2. The compound OOOO 8 0000 CH3CHCHCHCHCHCHCH3 CH3CH-CH3 4-isopropyloctane 5-isopropyloctane 5-sec-butyloctane 4-propyloctane 3. Complete combustion of...
Fantastic news! We've Found the answer you've been seeking!
Question:

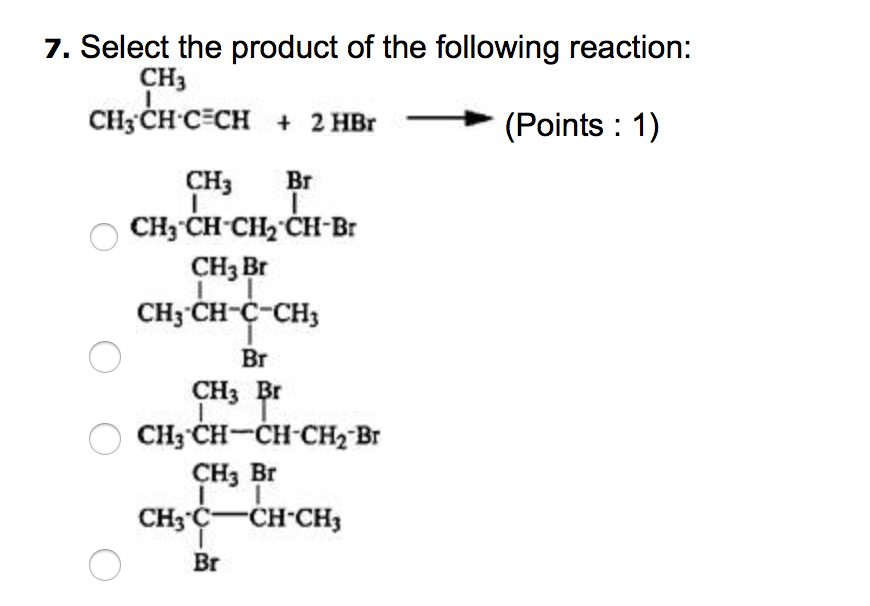

Transcribed Image Text:
2. The compound OOOO 8 0000 CH3CH₂CH₂CH₂CHCH₂CH₂CH3 CH3CH-CH3 4-isopropyloctane 5-isopropyloctane 5-sec-butyloctane 4-propyloctane 3. Complete combustion of an alkane produces what products? (Points : 1) has the IUPAC name (Points : 1) CO2 and O2 CO₂ and H₂O CO and H₂0 C and H₂O 7. Select the product of the following reaction: CH3 CH, CH-C=CH + 2 HBr (Points : 1) CH3 Br I CH3 CH-CH₂ CH-Br CH3 Br CH3-CH-C-CH3 Br CH3 Br 1 CH3 CH-CH-CH₂ Br CH3 Br I I CH3 C T Br CH-CH3 14. Select the correct IUPAC name of CH3CH-CH₂CH₂-O-CH3 CH3 (Points: 1) O3-methyl-1-methylbutane 1-methoxy-3-methylpentane methyl-3-methylpentyl ether O 1-methoxy-3-methylbutane 2. The compound OOOO 8 0000 CH3CH₂CH₂CH₂CHCH₂CH₂CH3 CH3CH-CH3 4-isopropyloctane 5-isopropyloctane 5-sec-butyloctane 4-propyloctane 3. Complete combustion of an alkane produces what products? (Points : 1) has the IUPAC name (Points : 1) CO2 and O2 CO₂ and H₂O CO and H₂0 C and H₂O 7. Select the product of the following reaction: CH3 CH, CH-C=CH + 2 HBr (Points : 1) CH3 Br I CH3 CH-CH₂ CH-Br CH3 Br CH3-CH-C-CH3 Br CH3 Br 1 CH3 CH-CH-CH₂ Br CH3 Br I I CH3 C T Br CH-CH3 14. Select the correct IUPAC name of CH3CH-CH₂CH₂-O-CH3 CH3 (Points: 1) O3-methyl-1-methylbutane 1-methoxy-3-methylpentane methyl-3-methylpentyl ether O 1-methoxy-3-methylbutane
Expert Answer:

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
The gasoline additive methyl t-butyl ether (MTBE) has been leaking into groundwater ever since its introduction in the 1990s. MTBE can be measured at parts per billion levels by solid-phase...
-
Complete and balance each combustion equation. a. C4H9OH + O2 ( ? b. CH3NO2 + O2 ( ?
-
Complete and balance each combustion equation. a. B2H6 + O2 ( ? (The oxide of boron formed is B2O3.) b. Al2S3 + O2 ( ? (The oxide of sulfur formed is SO2.) c. Al2S3 + O2 ( ? (The oxide of sulfur...
-
Consider the utility function U(x 1 , x 2 ) = x 1 x 2 with budget constraint p 1 x 1 + p 2 x 2 = c. (a) Show that the maximum of U(x 1 , x 2 ) subject to the budget constraint is equal to c 2 /(4p 1...
-
Mostek Corp., a Texas corporation, made a contract to sell computer-related products to North American Foreign Trading Corp., a New York corporation. North American used its own purchase order form,...
-
Let X and Y be independent continuous random variables with respective hazard rate functions X(t) and Y(t), and set W = min(X,Y). (a) Determine the distribution function of W in terms of those of X...
-
Consider the system \[\begin{array}{r} x^{\prime}=-4 x-y \\ y^{\prime}=x-2 y \end{array}\] a. Determine the second-order differential equation satisfied by \(x(t)\). b. Solve the differential...
-
Chaplin Arts, Inc.s comparative balance sheets for December 31, 2014 and 2013 follow. The following additional information about Chaplin Artss operations during 2013 is available: (a) Net income,...
-
Liza's budgets sales of $40,000 for May and $52,000 for June. Assume 60% of Liza's sales are for cash. The remaining 40% are credit sales; credit customers pay the entire amount owed in the month...
-
Your firm represents Amanda and Sam Baker, grandparents of two year old Brian Baker. Brian was recently injured in a home accident. The two year old stuck a hairpin into an electrical outlet and was...
-
Company "A" paid 35 percent premium for the acquisition of Company "B" in 2019. Company "A" subsequently needed to issue more stock. This illustrates the acquisition problem of: a. integration...
-
Suppose a box of mass m slides toward a spring with a speed of vii, presses 1.20 mm into the spring, and then bounces away with a final speed of 17.0 m/s. Find the initial speed of the box. Take into...
-
Focuses on Organizational Behavior and the application of theory to the workplace. Take two concepts or theories covered in this course and apply the concepts or theories to your current or past...
-
1. What are the business practices of failed mergers and acquisitions in the beverage industry in line with the significant money wastage and the new regulations? 2. What are the general and specific...
-
1. Langerhans cells, which play a role in defense against microbes, are located in the a. epidermis. b. dermis. c. hypodermis. d. subcutaneous layer. 2. The papillary layer of the skin is part of the...
-
In 2018, Apple hit $1 trillion in market capitalization, a value that was much greater than Amazon's market cap despite Amazon having a much higher stock share price. In 2020, Apple was the first to...
-
We all have different perspectives and ideas of appropriate family/parent involvement. Please share with the class, at least two ideas that you have on this topic. How did you find these ideas -...
-
The figure shows six containers, each of which is filled from the top. Assume that water is poured into the containers at a constant rate and each container is filled in 10 seconds. Assume also that...
-
Using the periodic table as a guide, write the condensed electron configuration and determine the number of unpaired electrons for the ground state of (a) Si, (b) Zn, (c) Zr, (d) Sn, (e) Ba, (f) Tl.
-
Given the following half-reactions and associated standard reduction potentials: (a) Write the equation for the combination of these half-cell reactions that leads to the largest positive emf and...
-
Proteins are polymers formed by condensation reactions of amino acids, which have the general structure In this structure, R represents ---- H, ----CH3, or another group of atoms. Draw the general...
-
Question: Sports Graphics, Inc. imports "Chill" brand coolers from Taiwan. Chill coolers have an outer shell of vinyl, with handles and pockets, and an inner layer of insulation. In a recent lawsuit,...
-
Question: In a recent WTO case, several nations claimed that American laws concerning shrimp fishing were unfair and illegal. The case demonstrated a conflict between two important values. What were...
-
Question: Flyby Knight (FK) contracts to sell 12 helicopters to Air Nigeria, for $8 million each. Payment is to be made by letter of credit, issued by the Bank of Nigeria, confirmed by Citibank in...

Study smarter with the SolutionInn App


