3. An equilibrium mixture contains 1.0 mol of iron metal, Fe(s); 1.0 x 10 mol of...
Fantastic news! We've Found the answer you've been seeking!
Question:
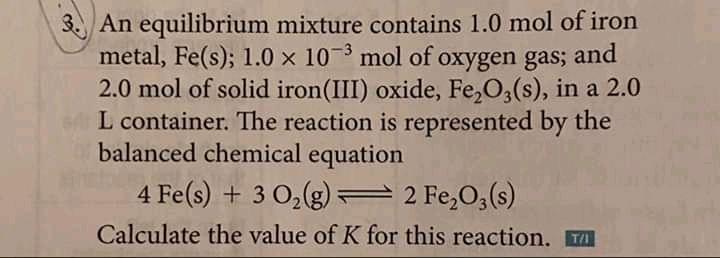
Transcribed Image Text:
3. An equilibrium mixture contains 1.0 mol of iron metal, Fe(s); 1.0 x 10 mol of oxygen gas; 2.0 mol of solid iron(III) oxide, Fe,O,(s), in a 2.0 L container. The reaction is represented by the balanced chemical equation and 4 Fe(s) + 3 0,(g) 2 Fe,O,(s) Calculate the value of K for this reaction. T 3. An equilibrium mixture contains 1.0 mol of iron metal, Fe(s); 1.0 x 10 mol of oxygen gas; 2.0 mol of solid iron(III) oxide, Fe,O,(s), in a 2.0 L container. The reaction is represented by the balanced chemical equation and 4 Fe(s) + 3 0,(g) 2 Fe,O,(s) Calculate the value of K for this reaction. T
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
A gas mixture of 1 kmol carbon monoxide, 1 k mol nitrogen, and 1 k mol oxygen at 25C, 150 kPa, is heated in a constant pressure SSSF process. The exit mixture can be assumed to be in chemical...
-
What is the shear capacity of the RC beam described below considering the steel reinforcement and using the formula: VRsyAw 2fyd cot 8/s The shear reinforcement in the beam is provided by sets of...
-
A metal cylinder with rigid walls contains 2.50 mol of oxygen gas. The gas is cooled until the pressure decreases to 30.0% of its original value. You can ignore the thermal contraction of the...
-
The comparative balance sheet of Merrick Equipment Co. for December 31, 20Y9 and 20Y8, is as follows: Dec. 31, 20Y9 Dec. 31, 20Y8 Assets Cash $70,720 $47,940 Accounts receivable (net) 207,230 188,190...
-
How would you search for potential businesses to buy? Would you use business broker, and why?
-
Describe the role of pictographs. What is the relationship between bar charts and histograms?
-
Write a computer program for finding numerically the mode shapes of thin fixed-simply supported beams by using the known values of the natural frequencies.
-
Jennifer has just been promoted to manager of the gear division of Machine Parts Co. The division, which manufactures gears for hydraulic drives, uses a standard cost system. The standard cost of a...
-
Puget Sound Divers provides diving services, such as underwater ship repairs, to its clients. The company's planning budget for May appears below: Puget Sound Divers Planning Budget For the Month...
-
Perfect Parties, Inc. has several divisions, one of which provides birthday parties at their facility, and has provided the actual and planning budget results for the month of June. The Controller...
-
Sally and Simon are in a pub. Sallys demand curve for cocktails is P= 30 2qc. Simons demand curve for cocktails is P = 40 3qf. If one cocktail costs $10, together how many cocktails do they buy?
-
Give a general description of PERT/CPM techniques.
-
Using real-world examples, evaluate the success of a country of your choice in achieving any two Sustainable Development Goals.
-
Using real-world examples, discuss the possible relationship between economic growth and economic development.
-
Murchison Technologies, Inc. recently developed a patient-billing software system that it markets to physicians and dentists. Jim Archer and Janice Johnson founded the company in Austin, Texas five...
-
Interview at least ten people and ask the following questions: What are your auto liability insurance limits and deductibles? If you own your own home, do you have an all-risk or named perils...
-
Fitness Fanatics is a regional chain of health clubs. The managers of the clubs, who have authority to make investments as needed, are evaluated based largely on return on Investment (ROI). The...
-
What is a content filter? Where is it placed in the network to gain the best result for the organization?
-
Give the structural formula for a portion of the chain polymer obtained by the reaction of ethylene glycol, CH2OHO CH2OH with malonic acid, HOOCCH2COOH.
-
You want to make up 3.00 L of aqueous hydrochloric acid, HCl(aq), that has a pH of 2.00. How many grams of concentrated hydrochloric acid will you need? Concentrated hydrochloric acid contains 37.2...
-
The photoelectric work function for magnesium is 5.90 1019 J. (The work function is the minimum energy needed to eject an electron from the metal by irradiating it with light.) Calculate the minimum...
-
A precision grinding machine (Fig. 3.53) is supported on an isolator that has a stiffness of \(1 \mathrm{MN} / \mathrm{m}\) and a viscous damping constant of \(1 \mathrm{kN}-\mathrm{s} /...
-
Derive the equation of motion and find the steady-state response of the system shown in Fig. 3.54 for rotational motion about the hinge \(O\) for the following data: \(k=5000 \mathrm{~N} /...
-
An air compressor of mass \(100 \mathrm{~kg}\) is mounted on an elastic foundation. It has been observed that, when a harmonic force of amplitude \(100 \mathrm{~N}\) is applied to the compressor, the...

Study smarter with the SolutionInn App



