount of strontium 90 and cesium 137 leaked into the Columbia River. In a...
-
For each of the following situations, indicate whether the taxpayer(s) is (are) required to file a tax return for 2012. Explain your answer. a. Helen is a single taxpayer with interest income in 2012...
-
Quince Corporation has taxable income of $450,000 for its 2012 calendar tax year. Calculate the corporation's income tax liability for 2012 before tax credits. $_________
-
Yolanda earns $112,000 in 2012. Calculate the FICA tax that must be paid by: Yolanda:.....................Soc.Sec..................$__________...
-
Initially, both legs of a mercury manometer are open to the atmosphere, and the mercury height in the left leg is \(40.0 \mathrm{~mm}\). You inflate a balloon until it has a surface area of \(0.300...
-
At an atmospheric pressure of \(1.00 \mathrm{~atm}\), the height of the liquid in the tube of a mercury barometer is \(760 \mathrm{~mm}\) only if you ignore capillary rise in the tube. Obtain an...
-
A hydrogen-filled balloon is used to lift a \(125-\mathrm{kg}\) stone off the ground. The basket holding the stone has a mass of \(15.0 \mathrm{~kg}\). What must the minimum radius \(R\) of the...

Study smarter with the SolutionInn App


