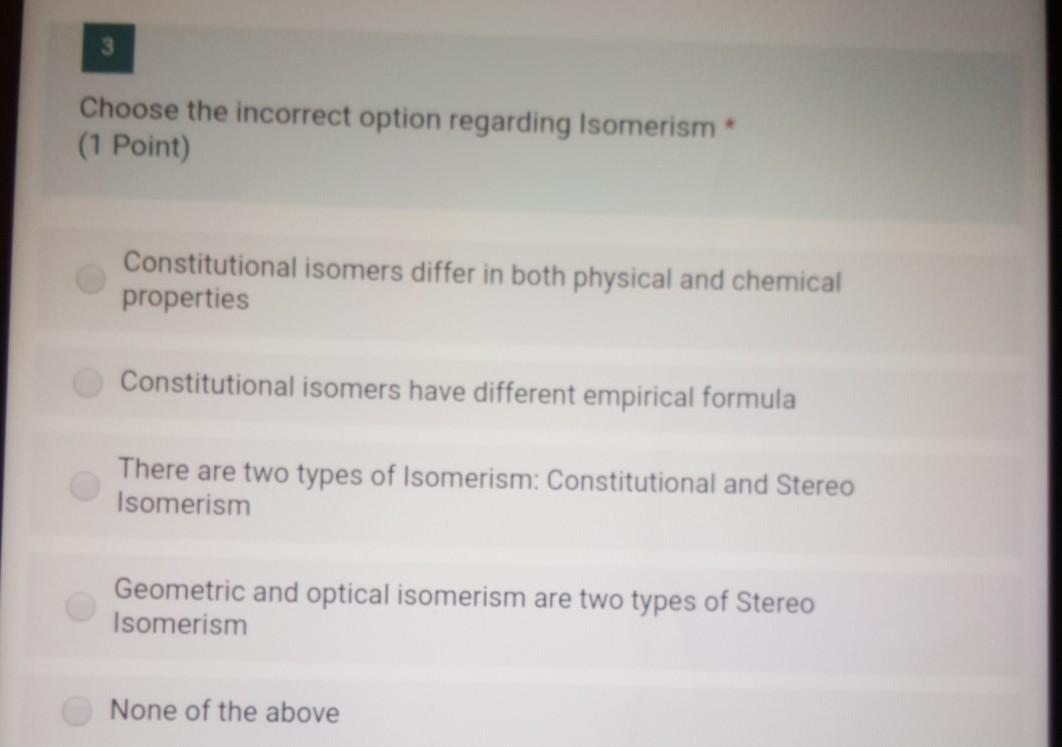
3 Choose the incorrect option regarding Isomerism * (1 Point) Constitutional isomers differ in both physical...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
3 Choose the incorrect option regarding Isomerism * (1 Point) Constitutional isomers differ in both physical and chemical properties Constitutional isomers have different empirical formula There are two types of Isomerism: Constitutional and Stereo Isomerism Geometric and optical isomerism are two types of Stereo Isomerism None of the above 3 Choose the incorrect option regarding Isomerism * (1 Point) Constitutional isomers differ in both physical and chemical properties Constitutional isomers have different empirical formula There are two types of Isomerism: Constitutional and Stereo Isomerism Geometric and optical isomerism are two types of Stereo Isomerism None of the above
Expert Answer:
Answer rating: 100% (QA)
Answer 8 The incorrect option regarding isomerism is b Constitutional isomers have different empirical formula Constitutional isomers have similar chemical properties ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
You go to the car dealer to have some service done on your car. Unfortunately, they find a lot of problems with the car. Today you will have to pay $2,000 to get the car fixed. Based on your estimate...
-
49. When developing a forecast and production plan; utilization refers to: a) The minimum number of labour hours required to produce full capacity b) The total number of units in the production plan...
-
There are two types of electric pencil-sharpener producers. High-quality manufacturers produce very good sharpeners that consumers value at $14. Low-quality manufacturers produce less good ones that...
-
What would a profile look like across a restraining bend? Releasing bend?
-
For 2010, its first year of operations, Quinn Advertising, Inc. earned pretax accounting income (on the income statement) of $375,000. Taxable income (on the tax return filed with the Internal...
-
Is a randomly generated 4-CNF sentence with n symbols and m clauses more or less likely to be solvable than a randomly generated 3-CNF sentence with n symbols and m clauses? Explain.
-
Sherene Lynch opened a medical practice titled Sherene Lynch M.D., Inc. During January, the first month of operations, the business experienced the following events: Requirement 1. Analyze the...
-
Does Monsanto maintain an ethical culture that can effectively respond to various stakeholders ? The original Monsanto was very different from the current company. It was started by John F. Queeny in...
-
Technology has an impact on society and individuals, even if we don't realize it because we're so immersed in our technology-centric society. Technology has also changed the way people interact in an...
-
San Francisco led the movement to ban plastic shopping bags and is now proposing to ensure people have access to safe high-quality tap water and ban bottled water. A supporter of the ban says The...
-
Find the area K of the triangle specified below. a =5, b= 16, c= 19 The area K is square units.
-
Jeff Krause purchased 1,000 shares of a speculative stock in January for $1.93 per share. Six months later, he sold them for $10.35 per share. He uses an online broker that charges him $10.00 per...
-
A new blu-ray player costs $94.99 in the store. What would your total cost be if the sales tax is 4.5 % ? Round your answer to the nearest cent, if necessary.
-
Have you ever witnessed ineffective vs. effective delegation while you've been at a job? In a short paragraph, describe the situation and reflect what behaviors made it ineffective or effective. (5...
-
Ha Magan is saving money and plans on making quarterly contributions into an account earning an annual interest rate of 6% compounded quarterly. If Magan would like to end up with $49,000 after 6...
-
Suppose that a hollow cylindrical object is floating on the surface of water. This object has a mass of 300g and is floating such that 4cm of its height is submerged under the surface of water, while...
-
Simon Company's year-end balance sheets follow. At December 31 Assets Cash Accounts receivable, net Merchandise inventory Prepaid expenses Plant assets, net Total assets Liabilities and Equity...
-
A condenser (heat exchanger) brings 1 kg/s water flow at 10 kPa quality 95% to saturated liquid at 10 kPa, as shown in Fig. P4.91. The cooling is done by lake water at 20C that returns to the lake at...
-
Suppose you have just synthesized heptanoic acid from heptan-1-ol. The product is contaminated by sodium dichromate, sulfuric acid, heptan-1-ol, and possibly heptanal. Explain how you would use...
-
There are two different ways of making 2-ethoxyoctane from octan-2-ol using the Williamson ether synthesis. When pure 1-2-octan-2-ol of specific rotation -8.24 is treated with sodium metal and then...
-
Step 2 of the iodination of benzene shows water acting as a base and removing a proton from the sigma complex. We did not consider the possibility of water acting as a nucleophile and attacking the...
-
An automobile's suspension system determines the vehicle's response to inputs from uneven road surfaces. Figure 3.56 shows a drawing of a suspension system that includes a coil spring, a shock...
-
For a mass-spring-damper system under harmonic loading per unit mass \(F(t) / m=\cos \omega t \mathrm{ft} / \mathrm{s}^{2}\), solve for the response amplitude for the case where \(k=\) \(20...
-
For the system in Figure 3.57, the forcing per unit mass is \(F(t) / m=10 \sin 15 t \mathrm{ft} / \mathrm{s}^{2}\). Solve for the transient response, the steady-state response, and the constants of...

Study smarter with the SolutionInn App


