Evaluate the iterated integral by converting to polar coordinates. 2x-x2 On converting into polar coordinates, (x2 +
Fantastic news! We've Found the answer you've been seeking!
Question:
Evaluate the iterated integral by converting to polar coordinates.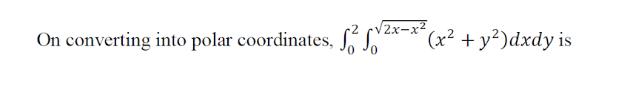
Transcribed Image Text:
2x-x2 On converting into polar coordinates, (x2 + y²)dxdy is 2x-x2 On converting into polar coordinates, (x2 + y²)dxdy is
Expert Answer:
Answer rating: 100% (QA)
On Conventing in to polas coondinaly into polar coordinales SCxty d... View the full answer

Posted Date:
Students also viewed these mathematics questions
-
Polar Coordinates Ï = x 2 + y 2 , θ = arctan (y/n) give where Ï' = dÏ/dθ. Derive this. Use it to find the total length of cardioid Ï = α(1...
-
FACTORING ACCOUNTS RECEIVABLE On February 1, Fotis Company factored $80,000 of accounts receivable without recourse with Fast Finance Company on a notification basis. Fast Finance charged a factoring...
-
In converting from polar coordinates to rectangular coordinates, what formulas will you use?
-
Based on Exhibit 1, the expected future value of Bond I at maturity is closest to: A. 98.80. B. 103.74. C. 105.00. Lena Liecken is a senior bond analyst at Taurus Investment Management. Kristel...
-
From 1789 through 1870, the last names of associate justices appointed to the U.S. Supreme Court were as coded below. Last names beginning with AM are shown as a 1, those beginning with NZ as a 2....
-
A product item for RMO is not the same as an inventory item. A product item is something like a mens leather hunting jacket supplied by Leather R Us. An inventory item is a specific size and color of...
-
We will use the Minitab macro NormNP, or the R function normnp, to nd the posterior distribution of the normal mean when we have a random sample of size n from a normal( , 2 ) distribution with...
-
Ramona Company incurred the following costs. 1. Sales tax on factory machinery purchased $ 5,000 2. Painting of and lettering on truck immediately upon purchase 700 3. Installation and testing of...
-
123456789 10 What is the need of MIS? What are the objectives of MRP? JIT introduced in which country? Advantages of TQM. Discuss about six sigma. Define various levels of CMM. What is mean by supply...
-
Case Study: Google Inc.s Acquisition of AdMob; Questions 1,5 1. How do investors evaluate young technology companies such as Google and how do these investor evaluations in turn impact management...
-
Ge is an indirect bandgap semiconductor. Describe the two-step and one-step light absorption processes in Ge at different wavelength.
-
Suppose a different company offers Ludo Corporation a "Loss Control" service which costs S (in dollars) and which cuts the probability of all losses in half (such that the probability of both the...
-
How much equity would Phil have in his home based on the following information: Purchased price of $425,000 Fair market value of $432,000 20% down payment Has made $15,000 in principal payments
-
Should civility be a criterion for job performance? Discuss the implications of leaders and managers who lack appropriate leadership skills.
-
For a large boiler, you have the choice of the following energy resources. Calculate the point of use cost per GJ for each Natural gas at $ 3.00 per kg, efficiency 75% Electricity at $ 0.18/kWh,...
-
When preparing the bank reconciling, how are NSF treated? Explain.
-
Suppose a country has national savings of $861.97 and net capital outflow at $(364.01). What is the value of total national investment? Round your answer to two (2) decimal places
-
You are standing at x = 9.0 km and your assistant is standing at x = 3.0 km. Lightning bolt 1 strikes at x = 0 km and lightning bolt 2 strikes at x = 12.0 km. You see the flash from bolt 2 at t = 10...
-
The latent heat of vaporization per unit mass of a pure substance at a given temperature, \(\lambda\), is defined as the difference in enthalpy between the saturated vapor and saturated liquid at the...
-
Calculate the absolute humidity, relative saturation, and dew-point temperature of a mixture of benzene-nitrogen at a pressure of \(1.5 \mathrm{bar}\) and temperature of \(320 \mathrm{~K}\) if the...
-
It is desired to dehumidify \(1.2 \mathrm{~m}^{3} / \mathrm{s}\) of air, available at \(311 \mathrm{~K}\) with a wet-bulb temperature of \(303 \mathrm{~K}\), to a wet-bulb temperature of \(288...

Study smarter with the SolutionInn App

