2. Predict the solubility of the following substances in water, using Table 8.1. These substances are...
Fantastic news! We've Found the answer you've been seeking!
Question:
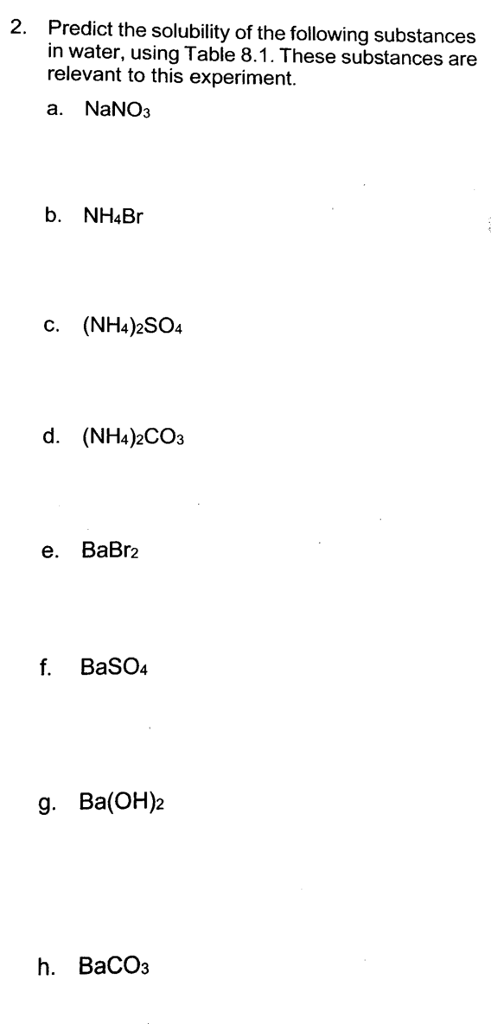

Transcribed Image Text:
2. Predict the solubility of the following substances in water, using Table 8.1. These substances are relevant to this experiment. a. NaNO3 b. NH4Br C. (NH4)2SO4 d. (NH4)2CO3 e. BaBr2 f. BaSO4 g. Ba(OH)2 h. BaCO3 Table 8.1: Solubility Rules of lonic Compounds Soluble lonic Compounds Compounds containing: Li+, Na+, K+, NH4+ NO3 CH³COO™, CIO4¯ CI, Br, I SO4²- Insoluble lonic Compounds Compounds containing: 0²-, CO3²-, PO4³- S²-, OH- Unless it contains No exception No exceptions Ag+, Cut, Hg₂+2, Pb+² Ba+2, Ca²+, Pb+², Sr+² Unless it contains NH4+, Li+, Na+, K+ NH4+, Li+, Na+, K+, Ca+2, Sr+2, Ba+² CH3COOH is an organic acid and it dissolved in water as whole - you should not separate it into CH3COO™ and H. 2. Predict the solubility of the following substances in water, using Table 8.1. These substances are relevant to this experiment. a. NaNO3 b. NH4Br C. (NH4)2SO4 d. (NH4)2CO3 e. BaBr2 f. BaSO4 g. Ba(OH)2 h. BaCO3 Table 8.1: Solubility Rules of lonic Compounds Soluble lonic Compounds Compounds containing: Li+, Na+, K+, NH4+ NO3 CH³COO™, CIO4¯ CI, Br, I SO4²- Insoluble lonic Compounds Compounds containing: 0²-, CO3²-, PO4³- S²-, OH- Unless it contains No exception No exceptions Ag+, Cut, Hg₂+2, Pb+² Ba+2, Ca²+, Pb+², Sr+² Unless it contains NH4+, Li+, Na+, K+ NH4+, Li+, Na+, K+, Ca+2, Sr+2, Ba+² CH3COOH is an organic acid and it dissolved in water as whole - you should not separate it into CH3COO™ and H.
Expert Answer:

Related Book For 

Income Tax Fundamentals 2013
ISBN: 9781285586618
31st Edition
Authors: Gerald E. Whittenburg, Martha Altus Buller, Steven L Gill
Posted Date:
Students also viewed these chemistry questions
-
What cultures are relevant to this project? Describe the project environment. MINI CASE Mr. Robert Rutland, founder of the National Jazz Hall of Fame, poured himself another drink as he listened to...
-
Using solubility guidelines, predict whether each of the following compounds is soluble or insoluble in water: (a) MgBr2 (b) PbI2 (c) (NH4)2CO3 (d) Sr(OH)2 (e) ZnSO4.
-
Evaluate the solubility of the following peptides at the given pH. For each pair, which peptide is more soluble and why? (a) pH 7: Gly20 and (Glu-Asp)10 (b) pH 5: (Cys-Ser-Ala)5 and (Pro-Ile-Leu)5...
-
"If nominal GDP rises, velocity must rise." Is this statement true, false, or uncertain? Explain your answer.
-
Andy Confer, production-line manager, had arranged a visit with Will Keating, plant manager. He had some questions about the new operational measures that were being used. Andy: Will, my questions...
-
a. Suppose that the number of workers doubles in Home. What happens to the Home PPF and what happens to the no-trade relative price of wheat? b. Suppose that there is technological progress in the...
-
2. Josephine Rutyna was a 60-year-old widow who suffered from high blood pressure and epilepsy. A bill collector from Collections Accounts Terminal, Inc., called her and demanded that she pay $56 she...
-
Goltra Clinic is considering investing in new heart-monitoring equipment. It has two options: Option A would have an initial lower cost but would require a significant expenditure for rebuilding...
-
ADCO sells a single product. 2,000 units were sold resulting in $1,000,000 of sales revenue, variable cost of $600,000. ontribution margin ratio of 40%, and fixed cost of 6,000. To achieve a target...
-
Draw a diagram similar to Figure 1.1 that places the following words in the proper relationships: theory, hypothesis, model, data, guess, and law. Figure 1.1 Geology Geochemistry (chemistry of the...
-
2. A stock's price is $100. Over each of the next two three month periods it is expected to go up by 15% or down by 10%. The risk free rate is 4% p.a. The stock pays a dividend of $1 per quarter....
-
Why do governments find it difficult to reduce spending?
-
What sort of assumptions underlie the policy that cuts in income taxes would increase the supply of labour hours offered in an economy?
-
Firms experience a rise in stocks. Explain why this might have occurred and what you expect firms response to this event might be and how this might affect output.
-
What are the potential costs and benefits of policies designed to improve the flexibility of labour markets?
-
What sort of trade-offs do governments face if a decision was being considered to reduce the level of welfare benefits as part of a market-orientated supply-side policy?
-
Case Study Cambridge Health Alliance set out to increase mammography rates among women 50 to 69 years of age. Screening rates for all women were below 60% when the initiative began. The initiative...
-
The following information is available for Partin Company: Sales $598,000 Sales Returns and Allowances 20,000 Cost of Goods Sold 398,000 Selling Expense 69,000 Administrative Expense 25,000 Interest...
-
Yolanda earns $112,000 in 2012. Calculate the FICA tax that must be paid by: Yolanda:.....................Soc.Sec..................$__________...
-
On February 2, 2012, Alexandra purchases a personal computer for her home. The computer cost $3,000. Alexandra uses the computer 80 percent of the time in her accounting business, 10 percent of the...
-
If Charles, a 16-year-old child model, earns $50,000 a year and is completely self supporting even though he lives with his parents, can his parents claim him as a dependent? Why or why not?...
-
Which statement is false with respect to the PCAOB? a. PCAOB issues standards describing auditor's attestation requirements. b. D.W. Squires was a former chief auditor of PCAOB. c. PCAOB is a...
-
Select five of these alleged fraudsters and prepare a two-paragraph discussion of them. Outline their modus operandi. 1. Frank Abagnale 2. Jack Abramoff 3. Kobi Alexander 4. Eddie Antar 5. Jim Bakker...
-
Which statement is false with respect to Section 404 of SOX? a. The section requires management's assessment of internal control over financial reporting. b. The auditor's report on internal control...

Study smarter with the SolutionInn App


