3. (6 pts) Use the answer bank and label the molecular shape around each of the...
Fantastic news! We've Found the answer you've been seeking!
Question:
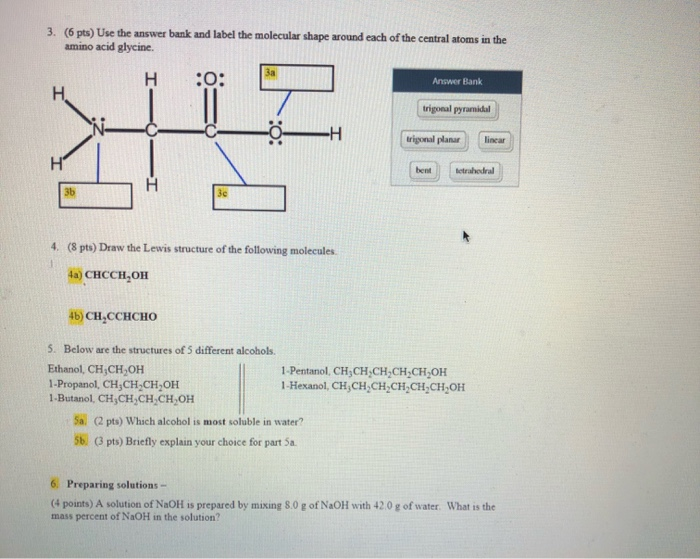
Transcribed Image Text:
3. (6 pts) Use the answer bank and label the molecular shape around each of the central atoms in the amino acid glycine. H Н. 3b :0: 30 3a 4. (8 pts) Draw the Lewis structure of the following molecules. 1 4a) CHCCH₂OH 4b) CH₂CCHCHO 5. Below are the structures of 5 different alcohols. Ethanol, CH₂CH₂OH 1-Propanol, CH3CH₂CH₂OH 1-Butanol, CH₂CH₂CH₂CH₂OH H 5a. (2 pts) Which alcohol is most soluble in water? 5b. (3 pts) Briefly explain your choice for part 5a. Answer Bank trigonal pyramidal trigonal planar linear bent tetrahedral 1-Pentanol, CH₂CH₂CH₂CH₂CH₂OH 1-Hexanol, CH₂CH₂CH₂CH₂CH₂CH₂OH 6. Preparing solutions - (4 points) A solution of NaOH is prepared by mixing 8.0 g of NaOH with 42.0 g of water. What is the mass percent of NaOH in the solution? 3. (6 pts) Use the answer bank and label the molecular shape around each of the central atoms in the amino acid glycine. H Н. 3b :0: 30 3a 4. (8 pts) Draw the Lewis structure of the following molecules. 1 4a) CHCCH₂OH 4b) CH₂CCHCHO 5. Below are the structures of 5 different alcohols. Ethanol, CH₂CH₂OH 1-Propanol, CH3CH₂CH₂OH 1-Butanol, CH₂CH₂CH₂CH₂OH H 5a. (2 pts) Which alcohol is most soluble in water? 5b. (3 pts) Briefly explain your choice for part 5a. Answer Bank trigonal pyramidal trigonal planar linear bent tetrahedral 1-Pentanol, CH₂CH₂CH₂CH₂CH₂OH 1-Hexanol, CH₂CH₂CH₂CH₂CH₂CH₂OH 6. Preparing solutions - (4 points) A solution of NaOH is prepared by mixing 8.0 g of NaOH with 42.0 g of water. What is the mass percent of NaOH in the solution?
Expert Answer:

Related Book For 

Cambridge International AS And A Level Biology
ISBN: 9781107636828
4th Edition
Authors: Mary Jones, Richard Fosbery, Jennifer Gregory, Dennis Taylor
Posted Date:
Students also viewed these chemistry questions
-
The amino acid glycine (H2N-CH2-COOH) can participate in the following equilibria in water: (a) Use the values of Ka and Kb to estimate the equilibrium constant for the intramolecular proton transfer...
-
Glycine is an amino acid that can be obtained from most proteins. In solution, glycine exists in equilibrium between two forms: (a) Consult Table 3.1 and state which form is favored at equilibrium. ...
-
The diffusion constant for the amino acid glycine in water has a value of 1.06 10-9 m2/s. In a 2.0-cm-long tube with a cross-sectional area of 1.5 10-4 m2, the mass rate of diffusion is m/t = 4.2 ...
-
Use the trapezium rule, with interval-halving and extrapolation, to evaluate 0 log(cosh x) dx to 4dp
-
Briefly discuss some of the service that international banks provide their customers and the marketplace.
-
Convert: a. 68oF to degrees Celsius b. 23oF to degrees Celsius c. 26oC to degrees Fahrenheit d. 70oC to degrees Fahrenheit
-
The chief accountant of Uncertain Ltd is not sure of the appropriate accounting treatment for a number of events occurring during the year 2005/6. (i) A significant number of employees have been made...
-
Thakin Stairs Co. of Moore designs and builds factory-made premium wooden stairways for homes. The manufactured stairway components (spindles, risers, hangers, hand rails) permit installation of...
-
Tamarisk Inc. has the following shares outstanding: 64,600 common and 16,500 $0.85 cumulative preferred. On December 15, the directors declared dividends of $67,915 to the shareholders of record on...
-
The following cross tabulation shows the average speed of the 25 winners by year of the Daytona 500 automobile race (The 2013 World Almanac). a. Calculate the row percentages. b. W hat is the...
-
Suppose 56%56% of the students in a university are baseball players. If a sample of 840840 students is selected, what is the probability that the sample proportion of baseball players will be greater...
-
Describe the concepts, principles and constraints underlying financial statements.
-
What form of representation would you recommend for this new market or would you consider setting up a manufacturing subsidiary? Give reasons for your decision.
-
Soon after the precipitous decline of Arizonas real estate market in 1989, BFA management decided to establish a number of related affiliates. These affiliates were controlled by individuals with...
-
Distinguish between the effect on a CPA firms practice of enforcing the rules of conduct by the AICPA versus a state Board of Accountancy.
-
What, if any, further research needs to be undertaken before attempting to export to the United States?
-
O HO: FOMO in participants who used social media for 2 hours/day < FOMO in participants who do not use social media O HO: FOMO in participants who used social media for 2 hours/day # FOMO in...
-
Cassandra Casey operates the Futuristic Antique Store. She maintains subsidiary ledgers for accounts payable and accounts receivable. She presents you with the following information for October 2019:...
-
Cigarette smoke contains tar, nicotine and carbon monoxide. Tar contains carcinogens. a. Describe the effect of tar on the lining of the bronchi in the lungs. b. Describe the effects of nicotine and...
-
Copy and complete the table to compare artificial selection with natural selection. Natural selection Artificial selection The selective agent is the total environment of the organism Adaptations to...
-
The electron micrograph on page 25 shows part of a secretory cell from the pancreas. The secretory vesicles are Golgi vesicles and appear as dark round structures. The magnification is 8000. a. Copy...
-
Markup. In this exercise, we introduce the concept of markup. The markup on an item is the difference between how much a store sells an item for and how much the store paid for the item. Suppose...
-
Repeated Discounts. In this exercise, we explore applying more than one discount to an item. Suppose a store cuts the price on an item by \(50 \%\), and then offers a coupon for \(25 \%\) off any...
-
Unit Conversion. There are 12 inches in a foot, 3 feet in a yard, and 1,760 yards in a mile. Convert 10 miles to inches. To do so, first convert miles to yards. Next, convert the yards to feet. Last,...

Study smarter with the SolutionInn App


