3. The mass spectrum of 2-heptanone is shown below. a) Draw structures for the fragment ions...
Fantastic news! We've Found the answer you've been seeking!
Question:
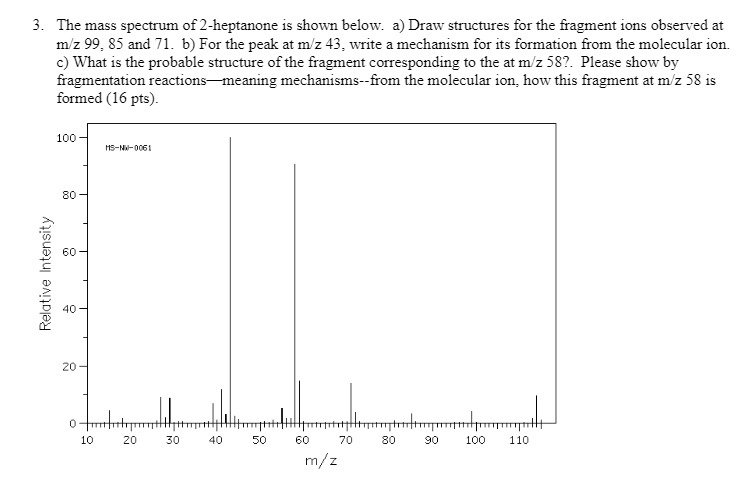
Transcribed Image Text:
3. The mass spectrum of 2-heptanone is shown below. a) Draw structures for the fragment ions observed at m/z 99, 85 and 71. b) For the peak at m/z 43, write a mechanism for its formation from the molecular ion. c) What is the probable structure of the fragment corresponding to the at m/z 587. Please show by fragmentation reactions meaning mechanisms--from the molecular ion, how this fragment at m/z 58 is formed (16 pts). 100 80 8 Relative Intensity 40 20 0 10 MS-NW-0061 20 30 40 50 60 70 m/z 80 Jump 90 100 110 3. The mass spectrum of 2-heptanone is shown below. a) Draw structures for the fragment ions observed at m/z 99, 85 and 71. b) For the peak at m/z 43, write a mechanism for its formation from the molecular ion. c) What is the probable structure of the fragment corresponding to the at m/z 587. Please show by fragmentation reactions meaning mechanisms--from the molecular ion, how this fragment at m/z 58 is formed (16 pts). 100 80 8 Relative Intensity 40 20 0 10 MS-NW-0061 20 30 40 50 60 70 m/z 80 Jump 90 100 110
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The molecular ion region in the mass spectrum of a large molecule, such as a protein, consists of a cluster of peaks differing by 1 Da. This pattern occurs because a molecule with many atoms has a...
-
10 20 30 40 50 60 70 Mancom 10 20 30 40 50 60 70 Macom B Ages of group of patients: The youngest participant has: Type of the graph: Ages of group of patients: The oldest participant has: Y/O Y/O ...
-
The mass spectrum of unknown compound A shows a molecular ion at m/z 116 and prominent peaks at m/z 87 and m/z 101. Its UV spectrum shows no maximum above 200 nm. The IR and NMR spectra of A follow....
-
Let B > 0. Show that the maximum of subject to the constraints x + this to conclude that f(x,...,xn) = X1 X2 Xn for all positive numbers a,..., an. . + xn = B and xj 0 for j = 1,. n occurs for x =...
-
Celtic Imports is an importer of silver, brass, and furniture items from Ireland. Eileen Sullivan is the general manager of Celtic Imports. Sullivan employs two other people in the business. Mary...
-
Refer to the RMO CSMS Marketing subsystem shown in Figure 3-10. Draw a use case diagram that shows all actors and all use cases. Use a drawing tool such as Microsoft Visio if it is available. Use...
-
Predict the value of y when x1 = 20, x2 = 20, and x3 = 30. Exercises 1115 refer to the following data set: x1 *2 x3 69.8 7.9 37.3 62.4 32.3 9.3 20.2 40.7 66.9 13.3 30.5 48.7 87.5 27.4 38.8 35.8 93.5...
-
Comparative balance sheets for 2013 and 2012, a statement of income for 2013, and additional information from the accounting records of Red, Inc., are provided below. Additional information from the...
-
The Ontario Line will be a 15.6-kilometre subway line that will make it faster and easier to travel within Toronto and beyond. The line will bring 15 new stations to the city and will run from...
-
(c) A Michelson interferometer is used to measure the displacement of a linearly moving object. The target mirror is mounted on the moving object. The laser source used in this system is laser diode...
-
What is the binary search tree rank of node E? What is the binary search tree successor of node E? What is the depth of node E? H D I B J E A F C G
-
Accounts Payable had a normal beginning balance of $1,200. During the period, there were debit postings of $300 and credit postings of $800. What was the ending balance?
-
Compute the total manufacturing cost for a manufacturer with the following information for the month. Raw materials purchased $ 3 6 , 0 0 0 Direct materials used 6 2 , 7 5 0 Direct labor used 1 2 , 4...
-
Divide: 32m-72m-56m 8m3
-
Swift Corporation has a beta of 1 . 2 5 . Based on current market conditions, the required return on the Swift Corporation's common stock is 1 1 . 0 % . Global events have introduced greater risk...
-
Iz Rizky Printing (IRP) uses a job costing system for all its products, through two production departments, Editing Department and the Printing Department before being transferred as a finished...
-
A hollow tube chimes are made of metal and are open at each end. these columns resonate best at their third resonant length. One chime is 2.50m long and the air temperature is 25 Celsius. what is the...
-
Explain what is meant by vicarious liability and when it is available?
-
In Section 9-7B, we saw how acetylide ions add to carbonyl groups in much the same way as Grignard and organolithium reagents. Acetylide ions also add to ethylene oxide much like Grignard and...
-
Show the rest of the mechanism for formation of the cyclized intermediate in Figure 14-6. squalene epoxidase (enzyme) HC CH H CH HC CH squalene squalene-2.3-epoxide cyclized intermediate CH . CH H,C...
-
Predict the products of the following reactions. (a) Phenol + acetic anhydride (b) Phenol + acetic formic anhydride (c) Aniline + phthalic anhydride (d) Anisole + succinic anhydride and aluminum...
-
Should government regulation of sport and business be increased, be decreased, or remain the same? Defend your position.
-
What guides your behavior now? Will you use one of the ethical guides from the text? If yes, which one and why?
-
Can ethics be taught and learned? Defend your position.

Study smarter with the SolutionInn App



