The solubility product, Ksp, for Mg(OH)2 is 8.9 x 10-12 Determine the following. a. The solubility...
Fantastic news! We've Found the answer you've been seeking!
Question:
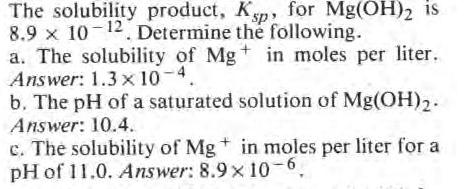
Transcribed Image Text:
The solubility product, Ksp, for Mg(OH)2 is 8.9 x 10-12 Determine the following. a. The solubility of Mg+ in moles per liter. Answer: 1.3 x 10-4. b. The pH of a saturated solution of Mg(OH)2. Answer: 10.4. c. The solubility of Mg + in moles per liter for a pH of 11.0. Answer: 8.9 x 10-6, The solubility product, Ksp, for Mg(OH)2 is 8.9 x 10-12 Determine the following. a. The solubility of Mg+ in moles per liter. Answer: 1.3 x 10-4. b. The pH of a saturated solution of Mg(OH)2. Answer: 10.4. c. The solubility of Mg + in moles per liter for a pH of 11.0. Answer: 8.9 x 10-6,
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Consider the diode bridge rectifier shown below with the voltage regulating capacitor in parallel with the load resistor. 60Hz 120V 10:1 C-470F IL R=1kQ a) For the input signal, sketch waveforms of...
-
In a bimolecular reaction A + B M, a moles per liter of a substance A and b moles per liter of a substance B are combined. Under constant temperature the rate of reaction is y' = k(a y) (b y) that...
-
Activity Optimistic Time Estimate (weeks) Most Likely Time Estimates (weeks) Pessimistic Time Estimates (weeks) Immediate Predecessor(s) A 3 5 10 none B 3 5 9 A C 5 7 13 A D 4 5 12 B E 5 11 16 C F 3...
-
This case will enable you to practice conducting planning and substantive analytical procedures for accounts in the revenue cycle. When analyzing the financial data, you may assume that the 2015...
-
Petey Ltd. has a policy of obtaining an actuarial pension valuation every three years. Based on the individual components of its annual pension expense, Petey Ltd.s accrued benefit obligation as at...
-
The rectangular block shown rotates about the diagonal OA with a constant angular velocity of 6.76 rad/s. Knowing that the rotation is counterclockwise as viewed from A, determine the velocity and...
-
Compute the expected frequencies under the null hypothesis of homogeneity. Exercises 79 refer to the following data: At an assembly plant for light trucks, routine monitoring of the quality of welds...
-
A firm has the following short run demand and cost schedule for a product. Q = 200 5P TC = 400 + 4Q a. What are price, quantity and profit for this company? b. Suppose the above demand shifted to Q...
-
Plan finances for new business ventures in Australia Profit, loss and cash flow projections Monthly projected $ sales Jul Aug Sep Oct Nov Dec Jan Feb Mar Apr May Jun Total Coffee regular $ 8,734...
-
Compte, Incorporated (a U.S.-based company), establishes a subsidiary in Croatia on January 1, 2023. The following account balances for the year ending December 31, 2024, are stated in kuna (K), the...
-
1. According to the midpoint method, the price elasticity of demand between points A and B on the initial graph is approximately (0, 0.33, 3, 7.5). 2. Suppose the price of bippitybops is currently...
-
A simple geometry of a slope is shown in Figure 2, with homogeneous deposit of soft clay on a stiffer clay at the bottom. Properties and thicknesses of the layers are as showed in the figure. a) Use...
-
1. Name two types of pathways that help a body maintain homeostasis, and an example of each. 2. What is the function of platelets? 3. Describe why air enters the lungs during inhalation. In your...
-
Now that you have assembled Sunset's raw data into useable income statements and balance sheets, and have evaluated the cash flow situation Sunset is faced with, Christina has asked you to assess any...
-
Journalize the following transactions using the direct write-off method of accounting for uncollectible receivables. April 1 Sold merchandise on account to Jim Dobbs, $6,600. The cost of goods sold...
-
(a) Consider an attractive spherical-well potential: V(x) = r < R and Vir) - 0, otherwise. Compute the a-wave phase shifts and -Vo for the scattering cross-sections for a particle of mass m in the...
-
Appendix 1 I Summary notes of your discussion with Scott Ginny GPP provides copying and small print job services from a single location close to a university. The business was organized in January...
-
An auto-parts manufacturer is considering establishing an engineering computing center. This center will be equipped with three engineering workstations each of which would cost $25,000 and have a...
-
When dinitrogen pentoxide, N2O5, a white solid, is heated, it decomposes to nitrogen dioxide and oxygen. If a sample of N2O5 produces 1.315 g O2, how many grams of NO2 are formed? 2N205(s) 4NO2(g) +...
-
Greater variation exists between the properties of the first and second members of a family in the periodic table than between other members. Discuss this observation for the oxygen family using the...
-
A sample of sodium peroxide, Na2O2, was reacted with an excess of water. 2Na2O2(s) + 2H2O(l ) 4NaOH(aq) + O2(g) All of the sodium peroxide reacted, and the oxygen was collected over water at 21oC....
-
The gross profit ratio is equal to: (a) profit divided by net sales. (b) cost of sales divided by net sales. (c) net sales minus cost of sales, divided by net sales. (d) net sales minus cost of...
-
What inventory cost flow method does Dominos Pizza Enterprises Ltd use for its inventories?
-
Which inventory cost flow method produces the highest profit in a period of rising prices?

Study smarter with the SolutionInn App


