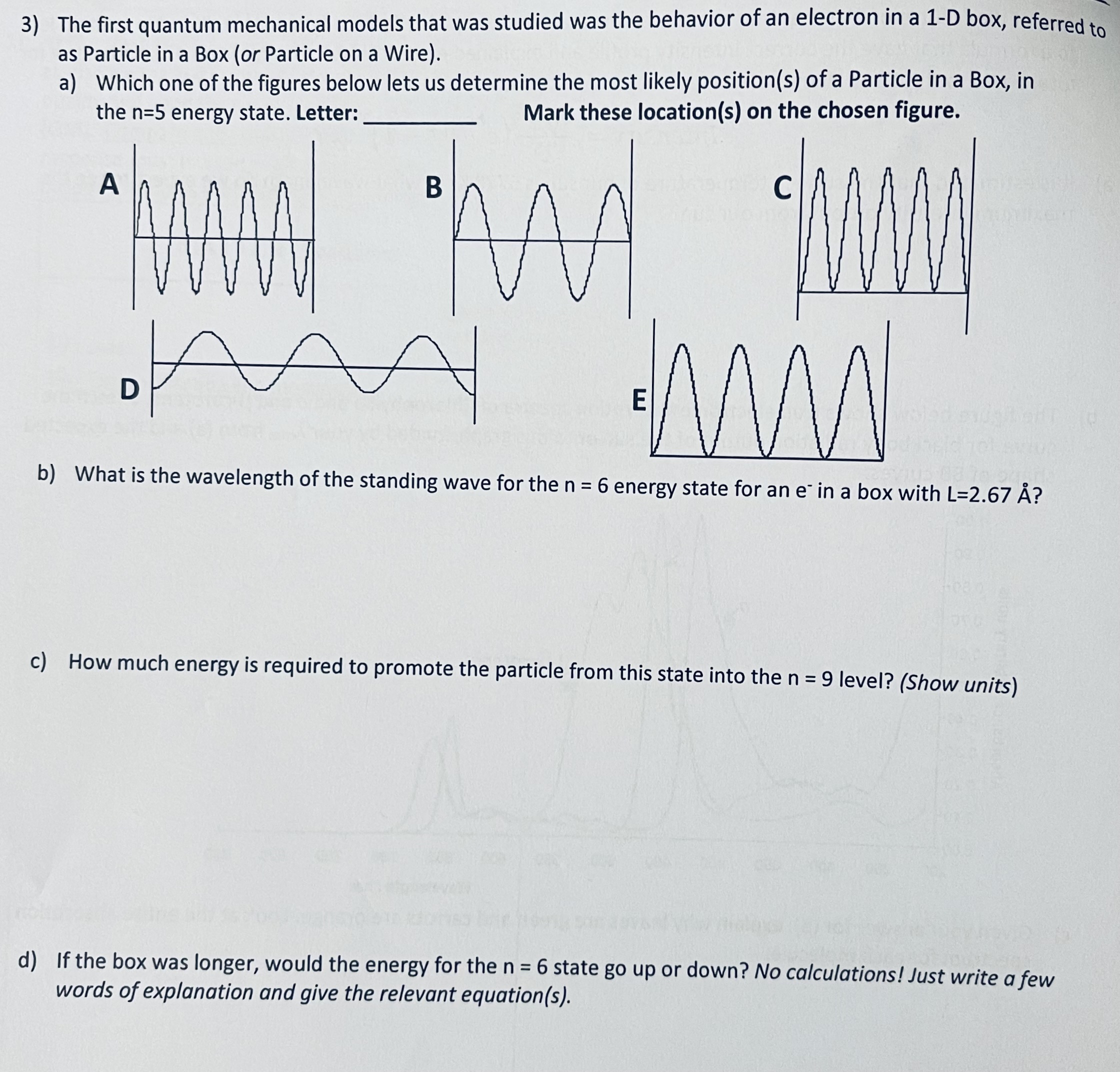
3) The first quantum mechanical models that was studied was the behavior of an electron in...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
3) The first quantum mechanical models that was studied was the behavior of an electron in a 1-D box, referred to as Particle in a Box (or Particle on a Wire). a) Which one of the figures below lets us determine the most likely position(s) of a Particle in a Box, in Mark these location(s) on the chosen figure. the n=5 energy state. Letter: A * B D AN mw b) What is the wavelength of the standing wave for the n = 6 energy state for an e in a box with L=2.67 ? E c) How much energy is required to promote the particle from this state into the n = 9 level? (Show units) d) If the box was longer, would the energy for the n = 6 state go up or down? No calculations! Just write a few words of explanation and give the relevant equation(s). 3) The first quantum mechanical models that was studied was the behavior of an electron in a 1-D box, referred to as Particle in a Box (or Particle on a Wire). a) Which one of the figures below lets us determine the most likely position(s) of a Particle in a Box, in Mark these location(s) on the chosen figure. the n=5 energy state. Letter: A * B D AN mw b) What is the wavelength of the standing wave for the n = 6 energy state for an e in a box with L=2.67 ? E c) How much energy is required to promote the particle from this state into the n = 9 level? (Show units) d) If the box was longer, would the energy for the n = 6 state go up or down? No calculations! Just write a few words of explanation and give the relevant equation(s).
Expert Answer:
Answer rating: 100% (QA)
The image youve provided shows a question related to the Particle in a Box model from Quantum Mechanics Here are the answers to the questions presente... View the full answer

Related Book For 

Posted Date:
Students also viewed these physics questions
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Let A, B be sets. Define: (a) the Cartesian product (A B) (b) the set of relations R between A and B (c) the identity relation A on the set A [3 marks] Suppose S, T are relations between A and B, and...
-
0.1 M CH3COOH with 0.1 M NH3. The relative conductivities of the solutions tested are as follows: 0.1 M CH3COOH/0.1 M NH3 measured in parallel: (b) 0.1 M CH,COOH with 0.1 M NH,. The relative...
-
Explain the term bounded rationality.
-
Is it better to use more or fewer years when performing a trend analysis? Why?
-
Extend the putcall parity relationship of Eq. (13.4) to the case in which the underlying stock share pays dividends, with present value D, between now and the maturity of the two options. Data From...
-
Holden Graham started The Graham Co., a new business that began operations on May 1. The Graham Co. completed the following transactions during its first month of operations. May 1 H. Graham invested...
-
T Corporation begin business May 1 , 2 0 2 3 and elected to end its tax year December 3 1 . The corporation s taxable income for the short period May 1 , 2 0 2 3 through December 3 1 , 2 0 2 3 was $...
-
Daniel B. Butler and Freida C. Butler, husband and wife, file a joint return. The Butlers live at 625 Oak Street in Corbin, KY 40701. Dan's Social Security number is 111-11-1112, and Freida's is...
-
Herb Marks built his enterprise on the faithful patronage of four specialty shops and a large contract from Elmore Distributors. But after two years, the maker of novelty pens and pencils had to...
-
Using these data from the comparative balance sheet of Oriole Spice Company is presented below. Prepare a schedule showing a horizontal analysis for 2021 using 2020 as the base year. (Round...
-
Statement of cost of goods manufactured for a manufacturing company Cost data for Johnstone Manufacturing Company for the month ended March 31 are as follows: Inventories Materials Work in process...
-
Gunco Industries invests a large sum of money in R&D; as a result, it retains and reinvests all of it earnings. In other words, Gunco does not pay any dividends and it has no plans to pay dividends...
-
Mariota Corp. just paid a dividend of $3.90 per share on its stock. The dividend growth rate is expected to be 3.6 forever and investors require a return of 12.8 percent on this stock. What will the...
-
Red Shoe Co. has concluded that additional equity financing will be needed to expand operations and that the needed funds will be best obtained through a rights offering. It has correctly determined...
-
Analyze the polynomial function f(x) = x + 0.3x - 1.4988x - 0.27285. Determine the end behavior of the graph of the function. The graph of f behaves like y = for large values of x].
-
1. Using the information from Problem 16-4B, prepare a statement of cash flows for Lim Garden Supplies Inc. using the direct method of presenting cash flows from operating activities. 2. How does Lim...
-
The magnetic flux through a single-loop coil is given by Figure 23-33. (a) Is the magnetic flux at t = 0.25 s greater than, less than, or the same as the magnetic flux at t = 0.55 s? Explain. (b) Is...
-
A mass moves back and forth in simple harmonic motion with amplitude A and period T. (a) In terms of T, how much time does it take for the mass to move through a total distance of 2A? (b) How much...
-
To tune middle C on a piano, a tuner hits the key and at the same time sounds a 261-Hz tuning fork. If the tuner hears 3 beats per second, what are the possible frequencies of the piano key?
-
How did the trade winds help traders in colonial America?
-
Do surface currents affect water temperature, air temperature, or both?
-
What drives surface currents?

Study smarter with the SolutionInn App


