2.4 mscf/d of the gas is compressed adiabatically and reversibly from 100 psi and 110F to...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
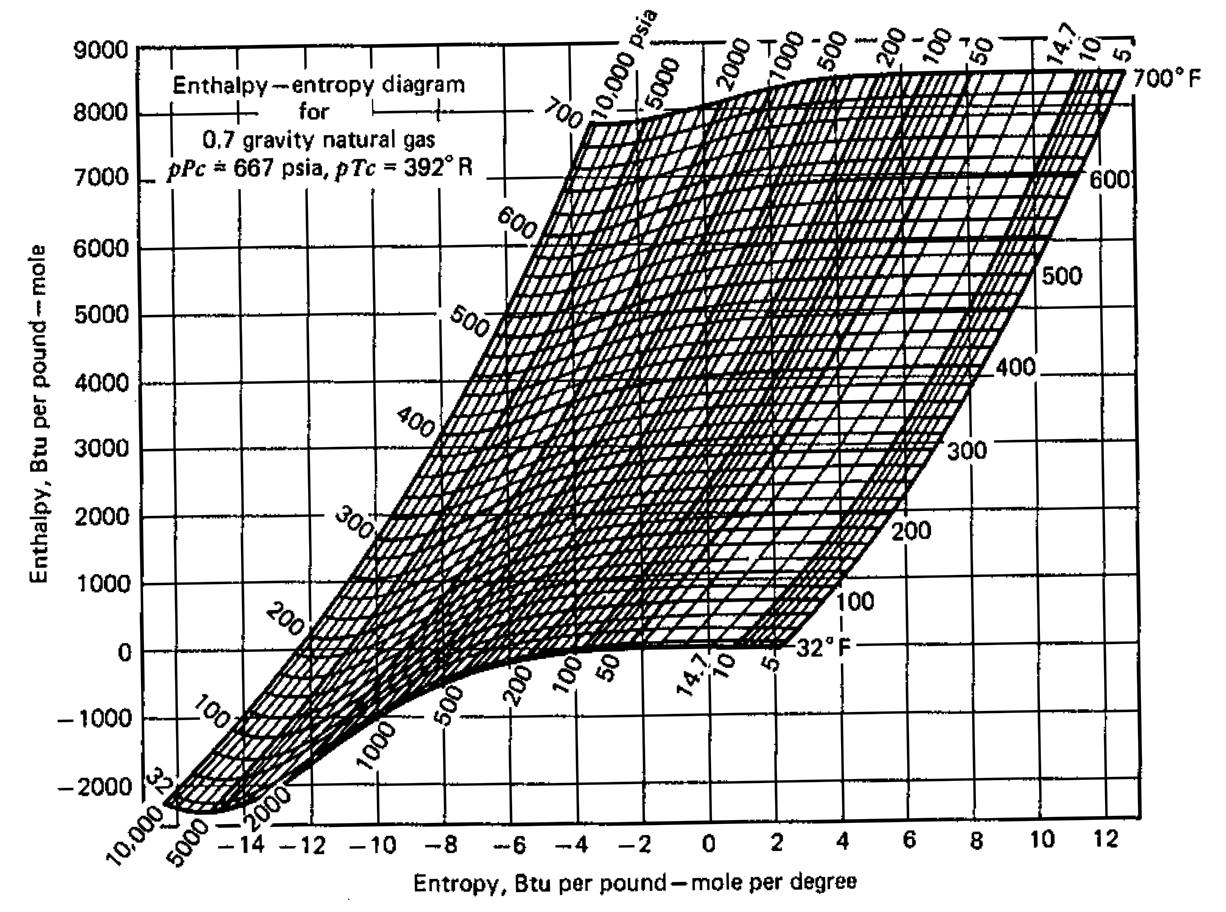
2.4 mscf/d of the gas is compressed adiabatically and reversibly from 100 psi and 110F to 1000 psi (i) What is the final temperature of this gas (ii) What is the energy requirement of this process (ii) What is the energy (in horsepower) requirement of the compressor 1 Horsepower = 2545 BTU/hr Use information provided at the end. Please explain how you calculate your answers. 700° F 9000 Enthalpy-entropy diagram for 700 8000 600. 0,7 gravity natural gas pPc * 667 psia, pTc = 392° R 7000 600 500 6000 500 400 5000 4000 400 300 3000 200 2000 100 1000 200 32° F 100 - 1000 10 12 -2000 4 8 2 -8 -6 -4 -2 - 14 -12 -10 Entropy, Btu per pound-mole per degree 200 00s 00 w00 00s 000 000, 0009 10,000 psia Enthalpy, Btu per pound -mole 2.4 mscf/d of the gas is compressed adiabatically and reversibly from 100 psi and 110F to 1000 psi (i) What is the final temperature of this gas (ii) What is the energy requirement of this process (ii) What is the energy (in horsepower) requirement of the compressor 1 Horsepower = 2545 BTU/hr Use information provided at the end. Please explain how you calculate your answers. 700° F 9000 Enthalpy-entropy diagram for 700 8000 600. 0,7 gravity natural gas pPc * 667 psia, pTc = 392° R 7000 600 500 6000 500 400 5000 4000 400 300 3000 200 2000 100 1000 200 32° F 100 - 1000 10 12 -2000 4 8 2 -8 -6 -4 -2 - 14 -12 -10 Entropy, Btu per pound-mole per degree 200 00s 00 w00 00s 000 000, 0009 10,000 psia Enthalpy, Btu per pound -mole
Expert Answer:
Answer rating: 100% (QA)
Given Initial pressure R10o psi 2 1000 psi Final ressure I psi 689476 Pa Also Init... View the full answer

Posted Date:
Students also viewed these physics questions
-
Please explain how you will be utilizing technology to make your business more effective and efficient. Explore the cloud technology concept. Discuss the importance of information technology planning...
-
Determine the specific volume (v) of the gas in a 1 m3 chamber filled with (a) Hydrogen (b) Carbon-dioxide. The pressure inside is 1 atm and the temperature is 25oC. Use the ideal gas (IG) state...
-
Refer to the case information provided at the end of Chapters 10 and 11 and the domain class diagram at the end of Chapter 9. Review and update your results from performing the tasks at the end of...
-
The jet plane starts from rest at s = 0 and is subjected to the acceleration shown. Determine the speed of the plane when it has traveled 1000 ft. Also, how much time is required for it to travel...
-
The following data relate to the operations of Slick Software, Inc., during 2011. Continuing operations: Net sales . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . ....
-
Suppose all firms in a competitive industry are operating at output level for which price is equal to long- run marginal cost. True or false: This industry is necessarily in long- run equilibrium.
-
What is the value of a put option if the underlying stock price is $60, the strike price is $65, the underlying stock volatility is 25 percent, and the risk-free rate is 5 percent? Assume the option...
-
The contribution margin income statement of Pepperpike Coffee for February follows: Pepperpike Coffee contribution margin Income Statement For month ended February 29 Sales...
-
washed off within 5 minutes. For the other 500 subjects, the oil was washed off after 5 minutes. The results are summarized in the following table. Time Within Which Oil Was Washed Off Reaction...
-
Below is the number of hand-washing episodes 20 patients with obsessive-compulsive disorder reported having the previous day. 21, 8, 11, 9, 12, 10, 10, 5, 9, 18, 17, 3, 6, 14, 18, 16, 19, 3, 22, 7...
-
I need help understanding the difference between, Quantitative forecasting Methods Using Time Series Methods. Briefly explain the following Quantitative Time Series methods and give an example of...
-
To determine the float, which formula should you use? a. Late finish-early finish (LF-EF) or late finish-late start (LF-LS) b. Late start-early start (LS-ES) or late finish-late start (LF-LS) c. Late...
-
What is ethics?
-
In the original network diagram question, if Activity D increases from four to five days, what is the critical path, and what is the length? a. BDE, 33 days b. ACE, 15 days c. BFG, 21 days d. BDG, 23...
-
What is the drill-down approach?
-
Using the original network diagram question, what is the slack of Activity F? a. Two days b. One day c. Four days d. Not enough information
-
3. ABC Corporation is a construction company. It borrowed money from a bank for the construction of Building X. Building X is a qualifying asset. ABC Corporation pays interest expense on the money it...
-
What recommendations would you make to Big Four firms to help them (1) avoid confrontations with governmental officials in an authoritarian society and (2) deal effectively with such confrontations...
-
Explain the difference between spot and forward exchange rates.
-
You have just been asked to conduct an executive program for the international controllers office of a company from your home country that is becoming increasingly involved in exports to countries in...
-
Find a currency of your choice. How has its value changed against the U.S. dollar over the last ninety days? What about since January 3, 1975?

Study smarter with the SolutionInn App

