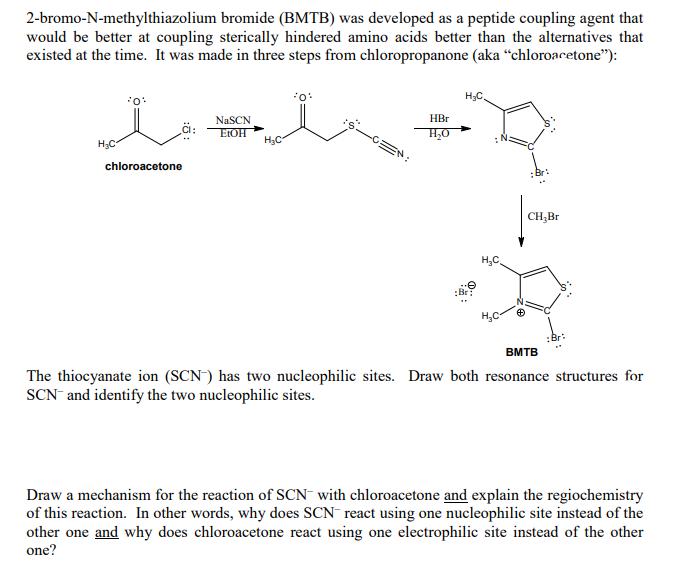
2-bromo-N-methylthiazolium bromide (BMTB) was developed as a peptide coupling agent that would be better at coupling...
Fantastic news! We've Found the answer you've been seeking!
Question:
![]()
Transcribed Image Text:
2-bromo-N-methylthiazolium bromide (BMTB) was developed as a peptide coupling agent that would be better at coupling sterically hindered amino acids better than the alternatives that existed at the time. It was made in three steps from chloropropanone (aka "chloroacetone"): H₂C *0* CI: chloroacetone NaSCN EtOH H₂C *0* HBr H₂O H₂C :Br: H₂C CH₂Br ;Br: BMTB The thiocyanate ion (SCN) has two nucleophilic sites. Draw both resonance structures for SCN and identify the two nucleophilic sites. Draw a mechanism for the reaction of SCN with chloroacetone and explain the regiochemistry of this reaction. In other words, why does SCN react using one nucleophilic site instead of the other one and why does chloroacetone react using one electrophilic site instead of the other one? (c) Propose a reasonable mechanism for the second step in the synthesis of BMTB. (shown below) 2-bromo-N-methylthiazolium bromide (BMTB) was developed as a peptide coupling agent that would be better at coupling sterically hindered amino acids better than the alternatives that existed at the time. It was made in three steps from chloropropanone (aka "chloroacetone"): H₂C *0* CI: chloroacetone NaSCN EtOH H₂C *0* HBr H₂O H₂C :Br: H₂C CH₂Br ;Br: BMTB The thiocyanate ion (SCN) has two nucleophilic sites. Draw both resonance structures for SCN and identify the two nucleophilic sites. Draw a mechanism for the reaction of SCN with chloroacetone and explain the regiochemistry of this reaction. In other words, why does SCN react using one nucleophilic site instead of the other one and why does chloroacetone react using one electrophilic site instead of the other one? (c) Propose a reasonable mechanism for the second step in the synthesis of BMTB. (shown below)
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Draw both resonance structures of the enolate formed when each of the following ketones is treated with a strong base: (a) (b) (c) (d) (e)
-
Are amino acids other than the usual 20 amino acids found in proteins? If so, how are such amino acids incorporated into proteins? Give an example of such an amino acid and a protein in which it...
-
Propose a mechanism for the reaction of (a) 1-methylcyclohexanol with HBr to form 1-bromo-1-methylcyclohexane. (b) 2-cyclohexylethanol with HBr to form 1-bromo-2-cyclohexylethane?
-
We consider the following CFG SE + SIE E 01|2|3|4|5|67|8|9|(S) Apply the leftmost derivation and rightmost derivation with a top-down parser for this sentence: (3+7+ (1+4)) + 2
-
Section 8.4 presents a second-order differential equation that can be used to analyze the unforced oscillations of an automobile shock absorber. Given m = 1.2 x 10 6 g , c = l x 10 7 g /s, and k...
-
Organelles compartmentalize a cell. What advantage does this offer a large cell? Cite two examples of organelles and the activities they compartmentalize.
-
How do purchasing decisions affect the amount of taxes you pay?
-
The normal capacity of a factory is 8,000 units per month. Cost and production data follow: Standard application rate for fixed overhead . . . . . . . . . . . . . . . . . . $0.50 per unit Standard...
-
Question 41 0/1 pts 1. Nina, age 54, earns $125.000 annually from ABC Incorporated. ABC sponsors a SIMPLE and matches all employee deferrals 100% up to a 2% contribution Assuming Nina defers the...
-
Extend Example 20.3 to calculate CVA when default can happen in the middle of each month. Assume that the default probability during the first year is 0.001667 per month and the default probability...
-
A firm needs to fully satisfy the demand, which is fixed to 900 units and generates unit revenue of $10. However, its production is subject to the random yield. That is, only a random percentage of...
-
Elon Musk decided to take over Twitter. Twitter has 50 million stocks outstanding with a current price of $40 per share. On 1/07/2022 Musk offered $48 per share conditioned on buying at least 50% of...
-
Calculate the predetermined over head rate assuring sandhill company estimates total manufacturing overhead costs of 1 0 0 8 0 0 0.
-
4. The orbit of asteroid Vesta has a semimajor axis of 2.36 AU and an eccentricity of 0.089. Its mass is 2.59x1020 kg and, like all asteroids, it orbits the Sun, whose mass is 1.99x1030 kg. Calculate...
-
Michael s company uses a single predetermined overhead rate using direct labor hours as the allocation base ( driver ) . a ) Calculate the Predetermined Overhead Rate given the following information:...
-
Convert the following decimal numbers to the base indicated: 1. 175 to binary 2. 25.86 to binary 3. 1938 to hexadecimal 4. 5386 to hexadecimal
-
After running an ANOVA to compare the mean price of my three vehicle models, I need help on determining if there is a significant difference between the means. I attached a photo.
-
Why is the national security argument for tariffs questionable?
-
Beleaguered State Bank (BSB) holds $250 million in deposits and maintains a reserve ratio of 10 percent. a. Show a T-account for BSB. b. Now suppose that BSBs largest depositor withdraws $10 million...
-
Using a diagram of the labor market, show the effect of an increase in the minimum wage on the wage paid to workers, the number of workers supplied, the number of workers demanded, and the amount of...
-
What kind of agreement is illegal for businesses to make? Why are the antitrust laws controversial?
-
\(13 \times\left(\frac{6}{11} \times 14 ight)=\left(13 \times \frac{6}{11} ight) \times 14\) Identify the property of real numbers that is being illustrated.
-
\(4+(8+\sqrt{7})=(4+8)+\sqrt{7}\) Identify the property of real numbers that is being illustrated.
-
\(6.3 \times \sqrt{14}=\sqrt{14} \times 6.3\) Identify the property of real numbers that is being illustrated.

Study smarter with the SolutionInn App



