23. 24. 25. 26. Examine the phase diagram for the substance Bogusium (Bo) and select the...
Fantastic news! We've Found the answer you've been seeking!
Question:
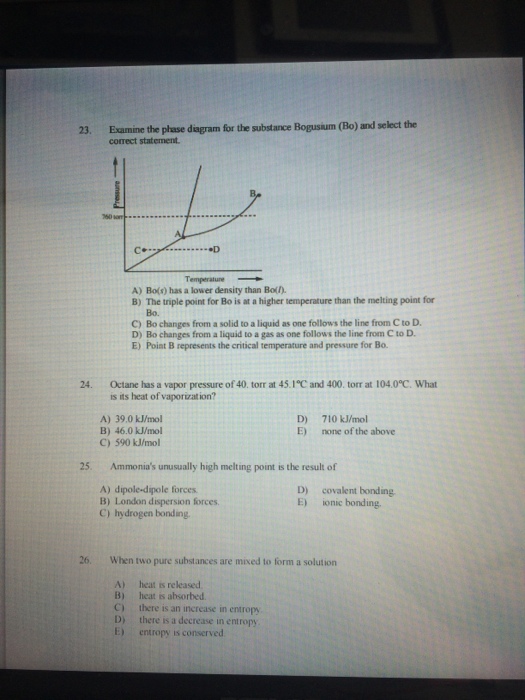
Transcribed Image Text:
23. 24. 25. 26. Examine the phase diagram for the substance Bogusium (Bo) and select the correct statement. •D Temperature A) Bo(s) has a lower density than Bo(). B) The triple point for Bo is at a higher temperature than the melting point for Bo. C) Bo changes from a solid to a liquid as one follows the line from C to D. D) Bo changes from a liquid to a gas as one follows the line from C to D. E) Point B represents the critical temperature and pressure for Bo. Octane has a vapor pressure of 40. torr at 45.1°C and 400. torr at 104.0°C. What is its heat of vaporization? A) 39.0 kJ/mol B) 46.0 kJ/mol C) 590 kJ/mol D) E) 710 kJ/mol none of the above Ammonia's unusually high melting point is the result of A) dipole-dipole forces. B) London dispersion forces. C) hydrogen bonding. D) E) covalent bonding ionic bonding. When two pure substances are mixed to form a solution A) heat is released. B) heat is absorbed. C) there is an increase in entropy D) there is a decrease in entropy. E) entropy is conserved. 23. 24. 25. 26. Examine the phase diagram for the substance Bogusium (Bo) and select the correct statement. •D Temperature A) Bo(s) has a lower density than Bo(). B) The triple point for Bo is at a higher temperature than the melting point for Bo. C) Bo changes from a solid to a liquid as one follows the line from C to D. D) Bo changes from a liquid to a gas as one follows the line from C to D. E) Point B represents the critical temperature and pressure for Bo. Octane has a vapor pressure of 40. torr at 45.1°C and 400. torr at 104.0°C. What is its heat of vaporization? A) 39.0 kJ/mol B) 46.0 kJ/mol C) 590 kJ/mol D) E) 710 kJ/mol none of the above Ammonia's unusually high melting point is the result of A) dipole-dipole forces. B) London dispersion forces. C) hydrogen bonding. D) E) covalent bonding ionic bonding. When two pure substances are mixed to form a solution A) heat is released. B) heat is absorbed. C) there is an increase in entropy D) there is a decrease in entropy. E) entropy is conserved.
Expert Answer:

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
From Figure 12.25, the phase diagram for the MgO-Al2O3 system, it may be noted that the spinel solid solution exists over a range of compositions, which means that it is non stoichiometric at...
-
The phase diagram for water is shown in FIGURE 17-37. (a) What is the temperature T1 on the phase diagram? (b) What is the temperature T2 on the phase diagram? (c) What happens to the...
-
The phase diagram for neon is Temperature (K) Use the phase diagram to answer the following questions. (a) What is the approximate value of the normal melting point? (b) Over what pressure range will...
-
Find the derivative of the function. y = e x-4
-
Refer to question 17 at the end of Chapter 2. Implement a spreadsheet model for this problem and solve it using Solver.
-
The trial balance for the General Fund of the City of Fairfield as of December 31, 2020, is presented here: Transactions for the year ended December 31, 2021, are summarized as follows: 1. The City...
-
Bonsai Boards income statement data for the year ended December 31,2010, follow. Assume that the ending inventory was accidentally overstated by $3,300. How would the inventory error affect Bonsai...
-
Julia Robertson is a senior at Tech, and she's investigating different ways to finance her final year at school. She is considering leasing a food booth outside the Tech stadium at home football...
-
1.Branson works for a firm that is expanding into a completely new line of business. He has been asked to determine an appropriate WACC for an average-risk project in the expansion division. Branson...
-
The proposed rates were not in the range the CEO expected given the pricing analysis. The CEO has asked the pricing actuary to verify the total projected loss cost excluding potential large storm...
-
There are few corporate blunders as staggering as Kodak's missed opportunities in digital photography, a technology that it invented. This strategic failure was the direct cause of Kodak's...
-
Is there anyone you would like to interview? If so, what do you want to learn from them?
-
What would you recommend (e.g., controls, monitoring) to deter future frauds from happening? Discuss and be specific.
-
In calculating common-sized ratios in a vertical and horizontal analysis, what is the base for calculating common-sized ratios for the income statement? A. Gross receipts B. Total assets C. Total...
-
What are the alleged frauds (e.g., accounts receivable skimming, disbursement fraud, embezzlement, larceny, payroll fraud, skimming, other)? Discuss each one.
-
In calculating common-sized ratios in a vertical and horizontal analysis, what is the base for calculating common-sized ratios for the balance sheet? A. Total assets B. Gross receipts C. Total...
-
echo " ' " id="MathJax-Element-1-Frame" role="presentation" style="font-size: 121%; position: relative;" tabindex="0"> " ' " ' Double ' " " id="MathJax-Element-2-Frame" role="presentation"...
-
Identify Thank You mission, strategy and core competencies. Identify strategy changes that have taken place at Thank You since its founding in 2008. Your answer must in text references and must be...
-
The rate of a first-order reaction is followed by spectroscopy, monitoring the absorbance of a colored reactant at 520 nm. The reaction occurs in a 1.00-cm sample cell, and the only colored species...
-
You have to prepare a pH 5.00 buffer, and you have the following 0.10 M solutions available: HCOOH, HCOONa, CH3COOH, CH3COONa, HCN,and NaCN.Which solutions would you use? How many milliliters of each...
-
Propylene, C3H6, is a gas that is used to form the important polymer called polypropylene. Its Lewis structure is (a) What is the total number of valence electrons in the propylene molecule? (b) How...
-
Differentiate between trace point and pitch point.
-
How the cam size is defined?
-
Define pressure angle of a cam.

Study smarter with the SolutionInn App


