3 A vessel containing water is put in a dry sealed room of volume 76 m...
Fantastic news! We've Found the answer you've been seeking!
Question:
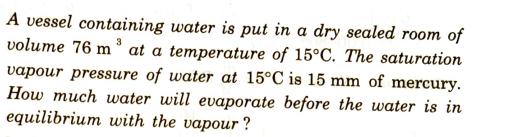
Transcribed Image Text:
3 A vessel containing water is put in a dry sealed room of volume 76 m at a temperature of 15C. The saturation vapour pressure of water at 15C is 15 mm of mercury. How much water will evaporate before the water is in equilibrium with the vapour? 3 A vessel containing water is put in a dry sealed room of volume 76 m at a temperature of 15C. The saturation vapour pressure of water at 15C is 15 mm of mercury. How much water will evaporate before the water is in equilibrium with the vapour?
Expert Answer:
Related Book For 

Physics
ISBN: 978-0077339685
2nd edition
Authors: Alan Giambattista, Betty Richardson, Robert Richardson
Posted Date:
Students also viewed these physics questions
-
A saturated water vapour is contained in a cylindrical vessel under a weightless piston at a temperature t = 100 C. As a result of a slow introduction of the piston a small fraction of the vapour m =...
-
A vessel is filled with gas at some equilibrium pressure and temperature. Can all gas molecules in the vessel have the same speed?
-
Mercury is a liquid at room temperature and pressure, but its vapor is present in the atmosphere from natural sources, such as volcanoes, and from human ("anthropogenic") activities such as coal...
-
When 12.0 V is applied to a DC motor it rotates at 24 rad/s. The current in the motor is 0.50 A and the "back" emf induced in the motor is 6.0 V. The resistance of the motor will be?
-
Preparing a balance sheet and an income statement The following information is based on accounting data for 2007 and 2008 for ChemAsia Limited (ChemAsia), a large petrochemicals company in China....
-
22. What are the gravitational constant and gravitational acceleration in each of the following unit systems? Unit System SI English (using slugs) English (using lbm) Gravitational constant...
-
The program computes federal withholding tax for each employee by multiplying gross pay times a tax rate determined from Table 9-9. The program next computes state withholding tax for each employee...
-
Birmingham Bowling Ball Company (BBBC) uses a job-order costing system to accumulate manufacturing costs. The companys work-in-process on December 31, 20x3, consisted of one job (no. 3088), which was...
-
In a year in which common stocks offered an average return of 1 8 % and Treasury bills offered 7 % . The risk premium for common stocks was: A . 3 % B . 1 % C . 1 1 % D . 1 8 %
-
Use Solver to create Answer and Sensitivity Reports for question 15 at the end of Chapter 2 and answer the following questions: a. How much excess wiring and testing capacity exists in the optimal...
-
Due to management bias, which of the following accounts tend to have a higher inherent risk of understatement? Multiple Choice Equity accounts. Asset and Revenue accounts. Contributed Surplus...
-
Assuming the following transaction details for an acquisition, calculate the amount of goodwill that will be recorded on the transaction: Purchase consideration given = 46.5 million shares issued,...
-
How do contemporary societies navigate the tension between normative regulation and individual freedom in the context of deviance and social sanctions ?
-
Consider the schema below, complete the following using the specified query language: Brands (brand id, brand_name) Products (product_id, product_name, brand_id, category_id, model_year, list_price)...
-
Cullumber-Line Inc. (CLI) is a manufacturer that produces parts for residential telephones. Recent indications are that the market for this product is likely to continue to decline significantly and...
-
How does the intersection of race, class, and gender impact the identification, treatment, and punishment of deviant behaviors within criminal justice systems ?
-
4) Execute the following function call. y 14 a) drawCircle(5, 6, 2) 12 b) You choose a new point, (A, B), to be the center of a new circle. -10- c) drawCircle(A, B, 2) d) Choose your point, so that...
-
Tell whether the angles or sides are corresponding angles, corresponding sides, or neither. AC and JK
-
What is the period of a pendulum formed by placing a horizontal axis: (a) Through the end of a meter stick (100-cm mark)? (b) Through the 75-cm mark? (c) Through the 60-cm mark?
-
Some bats determine their distance to an object by detecting the difference in intensity between echoes. (a) If intensity falls off at a rate that is inversely proportional to the distance squared,...
-
The efficiency of an engine is 0.21. For every 1.00 kJ of heat absorbed by the engine, how much (a) Net work is done by it and (b) Heat is released by it?
-
Presented below are summary financial data from the General Electric Company 2015 annual report. Using the ratio definitions from Exhibit 4.6, calculate the following liquidity and solvency ratios:...
-
Presented below are summary financial data from the The Coca-Cola Company 2015 annual report. Using the ratio definitions from Exhibit 4.6, calculate the following liquidity and solvency ratios: cash...
-
Presented below are summary financial data from The Boeing Company 2015 annual report. Using the ratio definitions from Exhibit 4.6, calculate the following liquidity and solvency ratios: cash and...

Study smarter with the SolutionInn App



