Lab 5: Equilibrium Data Standard Solutions Molar concentration of Fe(NO3)3 0.200 M Molar concentration of NaSCN...
Fantastic news! We've Found the answer you've been seeking!
Question:
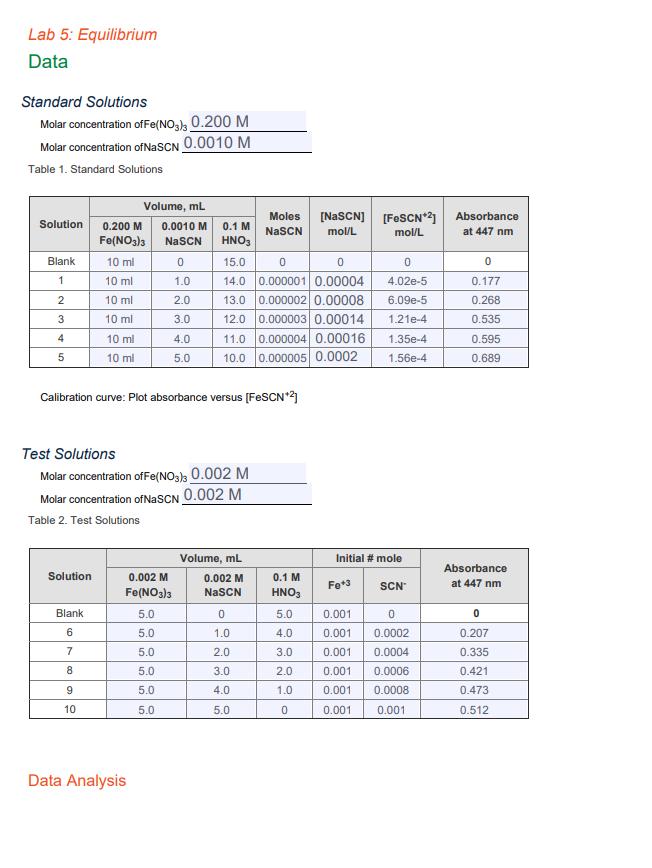
![Table 3: Equilibrium Conditions Using theequation ( K_{e q}=frac{left[mathrm{FeSCN}^{+2}ight]}{left[F e^{+3} mathrm{I](https://dsd5zvtm8ll6.cloudfront.net/si.experts.images/questions/2022/08/62e9062cb9e05_1659438634771.jpg)
Transcribed Image Text:
Lab 5: Equilibrium Data Standard Solutions Molar concentration of Fe(NO3)3 0.200 M Molar concentration of NaSCN 0.0010 M Table 1. Standard Solutions Solution 0.200 M Fe(NO3)3 10 ml 10 ml 10 ml 10 ml Blank 1 2 3 4 5 Solution Blank 6 10 ml 10 ml 7 8 Volume, mL 0.0010 M NaSCN 0 1.0 2.0 3.0 4.0 5.0 9 10 Calibration curve: Plot absorbance versus [FeSCN*2] Test Solutions Molar concentration of Fe(NO3)3 0.002 M Molar concentration of NaSCN 0.002 M Table 2. Test Solutions 0.1 M HNO3 0.002 M Fe(NO3)3 5.0 5.0 5.0 5.0 5.0 5.0 Data Analysis 15.0 0 0 14.0 0.000001 0.00004 13.0 0.000002 0.00008 12.0 0.000003 0.00014 Moles NaSC 11.0 0.000004 0.00016 1.35e-4 10.0 0.000005 0.0002 1.56e-4 Volume, mL 0.002 M NaSCN 0 1.0 2.0 3.0 4.0 5.0 [NaSCN] [FeSCN*2] Absorbance mol/L mol/L at 447 nm 0.1 M HNO3 5.0 4.0 3.0 2.0 1.0 0 0 4.02e-5 6.09e-5 1.21e-4 Initial # mole Fe+3 0.001 0.001 0.001 0.001 0.001 0.001 SCN 0 0.0002 0.0004 0.0006 0.0008 0.001 0 0.177 0.268 0.535 0.595 0.689 Absorbance at 447 nm 0 0.207 0.335 0.421 0.473 0.512 Table 3: Equilibrium Conditions Solution 6 7 8 9 10 Solution 6 7 8 9 10 [FeSCN¹21" mol/L [FeSCN*2]** mol/L Iron Equilibrium Moles Moles Fe*3 FeSCN¹2 reacted Using the equation K = Solution 6 Keq = Solution 7 Keq = Solution 8 Keq= Solution 9 Keq = Solution 10 Keq = Ave Keq = [FeSCN+2] eq [Fe +³[SCN] Moles Fe*3 unreacted [Fe*3] unreacted, mol/L Thiocyanate Equilibrium Moles Moles SCN Moles SCN [SCN] unreacted, SCN reacted unreacted mol/L calculate the equilibrium constant for solution 6 below: Lab 5: Equilibrium Data Standard Solutions Molar concentration of Fe(NO3)3 0.200 M Molar concentration of NaSCN 0.0010 M Table 1. Standard Solutions Solution 0.200 M Fe(NO3)3 10 ml 10 ml 10 ml 10 ml Blank 1 2 3 4 5 Solution Blank 6 10 ml 10 ml 7 8 Volume, mL 0.0010 M NaSCN 0 1.0 2.0 3.0 4.0 5.0 9 10 Calibration curve: Plot absorbance versus [FeSCN*2] Test Solutions Molar concentration of Fe(NO3)3 0.002 M Molar concentration of NaSCN 0.002 M Table 2. Test Solutions 0.1 M HNO3 0.002 M Fe(NO3)3 5.0 5.0 5.0 5.0 5.0 5.0 Data Analysis 15.0 0 0 14.0 0.000001 0.00004 13.0 0.000002 0.00008 12.0 0.000003 0.00014 Moles NaSC 11.0 0.000004 0.00016 1.35e-4 10.0 0.000005 0.0002 1.56e-4 Volume, mL 0.002 M NaSCN 0 1.0 2.0 3.0 4.0 5.0 [NaSCN] [FeSCN*2] Absorbance mol/L mol/L at 447 nm 0.1 M HNO3 5.0 4.0 3.0 2.0 1.0 0 0 4.02e-5 6.09e-5 1.21e-4 Initial # mole Fe+3 0.001 0.001 0.001 0.001 0.001 0.001 SCN 0 0.0002 0.0004 0.0006 0.0008 0.001 0 0.177 0.268 0.535 0.595 0.689 Absorbance at 447 nm 0 0.207 0.335 0.421 0.473 0.512 Table 3: Equilibrium Conditions Solution 6 7 8 9 10 Solution 6 7 8 9 10 [FeSCN¹21" mol/L [FeSCN*2]** mol/L Iron Equilibrium Moles Moles Fe*3 FeSCN¹2 reacted Using the equation K = Solution 6 Keq = Solution 7 Keq = Solution 8 Keq= Solution 9 Keq = Solution 10 Keq = Ave Keq = [FeSCN+2] eq [Fe +³[SCN] Moles Fe*3 unreacted [Fe*3] unreacted, mol/L Thiocyanate Equilibrium Moles Moles SCN Moles SCN [SCN] unreacted, SCN reacted unreacted mol/L calculate the equilibrium constant for solution 6 below:
Expert Answer:
Answer rating: 100% (QA)
Solution Since So Solution 1 2 345 02 M fet3 10 Solution 1 Nasen is ... View the full answer

Related Book For 

Financial Reporting and Analysis Using Financial Accounting Information
ISBN: 978-1439080603
12th Edition
Authors: Charles H Gibson
Posted Date:
Students also viewed these chemistry questions
-
J Meyers has the following budgeted figures for the year ended 30 June 2021: Units produced Machine-hours Direct labor-hours Manufacturing overheads Direct materials Direct wages 194 400 7 920 13 440...
-
General Long-term Liability and Capital Asset Transactions The Village of Nassau issued a 3-year, 6 percent note in the amount of $100,000 to finance the purchase of vehicles for the Public Works...
-
Capital Leases that have not been capitalized will decrease the times interest earned ratio. Comment.
-
Explain why each of the following statements about profit-maximizing competitive firms is incorrect. Restate each one correctly. a. A competitive firm will produce output up to the point where price...
-
A company manufactures tennis balls. When its tennis balls are dropped onto a concrete surface from a height of 100 inches, the company wants the mean height the balls bounce upward to be 55.5...
-
Westerfi eld contracted with Bradley Building Company for the construction of a ranch house as a wedding gift for his daughter. Before construction began, Westerfi eld died and Bradley announced that...
-
For a given x-value, the point estimate for a 95% confidence interval for the mean response is the same as the one for the 95% prediction interval. In Exercises 5 and 6, determine whether the...
-
Kitchen Electrics uses a perpetual inventory system. The following are three recent merchandising transactions: Apr. 10 Purchased 10 refrigerators from Polar Co. on account. Invoice price, $600 per...
-
A driver containing a main function that will allow the user to change their password. PasswordManager Class The PasswordManager class should have two member variables (both strings): a username an...
-
Chelsea Bush is an emerging candidate for her partys nomination for President of the United States. She now is considering whether to run in the high-stakes Super Tuesday primaries. If she enters the...
-
Read the following article: The Impact of Emerging Technologies on Knowledge Management in Organizations On the basis of your reading, answer the following questions: What are the objectives and main...
-
XYZ Corporation issued 20-year, noncallable, 4.5% annual coupon bonds at their par value of $1,000 one year ago. Today, the market interest rate on these bonds is 3.0%. a. What is the current price...
-
Thelma and Louise sit at rest in their 1966 Ford Thunderbird.They accelerate at a rate of 3.0 m/s/s for 9 seconds before drivingoff the edge of Dead Horse Point, 1900 feet above the bottom of...
-
After Robbie ran away from the accident scene, he was chased by the police and apprehended quickly. He agreed to a breathalyzer at the scene, which showed a result of .02. Robbie was charged with...
-
Explain in depth the following statement: - The relationship between project s objectives and project s parties provides insight into how uncertainty can be managed .
-
Calculate the average inventory (in $) and inventory turnover ratio for the given company. (Round the inventory turnover ratio to one decimal place.) Company Beginning Ending Inventory Inventory...
-
(a) The difference equation y[n] 0.75y[n-1]+ 0.125y[n-2]= x[n]. models an LTI system. Find the system transfer function. (b) Find the unit step response for the system of part (a). (e) Use MATLAB to...
-
The bookkeeper for Riley, Inc., made the following errors: a. A cash purchase of supplies of $357 was recorded as a debit to Supplies for $375 and a credit to Cash of $375. b. A cash sale of $3,154...
-
Indicate some sources that contain an appraisal of the outlook for particular industries.
-
The following material relates to Darrow Company: Required Place an X in the appropriate columns for each of thesituations. Cash Flows Classification Noncash Effect on Cash Operating Investing...
-
Hess Corporation* included the information in this case as part of the supplementary oil and gas data. This case only represents a small portion of the supplementary oil and gas data. Results of...
-
Museum directors today need both an advanced degree in art history and curatorial experience, as well as the financial acumen and managerial skills of a chief executive. In this economic environment,...
-
Discuss examples of how the changing economic condition has called for qualifi ed employees who are fl exible and willing to deal with rapid and unstructured changes. Give examples of impacts of...
-
Visit the US Department of Labor, Bureau of Labor Statistics web page and review Occupation Outlooks and Demographics Data. What additional workforce changes do you believe might take place in the...

Study smarter with the SolutionInn App


