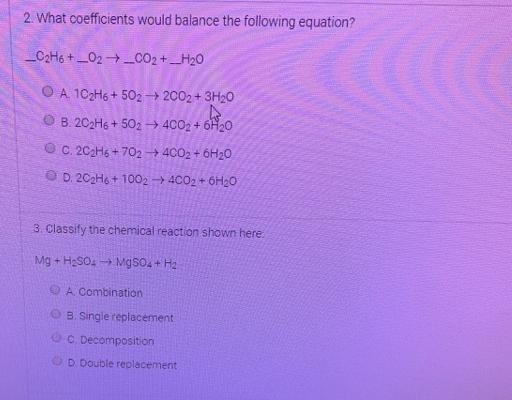
2. What coefficients would balance the following equation? _C2H6 +_02_002 +_H20 A 102H6 + 502 -...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
2. What coefficients would balance the following equation? _C2H6 +_02_002 +_H20 A 102H6 + 502 - 2002 + 3H20 B. 202H6 + 502 - 4C02 + 6H20 C. 202H6 + 702 4CO2 + 6H20 O D. 202H6 + 1002 - 4CO2 + 6H20 3. Classify the chemical reaction shown here Mg + H2SO. MgSO4 + H2 A Combination B. Single replacement C. Decomposition D Double replacement 2. What coefficients would balance the following equation? _C2H6 +_02_002 +_H20 A 102H6 + 502 - 2002 + 3H20 B. 202H6 + 502 - 4C02 + 6H20 C. 202H6 + 702 4CO2 + 6H20 O D. 202H6 + 1002 - 4CO2 + 6H20 3. Classify the chemical reaction shown here Mg + H2SO. MgSO4 + H2 A Combination B. Single replacement C. Decomposition D Double replacement
Expert Answer:
Answer rating: 100% (QA)
Answer 2 The balanced equation is Option C 2C2H67O24CO... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
Balance the following equation using the half-reaction method. H2O2+ ClO2 ClO2 + O2
-
A double replacement reaction between lead(IV) nitrate and sodium chloride will produce lead(IV) chloride and sodium nitrate. If 1.02 moles of lead(IV) nitrate is reacted, how many grams of sodium...
-
The chemical reaction A B + C has a rate constant that obeys the Arrhenius equation. Predict what happens to both the rate constant k and the rate of the reaction if the following were to occur. a....
-
Recent trends in recruiting rely on social capital by ______. Multiple choice question. making use of structural holes between groups in a social network where there are few relationships bridging...
-
Twilight Companys stock is selling for $60.25 per share, and the firms managers have just announced a $1.50 per share dividend payment. a. What should happen to Twilight Companys stock price on the...
-
Ian retired in December of 2021 at the age of 71 (his birthday is January 6). Ians retirement account was valued at $490,000 at the end of 2020 and $500,000 at the end of 2021. He has had all of his...
-
What three factors change air density?
-
Describe in detail all the facets of JetBlues product. What is being exchanged in a JetBlue transaction? In 2007, JetBlue was a thriving young airline with a strong reputation for outstanding...
-
The electric field in a particular space is E = (x + 2)1 N/C, with x in meters. Consider a cylindrical Gaussian surface of radius 39 cm that is coaxial with the x axis. One end of the cylinder is at...
-
The first quarter tax return needs to be filed for Prevosti Farms and Sugarhouse by April 15, 2019. For the purpose of the taxes, assume the second February payroll amounts were duplicated for the...
-
Vanessica, a CPA, used to work for Valley Girl Products in the earlier portion of 2020. Now, Vanessica works at Getty DaGreen Inc, CPA firm; she is the firm's specialist on various technical matters,...
-
Suppose two TCP connections share a path through a router R. The routers queue size is six segments; each connection has a stable congestion window of three segments. No congestion control is used by...
-
You are hired to design a reliable byte-stream protocol that uses a sliding window (like TCP). This protocol will run over a 1-Gbps network. The RTT of the network is 140 ms, and the maximum segment...
-
Use the formula in Example 1 to determine the sample size in Problems 5-14. The estimated prevalence is \(15 \%\), and the desired confidence level is \(92 \%\) with a margin of error of \(3 \%\).
-
For the network given in Figure 3.43, give the datagram forwarding table for each node. The links are labeled with relative costs; your tables should forward each packet via the lowest-cost path to...
-
Using the example network given in Figure 3.42, give the virtual circuit tables for all the switches after each of the following connections is established. Assume that the sequence of connections is...
-
Question 2 [15 marks] Create a Java application using arrays that sorts a list of integers in descending order. For example, if an array has values 106, 33, 69, 52, 17 your program should have an...
-
On July 1, 2011, Flashlight Corporation sold equipment it had recently purchased to an unaffiliated company for $480,000. The equipment had a book value on Flashlights books of $390,000 and a...
-
Phosphoric acid is usually obtained as an 85% phosphoric acid solution. If it is 15 M, what is the density of this solution? What is its molality?
-
Lead(IV) oxide is a strong oxidizing agent. For example, lead(IV) oxide will oxidize hydrochloric acid to chlorine, Cl2. Write the balanced equation for this reaction.
-
A reaction of the form aA Products is second order with a rate constant of 0.413 L/(mols). What is the half-life, in seconds, of the reaction if the initial concentration of A is 5.25 103 mol/L?
-
Use SecureRandom method ints to generate a stream of 50 random numbers in the range 1 to 999, then filter the resulting stream elements to select only the odd numbers and display the results in...
-
What percentage of the 9,449 survey respondents live in the Eastern part of the county?
-
(a) What percentage of those not completing high school were females? (b) What percentage of those not completing high school were males? (c) What percentage of those completing high school were...

Study smarter with the SolutionInn App


