3. You ran column chromatography and collected a fraction each minute for 25 minutes, then you...
Fantastic news! We've Found the answer you've been seeking!
Question:
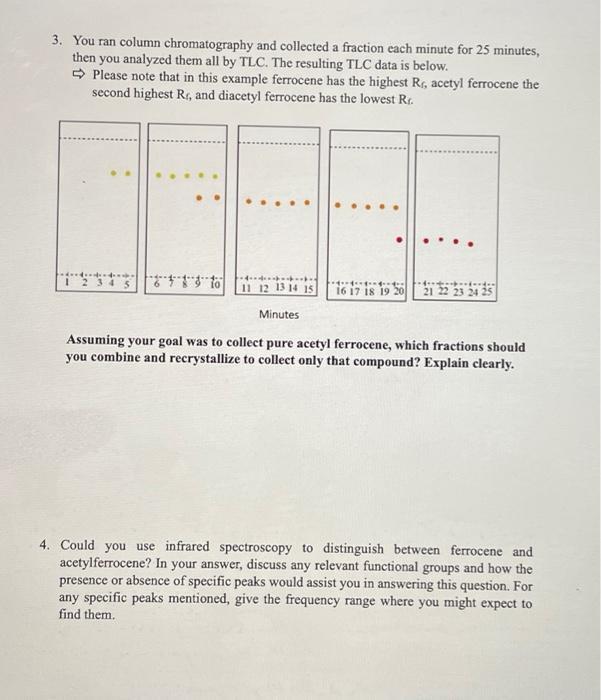
Transcribed Image Text:
3. You ran column chromatography and collected a fraction each minute for 25 minutes, then you analyzed them all by TLC. The resulting TLC data is below. Please note that in this example ferrocene has the highest Rr, acetyl ferrocene the second highest Rr, and diacetyl ferrocene has the lowest Rr. 4 11 12 13 14 15 Minutes e 16 17 18 19 20 Assuming your goal was to collect pure acetyl ferrocene, which fractions should you combine and recrystallize to collect only that compound? Explain clearly. 4. Could you use infrared spectroscopy to distinguish between ferrocene and acetylferrocene? In your answer, discuss any relevant functional groups and how the presence or absence of specific peaks would assist you in answering this question. For any specific peaks mentioned, give the frequency range where you might expect to find them. 3. You ran column chromatography and collected a fraction each minute for 25 minutes, then you analyzed them all by TLC. The resulting TLC data is below. Please note that in this example ferrocene has the highest Rr, acetyl ferrocene the second highest Rr, and diacetyl ferrocene has the lowest Rr. 4 11 12 13 14 15 Minutes e 16 17 18 19 20 Assuming your goal was to collect pure acetyl ferrocene, which fractions should you combine and recrystallize to collect only that compound? Explain clearly. 4. Could you use infrared spectroscopy to distinguish between ferrocene and acetylferrocene? In your answer, discuss any relevant functional groups and how the presence or absence of specific peaks would assist you in answering this question. For any specific peaks mentioned, give the frequency range where you might expect to find them.
Expert Answer:
Answer rating: 100% (QA)
Sol Since our will collect green dots denote acetyl ferrocene and G 11... View the full answer

Related Book For 

Introduction To Federal Income Taxation In Canada
ISBN: 9781554965021
33rd Edition
Authors: Robert E. Beam, Stanley N. Laiken, James J. Barnett
Posted Date:
Students also viewed these chemistry questions
-
List three specific parts of the Case Guide, Objectives and Strategy Section (See below) that you had the most difficulty understanding. Describe your current understanding of these parts. Provide...
-
"internet radios" for streaming audio, and personal video recorders and players. Describe design and evaluation processes that could be used by a start-up company to improve the usability of such...
-
All of the following companies manufacture toasters. The following costs were incurred by each company during September: Required: 1. What is the total conversion cost for Warner Co.? 2. What is the...
-
In the market for coffee, the elasticity of demand is estimated to be -0.6 and the elasticity of supply is estimated to be 1.2. If the government imposes a $1 tax on each cup of coffee sold, what...
-
Considering A-L to represent the major products formed in each of the following reactions, provide a structure for each of A through L. If more than one product can reasonably be conceived from a...
-
The Howell Corporation has the following account balances (in millions): Prepare an income statement and a supporting schedule of cost of goods manufactured for the year ended December 31, 2017. (For...
-
Last year (2013), Richter Condos installed a mechanized elevator for its tenants. The owner of the company, Ron Richter, recently returned from an industry equipment exhibition where he watched a...
-
Compare and contrast online transaction processing (OLTP) and online analytical processing (OLAP) databases.
-
Cardinal Company is considering a five-year project that would require a $2,975,000 investment in equipment with a useful life of five years and no salvage value . The company's discount rate is 14%....
-
Cheyenne Corp. issued 1,400 9%, 5-year, $1,000 bonds dated January 1, 2022, at face value. Interest is paid each January 1. (a) Prepare the journal entry to record the sale of these bonds on January...
-
If the authors of some essays that they wrote were alive today, would they still believe their original arguments regarding the Constitution, the size of the United States, and the ability of a...
-
Perfect Competition is a model of which examples are few and far between. Yet economists love to discuss this model. Explain why.
-
If the exchange rate is defined by AUD/USD. According to the purchasing power parity, how does the exchange rate change if the price level in Australia increases relative to that in the US? Explain...
-
To cater to the growing demand for luxury carpets, Germany agrees to buy 20,000 carpets from Plush Floors Inc. in exchange for 500 barrels of oil. Since Plush Floors Inc. is not seen as creditworthy,...
-
One of the criticisms of oligopolies is the adverse impacts these firms have on income distribution. Do you believe that is a valid criticism? Discuss with appropriate examples.
-
H H 3. H-C-C-0H k-C-H H H 3 Figone 1-6 In figure 1.6, which compound will have the highest boiling point and highest vapor pressure respectively? O 2 and 4 O 1 and 2 O 4 and 2 O 3 and 4
-
Ex. (17): the vector field F = x i-zj + yz k is defined over the volume of the cuboid given by 0x a,0 y b, 0zc, enclosing the surface S. Evaluate the surface integral ff, F. ds?
-
Mrs. Diet owns all 100 common shares of Low-Cal Caterers Inc. (Low-Cal), a Canadian-controlled private corporation with a December 31 year-end. Low-Cal was incorporated in 1983 when Mrs. Diet...
-
Pamela is 38 years old and lives in Welland, Ontario. She commutes daily to Niagara University, in Niagara Falls, New York. She is completing a full-time Masters in Education that is 12 consecutive...
-
Ed Sigmond was transferred from England to Ottawa by Pharmadyne Supplies Inc. on April 1, to assume the permanent position as Vice President, Canadian operations. Ed was a permanent resident of...
-
Management is considering three alternatives to satisfy an urgent need. Each of the alternatives will completely satisfy the need, so no combinations have to be considered. The first costs, operating...
-
Consider the following cash flow profile and assume MARR is 10 percent/year and the finance rate is 4 percent/year. a. Determine the MIRR for this project. b. Is this project economically attractive?...
-
Consider the following cash flow profile and assume MARR is 10 percent/year and the finance rate is 4 percent/ year. a. Determine the MIRR for this project. b. Is this project economically...

Study smarter with the SolutionInn App


