3. For the following transformations draw the product in the box below. Draw only one stereoisomer...
Fantastic news! We've Found the answer you've been seeking!
Question:
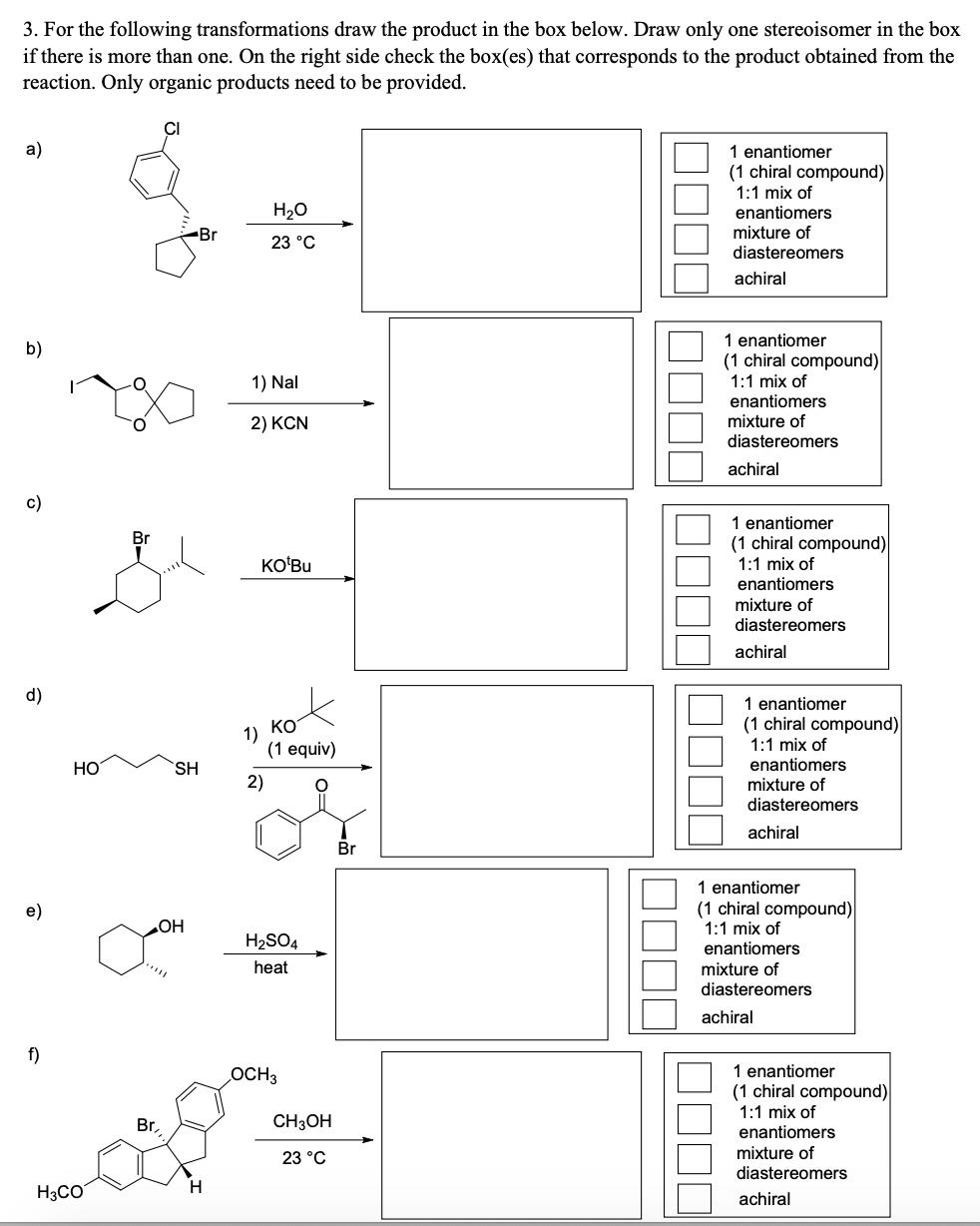
Transcribed Image Text:
3. For the following transformations draw the product in the box below. Draw only one stereoisomer in the box if there is more than one. On the right side check the box(es) that corresponds to the product obtained from the reaction. Only organic products need to be provided. a) 1 enantiomer (1 chiral compound) 1:1 mix of H20 enantiomers Br mixture of 23 °C diastereomers achiral 1 enantiomer b) (1 chiral compound) 1) Nal 1:1 mix of enantiomers mixture of 2) KCN diastereomers achiral c) 1 enantiomer Br (1 chiral compound) 1:1 mix of KOʻBU enantiomers mixture of diastereomers achiral d) 1 enantiomer (1 chiral compound) 1:1 mix of enantiomers mixture of KO 1) (1 equiv) HO SH 2) diastereomers achiral Br 1 enantiomer e) (1 chiral compound) 1:1 mix of HO H2SO4 enantiomers heat mixture of diastereomers achiral f) OCH3 1 enantiomer (1 chiral compound) 1:1 mix of Br, CH3OH enantiomers mixture of diastereomers 23 °C H3CO H. achiral 3. For the following transformations draw the product in the box below. Draw only one stereoisomer in the box if there is more than one. On the right side check the box(es) that corresponds to the product obtained from the reaction. Only organic products need to be provided. a) 1 enantiomer (1 chiral compound) 1:1 mix of H20 enantiomers Br mixture of 23 °C diastereomers achiral 1 enantiomer b) (1 chiral compound) 1) Nal 1:1 mix of enantiomers mixture of 2) KCN diastereomers achiral c) 1 enantiomer Br (1 chiral compound) 1:1 mix of KOʻBU enantiomers mixture of diastereomers achiral d) 1 enantiomer (1 chiral compound) 1:1 mix of enantiomers mixture of KO 1) (1 equiv) HO SH 2) diastereomers achiral Br 1 enantiomer e) (1 chiral compound) 1:1 mix of HO H2SO4 enantiomers heat mixture of diastereomers achiral f) OCH3 1 enantiomer (1 chiral compound) 1:1 mix of Br, CH3OH enantiomers mixture of diastereomers 23 °C H3CO H. achiral
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
(A) (B) (C) For the following transformations please predict the products and provide transition state rationale for the predicted selectivity. (D) (E) OH Ph VO(acac)2 TBHP 3 mol % R.R-Mn-Salen-Cl...
-
Draw the following signals a)x (t) = -5 rect (3) b)x(t) = 2sgn () (u(t 2) u(t + 3)) - - c) x(n)= = Sinc(t )e-lt| d)Given the following sequences x(n)= {2, -2, 1,0.2,3} draw the signal...
-
Draw either one of the enantiomers for each chiral molecule in Problem 34 and label its stereocenter as R or S. Problem 34 OH OH (d)OH OH OH
-
Why does the cost to load in an unconstrained system is lower compared to contrained with N - 1 Contingency in PLEXOS Modelling? What are the factors that affect the increase in cost to load during...
-
If Susan overpays her state income tax due to excess withholdings, can she deduct the entire amount in the year withheld? When Susan receives a refund from the state how must she treat that refund...
-
Consider the pdf for the rv X = magnitude (in newtons) of a dynamic load on a bridge, given in Example 4.7: Write a program to simulate values from this distribution using the inverse cdf method....
-
Eco-efficiency provides a measure of winwinan action is undertaken if it saves money and environmental interventions are reduced. Is eco-efficiency too concerned about saving money rather than...
-
Kawmin is a small country that produces and consumes jelly beans. The world price of jelly beans is $1 per bag, and Kawmins domestic demand and supply for jelly beans are governed by the following...
-
If the average return on common stocks was 13.3%, the average Treasury bill rate was 3.8%, and the average inflation rate was 3.2% what would be the expected nominal and approximate real market...
-
Analyzing, Forecasting, and Interpreting Both Income Statement and Balance Sheet Following are the income statements and balance sheets of Best Buy Co., Inc. Income Statement, Feb. 26, 2011 Feb. 27,...
-
Question 5 (4 points) P. A cylinder made of many different materials is loaded as shown. P = 9.00 kN Increase in & = 0.0019 O m Decrease in L = 0.031 O mm & = 55.0 mm L = 337 !3! mm Determine...
-
Equipment is to be depreciated using the units of use method with a preset unit total of 11 000. The equipment was in production from: 1 December 2021 to 30 November 2022, producing 2500 units 1...
-
Hemophilia is an X-linked recessive disorder in humans. If a heterozygous woman has children with an unaffected man, what is the probability of each of the following combinations of offspring? A. An...
-
Demand is elastic when ______. a) percentage change in price is greater than percentage change in quantity b) percentage change in quantity is greater than percentage change in price c) the demand...
-
Empire Electric Company (EEC) uses only debt and common equity. It can borrow unlimited amounts at an interest rate of rd = 9% as long as it finances at its target capital structure, which calls for...
-
\(\$ 20\) million of nondealer real estate obligations arose in and are outstanding at the end of calendar-year 2018. Deferred gross profit on such installment obligations equals \(\$ 5\) million....
-
Year Quantities Produced Prices CDs Tennis Rackets $/CD $/Tennis Racket Nominal GDP Real GDP Price Index 2011 2012 100 200 $20 $110 120 210 $22 $120 A) What is nominal GDP for year 2011? (Hint:...
-
A crop-dusting plane flies over a level field at a height of 25 ft. If the dust leaves the plane through a 30 angle and hits the ground after the plane travels 75 ft, how wide a strip is dusted? See...
-
A 5.00-g sample of aluminum pellets (specific heat capacity = 0.89 JoC-1g-1) and a 10.00-g sample of iron pellets (specific heat capacity = 0.45 JoC-1g-1) are heated to 100.0oC. The mixture of hot...
-
High- quality audio amplifiers generate large amounts of heat. To dissipate the heat and prevent damage to the electronic devices, manufacturers use heat- radiating metal fins. Would it be better to...
-
Determine the forms of the integrated and the differential rate laws for the decomposition of benzene diazonium chloride C6H5N2Cl(aq) C6H5Cl(l) + N2(g) From the following data, which were collected...
-
Refer to exercise E2-28 of Chapter 2. Start from the trial balance and the posted T-accounts prepared at January 18, 2020. Later in January, the business completed these transactions: Requirements 1....
-
Refer to exercise E3-31. Requirements 1. After solving exercise E3-30, use the data in that exercise to prepare Yosaf Portraits Ltd.'s classified balance sheet at December 31, 2020. Use the report...
-
Satterfield Corporation reported the following current accounts at Deccmber 31, 2019 (amounts in thousands): During January 2020, Satterfield completed these selected transactions: - Sold services on...

Study smarter with the SolutionInn App


