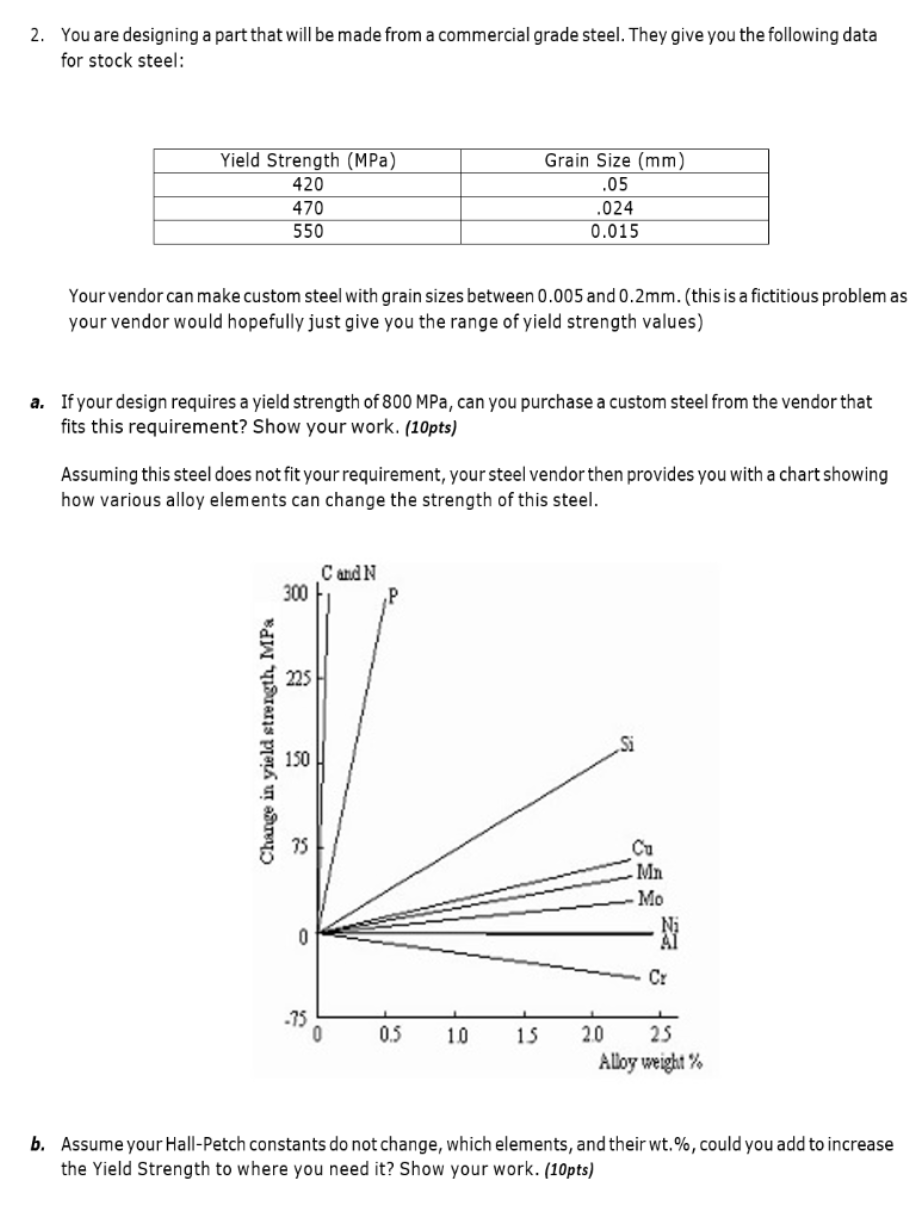
2. You are designing a part that will be made from a commercial grade steel. They...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
2. You are designing a part that will be made from a commercial grade steel. They give you the following data for stock steel: Yield Strength (MPa) 420 Grain Size (mm) .05 470 550 .024 0.015 Your vendor can make custom steel with grain sizes between 0.005 and 0.2mm. (this is a fictitious problem as your vendor would hopefully just give you the range of yield strength values) a. If your design requires a yield strength of 800 MPa, can you purchase a custom steel from the vendor that fits this requirement? Show your work. (10pts) Assuming this steel does not fit your requirement, your steel vendor then provides you with a chart showing how various alloy elements can change the strength of this steel. 300 C and N Change in yield strength, MPa 225 50 150 75 0 Si Ca Ma Mo Cr -75 0 0.5 10 15 20 25 Alloy weight% b. Assume your Hall-Petch constants do not change, which elements, and their wt.%, could you add to increase the Yield Strength to where you need it? Show your work. (10pts) 2. You are designing a part that will be made from a commercial grade steel. They give you the following data for stock steel: Yield Strength (MPa) 420 Grain Size (mm) .05 470 550 .024 0.015 Your vendor can make custom steel with grain sizes between 0.005 and 0.2mm. (this is a fictitious problem as your vendor would hopefully just give you the range of yield strength values) a. If your design requires a yield strength of 800 MPa, can you purchase a custom steel from the vendor that fits this requirement? Show your work. (10pts) Assuming this steel does not fit your requirement, your steel vendor then provides you with a chart showing how various alloy elements can change the strength of this steel. 300 C and N Change in yield strength, MPa 225 50 150 75 0 Si Ca Ma Mo Cr -75 0 0.5 10 15 20 25 Alloy weight% b. Assume your Hall-Petch constants do not change, which elements, and their wt.%, could you add to increase the Yield Strength to where you need it? Show your work. (10pts)
Expert Answer:

Related Book For 

Materials Science and Engineering An Introduction
ISBN: 978-0470419977
8th edition
Authors: William D. Callister Jr., David G. Rethwisch
Posted Date:
Students also viewed these mechanical engineering questions
-
respond to the instruction using the following criteria: Individual Written Case Exercise Respond to the specific questions provide in the case document it will be seven documents. Please make sure...
-
The U.S. Census Bureau classifies the 50 U.S. states into four regions: Northeast (9 states, New England and Middle Atlantic): CT, ME, MA, NH, NJ, NY, PA, RI, VT South (16 states, South Atlantic,...
-
Simplify each expression. Assume that all variables represent nonzero real numbers. 52
-
Simplify E(MSTr) for the random effects model when J1 = J2 = ... = JI = J?
-
During the global recession of 2008 and 2009, there were many accusations of unethical behavior by Wall Street executives, financial managers, and other corporate officers. At that time, an article...
-
Give reasons as to why the shape of the curve of the product life cycle is similar to that of the adoption of innovations curve.
-
Financial data for Bridger, Inc., for last year are as follows: The company paid dividends of $197,000 last year. The "Investment in Brier Company" on the balance sheet represents an investment in...
-
Lavage Rapide is a Canadian company that owns and operates a large automatic car wash facility near Montreal. The following table provides estimates concerning the company's costs: Fixed Cost Cost...
-
1. If you were in Jimmies shoes, would you sell Greg an equity stake in Lees Ice Cream? Explain. If Jimmie does sell equity to Greg for $3,300, what percentage of the business should he offer? 2....
-
You will develop a narrative report that provides detailed context on the key performance indicators and budget items along with a variety of institutional characteristics about the selected...
-
In Canada, education and healthcare systems are controlled by the governments and the market controls most of the businesses. Which term describes Canada's economy?
-
Describe the behaviour you wanted to change by stating your precise goal. Explain how your goal adheres to the SMART goals framework. The value of studying human behaviour and mental processes is in...
-
Assume that the company produces and sells 8 9 , 0 0 0 units during the year at a selling price of $ 8 . 1 6 per unit. Prepare a contribution format income statement for the year.
-
The following information is available from Ruby's financial records as at December 1, 2022. Cash $25,000 Furniture 8,000 Content of Home 5,000 House 300,000 Line of Credit 45,000 Mortgage 285,000...
-
Determine the future value of the following single amounts. Note: Use tables, Excel, or a financial calculator. Round your final answers to nearest whole dollar amount. (FV of $1, PV of $1, FVA of...
-
RECRUITMENT OF CANDIDATES Data Collection : Start by doing a thorough job analysis of Program Coordinators working in the Student Affairs department of your respective campus. Study the relevant...
-
The Higher the time period of the financial security the higher the. ............... risk. O a. Maturity O b. Default and Maturity Oc. Default O d. Liquidity
-
(a) Calculate and compare the specific longitudinal strengths of the glass-fiber, carbon-fiber, and aramid-fiber reinforced epoxy composites in Table 16.5 with the following alloys: tempered (315C)...
-
A polystyrene component must not fail when a tensile stress of 1.25 MPa (180 psi) is applied. Determine the maximum allowable surface crack length if the surface energy of polystyrene is 0.50 J/m2...
-
In Figure 9.38 is shown the pressure-temperature phase diagram for H2O. Apply the Gibbs phase rule at points A, B, and C; that is, specify the number of degrees of freedom at each of the points-that...
-
A system, consisting of \(2 \mathrm{~mol}\) of \(\mathrm{N}_{2}, 5 \mathrm{~mol}\) of \(\mathrm{H}_{2}\) and \(2 \mathrm{~mol}\) of \(\mathrm{NH}_{3}\) initially, is undergoing the following...
-
The following reaction takes place in a system consisting of \(3 \mathrm{~mol} \mathrm{CH}_{4}, 5 \mathrm{~mol} \mathrm{H}_{2} \mathrm{O}\), \(1 \mathrm{~mol} \mathrm{CO}\) and \(4 \mathrm{~mol}...
-
The study of thermodynamics enables us to understand (a) Whether the transformation of energy is feasible or not (b) To what extent the transformation will take place (c) In which direction the...

Study smarter with the SolutionInn App


